Epidemic Trends
CDC uses data from emergency department visits to model epidemic trends. This model helps tell whether the number of new respiratory infections is growing or declining in your state. While this model tells us the trend, it does not tell us the actual number of current infections with SARS-CoV-2 (the virus that causes COVID-19), influenza virus, or RSV. Refer to data notesfor more details.

Severe Viral Respiratory Illness
JAN. 9, 2026
ABOUT
Provides an update on how respiratory viruses are contributing to serious health outcomes, like hospitalizations and deaths.
This Week’s Illness Severity Update:
Reported on Friday, January 9, 2026
CDC uses data from emergency department visits to model epidemic trends. This model helps tell whether the number of new respiratory infections is growing or declining in your state. While this model tells us the trend, it does not tell us the actual number of current infections with SARS-CoV-2 (the virus that causes COVID-19), influenza virus, or RSV. Refer to data notesfor more details.
Severe Viral Respiratory Illness
JAN. 9, 2026
ABOUT
Provides an update on how respiratory viruses are contributing to serious health outcomes, like hospitalizations and deaths.
This Week’s Illness Severity Update:
Reported on Friday, January 9, 2026
- Influenza hospitalizations are elevated.
- Hospitalizations for RSV are elevated.
- COVID-19 hospitalizations are low.
- Deaths associated with COVID-19 (0.3%) and influenza (0.2%) are low. Deaths associated with RSV are very low. However, these respiratory death data are current as of the week ending December 6, 2025, and will be updated as soon as new data are available.


 )
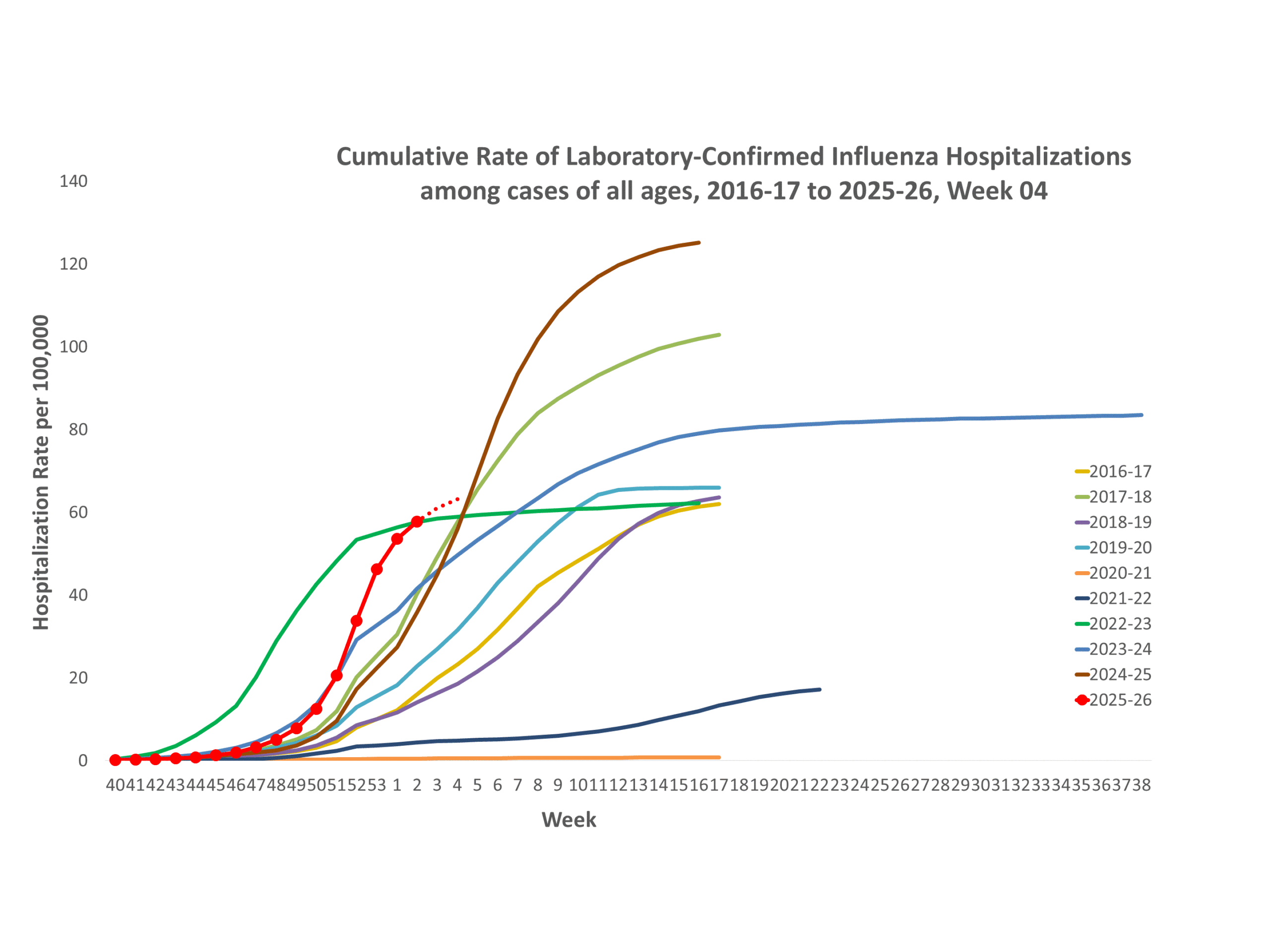
) ) weekly hospitalization rate
) weekly hospitalization rate Influenza Positive Tests Reported to CDC by Clinical Laboratories, National Summary, 2025-26 Season, week ending Jan. 24, 2026
Influenza Positive Tests Reported to CDC by Clinical Laboratories, National Summary, 2025-26 Season, week ending Jan. 24, 2026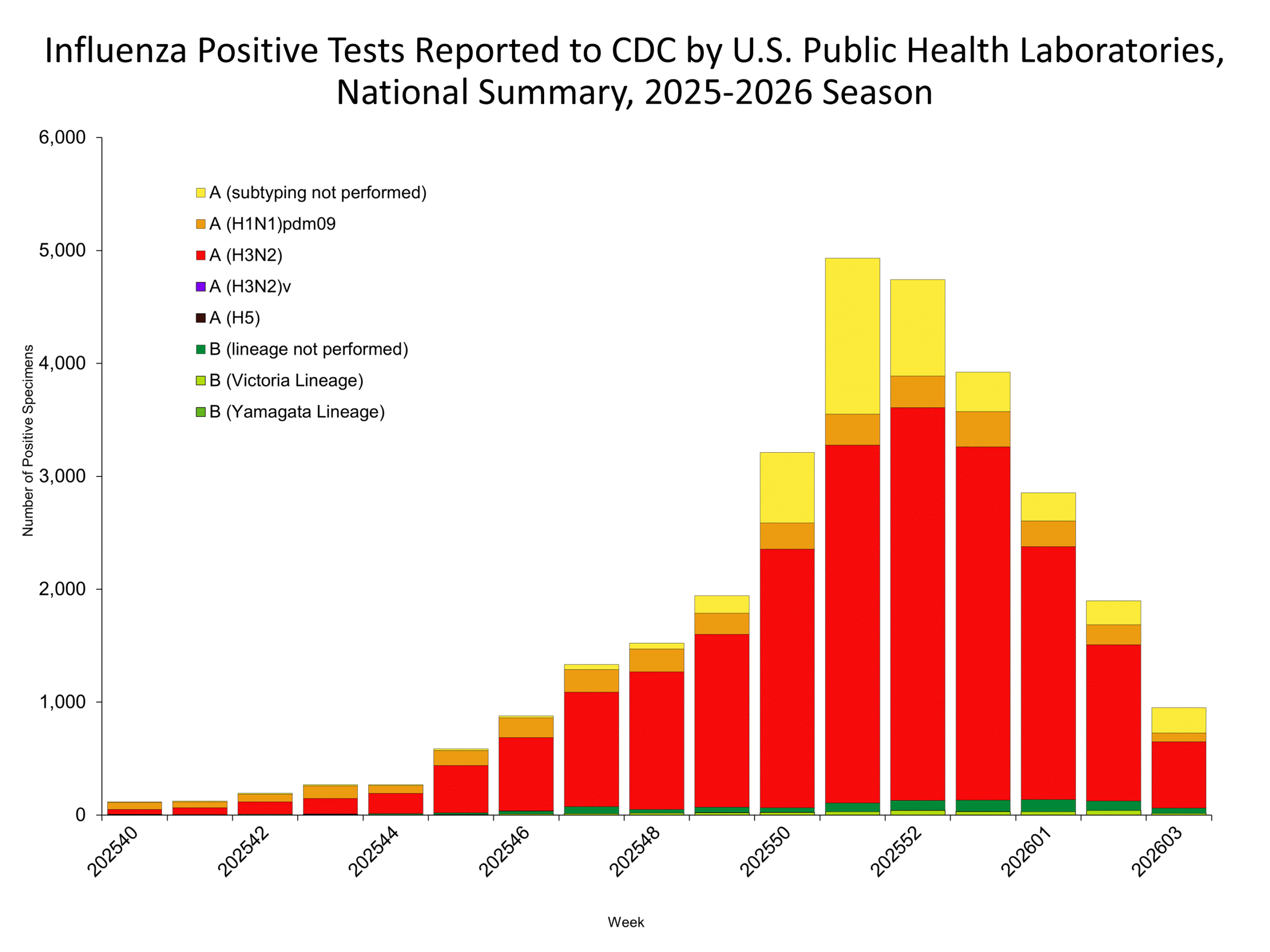 This graph reflects the number of specimens tested and the number determined to be positive for influenza viruses at the public health lab (specimens tested is not the same as cases). It does not reflect specimens tested only at CDC and could include... Show More
This graph reflects the number of specimens tested and the number determined to be positive for influenza viruses at the public health lab (specimens tested is not the same as cases). It does not reflect specimens tested only at CDC and could include... Show More Percentage of Outpatient Visits for Respiratory Illness Reported by. The U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet)
Percentage of Outpatient Visits for Respiratory Illness Reported by. The U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet) Percent of Outpatient Visits for Respiratory Illness by Age Group. Reported by the U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet)
Percent of Outpatient Visits for Respiratory Illness by Age Group. Reported by the U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet)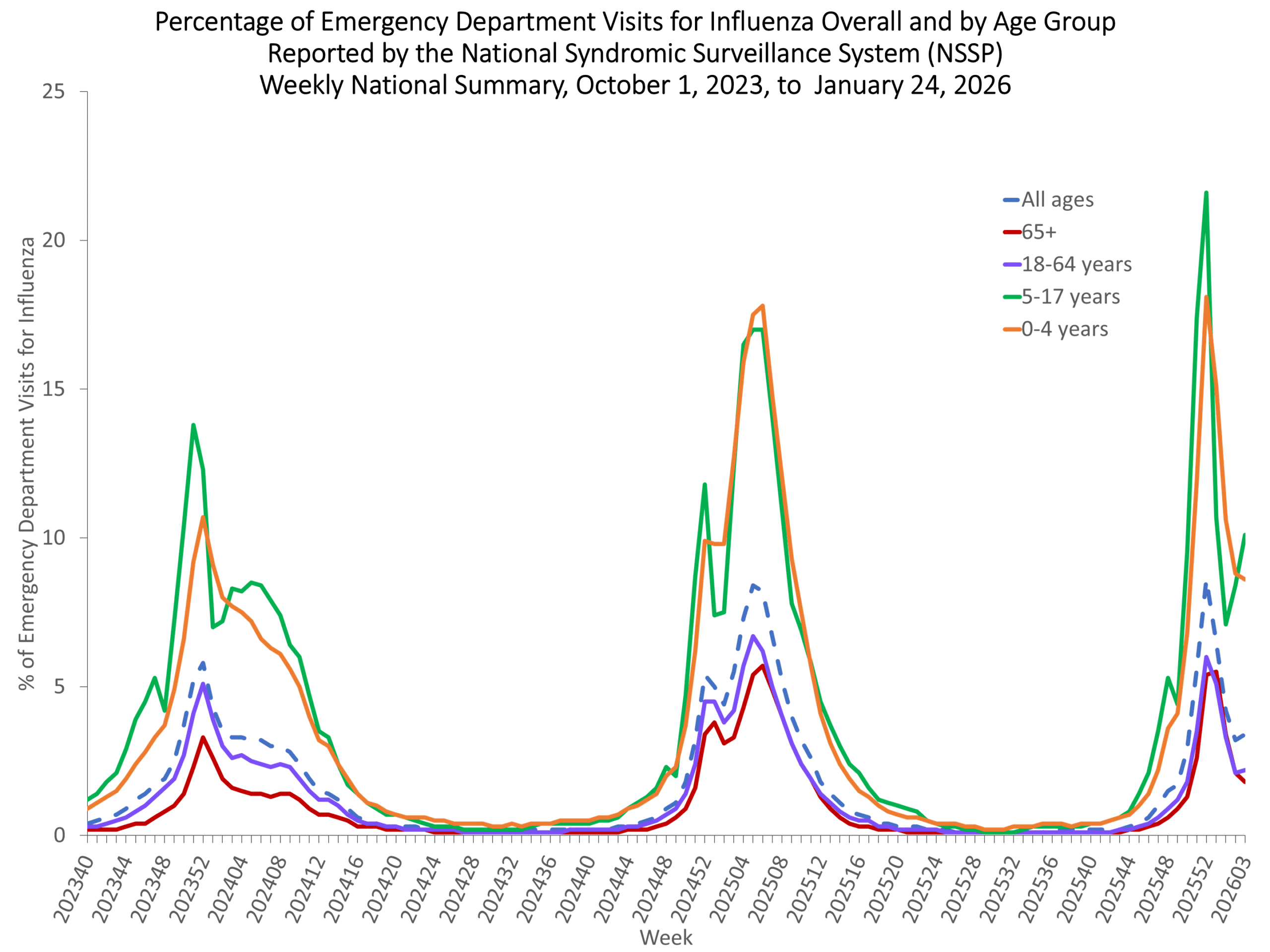 NSSP week 3 Additional information about emergency department visits for flu for current and past seasons:
NSSP week 3 Additional information about emergency department visits for flu for current and past seasons: **In this figure, weekly rates for all seasons prior to the 2024-2025 season reflect end-of-season rates. For the 2024-2025 season, rates for recent hospital admissions are subject to reporting delays and are shown as a dashed line for the current se... Show More
**In this figure, weekly rates for all seasons prior to the 2024-2025 season reflect end-of-season rates. For the 2024-2025 season, rates for recent hospital admissions are subject to reporting delays and are shown as a dashed line for the current se... Show More NHSN week 3 Additional NHSN Hospital Respiratory Data information:
NHSN week 3 Additional NHSN Hospital Respiratory Data information: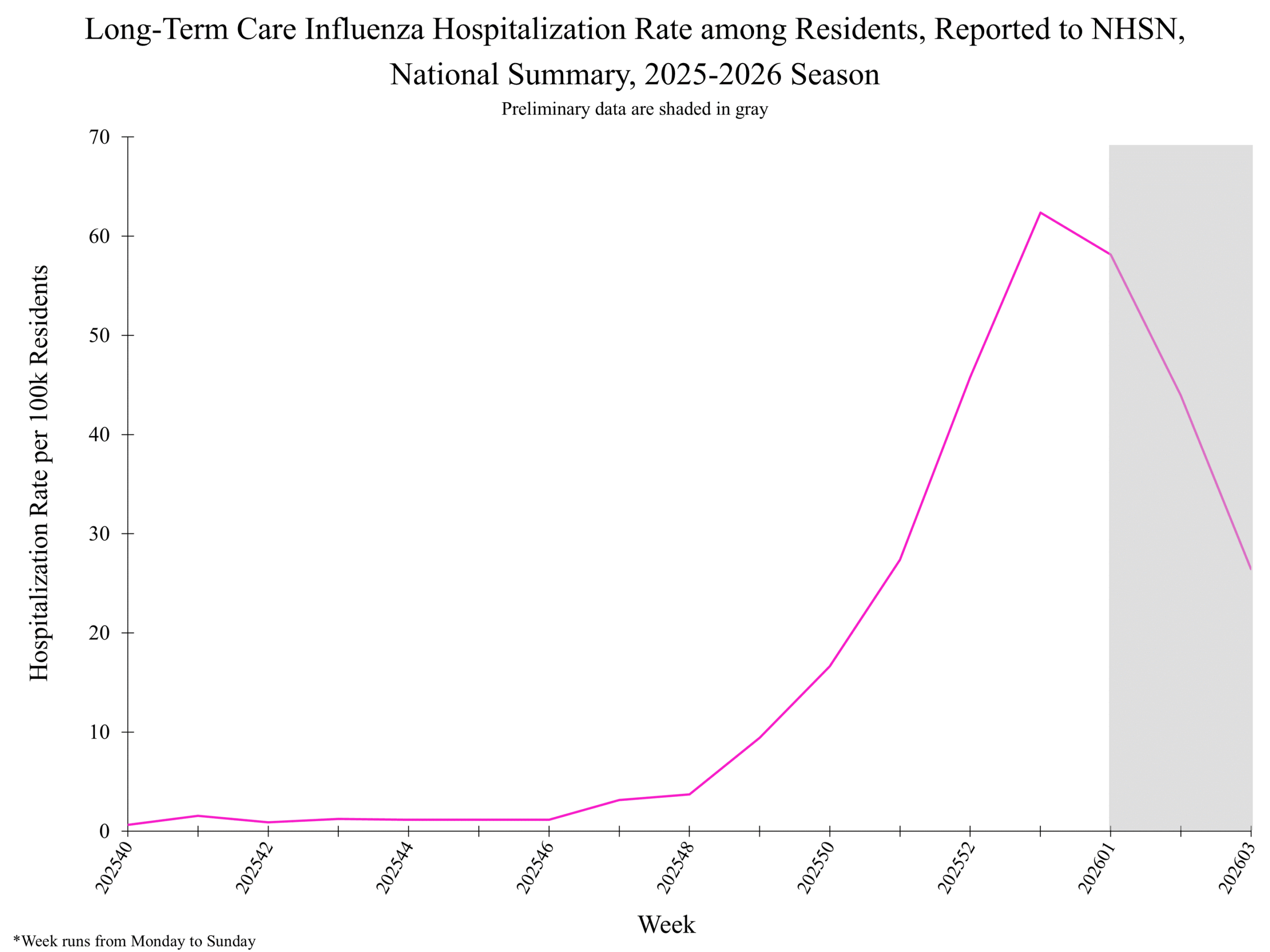 National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module
National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module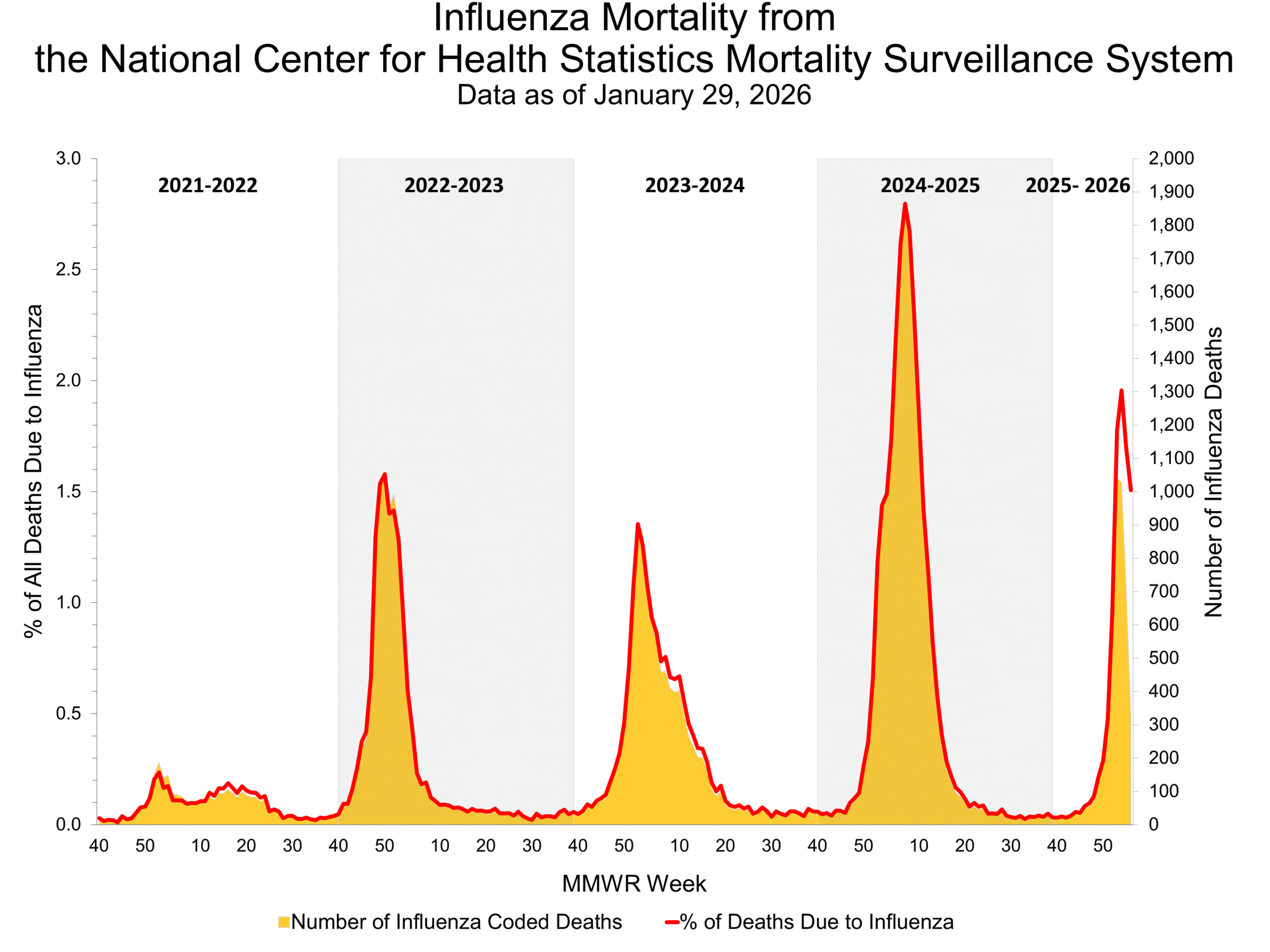 Influenza Mortality from the National Center for Health Statistics Mortality Surveillance System
Influenza Mortality from the National Center for Health Statistics Mortality Surveillance System Influenza-Associated Pediatric Deaths by Week of Death, 2022-23 season to 2025-26 season Additional pediatric mortality surveillance information for current and past seasons:
Influenza-Associated Pediatric Deaths by Week of Death, 2022-23 season to 2025-26 season Additional pediatric mortality surveillance information for current and past seasons:
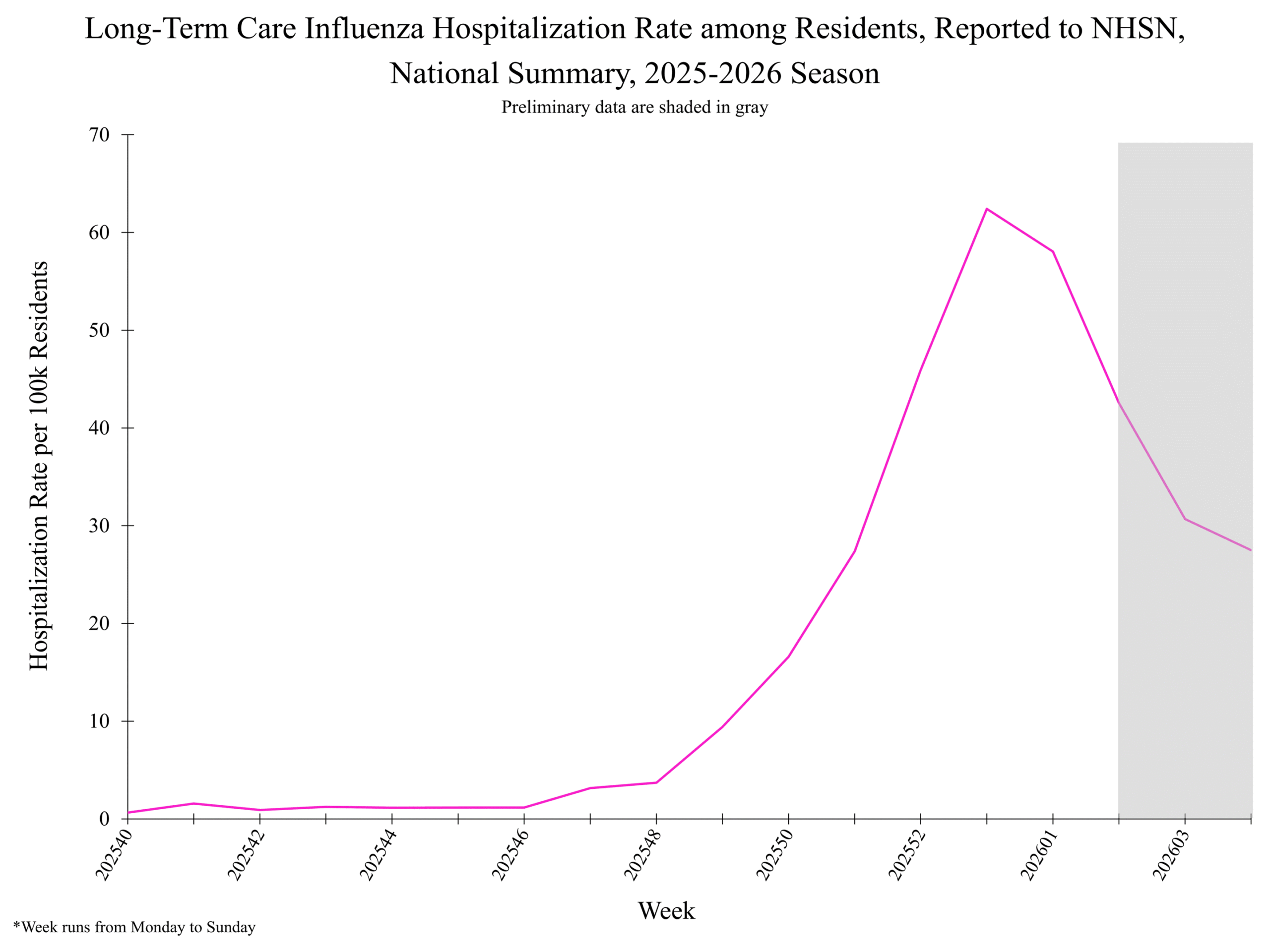 National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module
National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module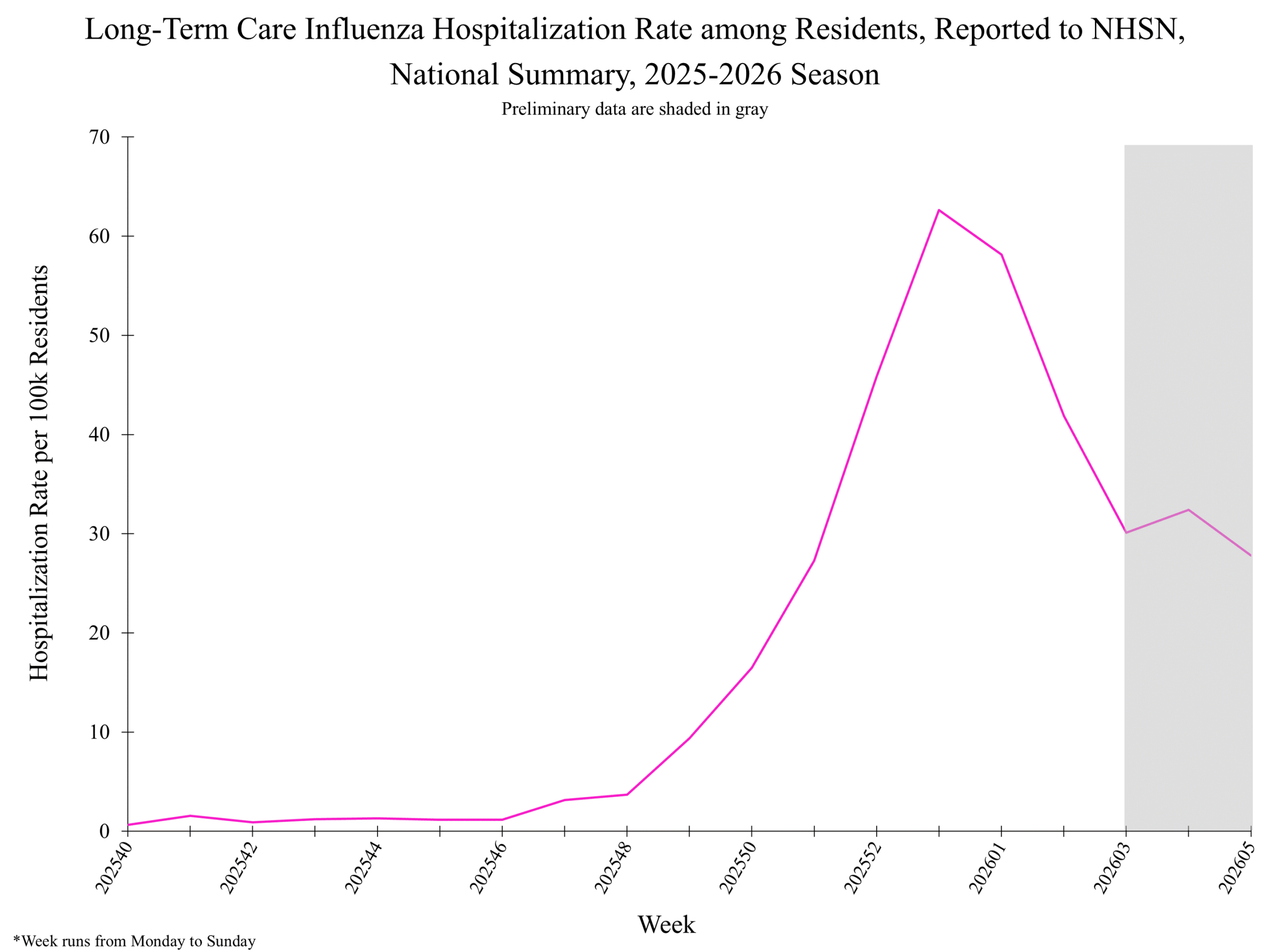 National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module
National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module Influenza Positive Tests Reported to CDC by Clinical Laboratories, National Summary, 2025-26 Season, week ending Feb. 14, 2026
Influenza Positive Tests Reported to CDC by Clinical Laboratories, National Summary, 2025-26 Season, week ending Feb. 14, 2026 This graph reflects the number of specimens tested and the number determined to be positive for influenza viruses at the public health lab (specimens tested is not the same as cases). It does not reflect specimens tested only at CDC and could include... Show More
This graph reflects the number of specimens tested and the number determined to be positive for influenza viruses at the public health lab (specimens tested is not the same as cases). It does not reflect specimens tested only at CDC and could include... Show More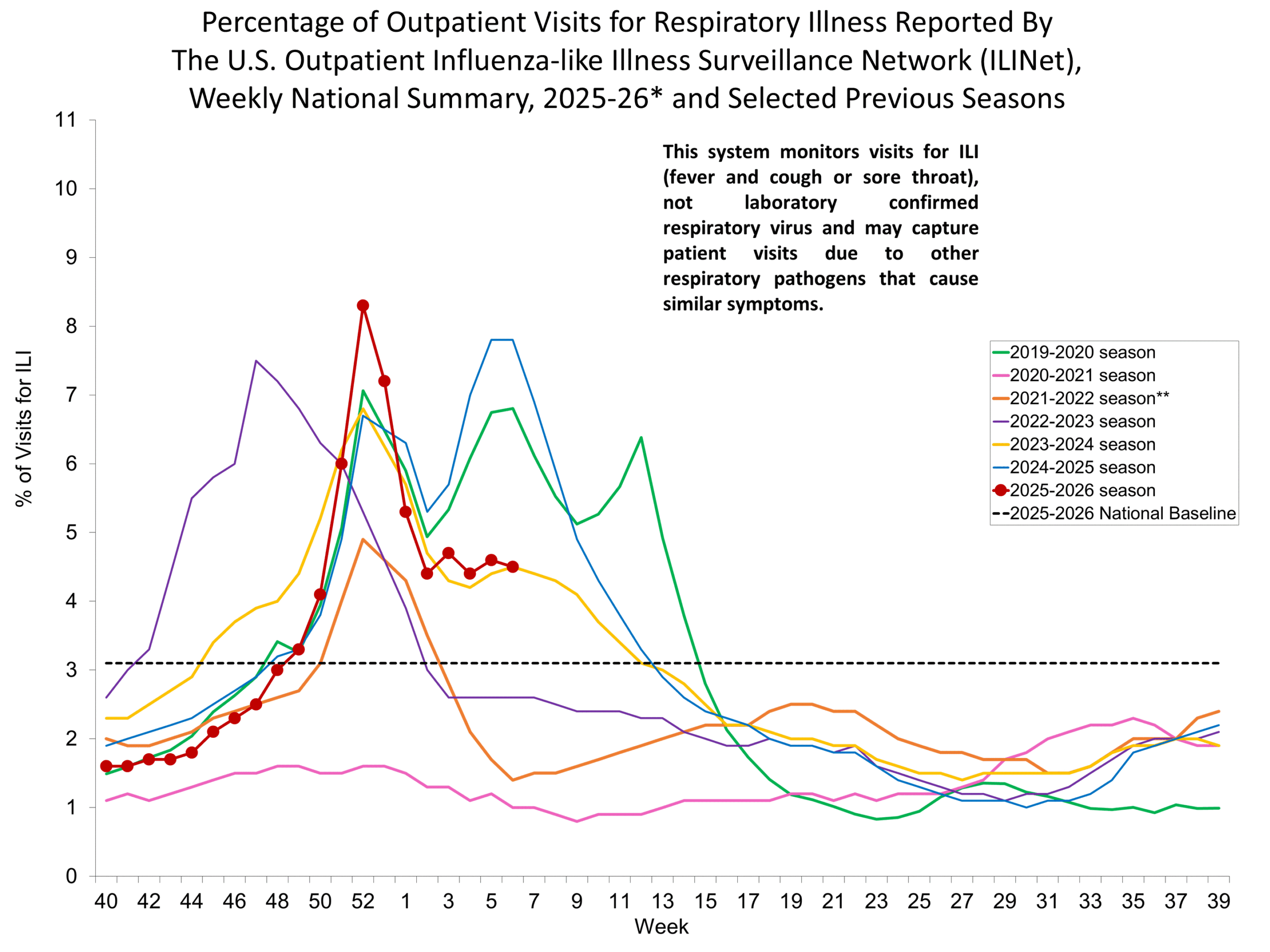 Percentage of Outpatient Visits for Respiratory Illness Reported by. The U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet)
Percentage of Outpatient Visits for Respiratory Illness Reported by. The U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet)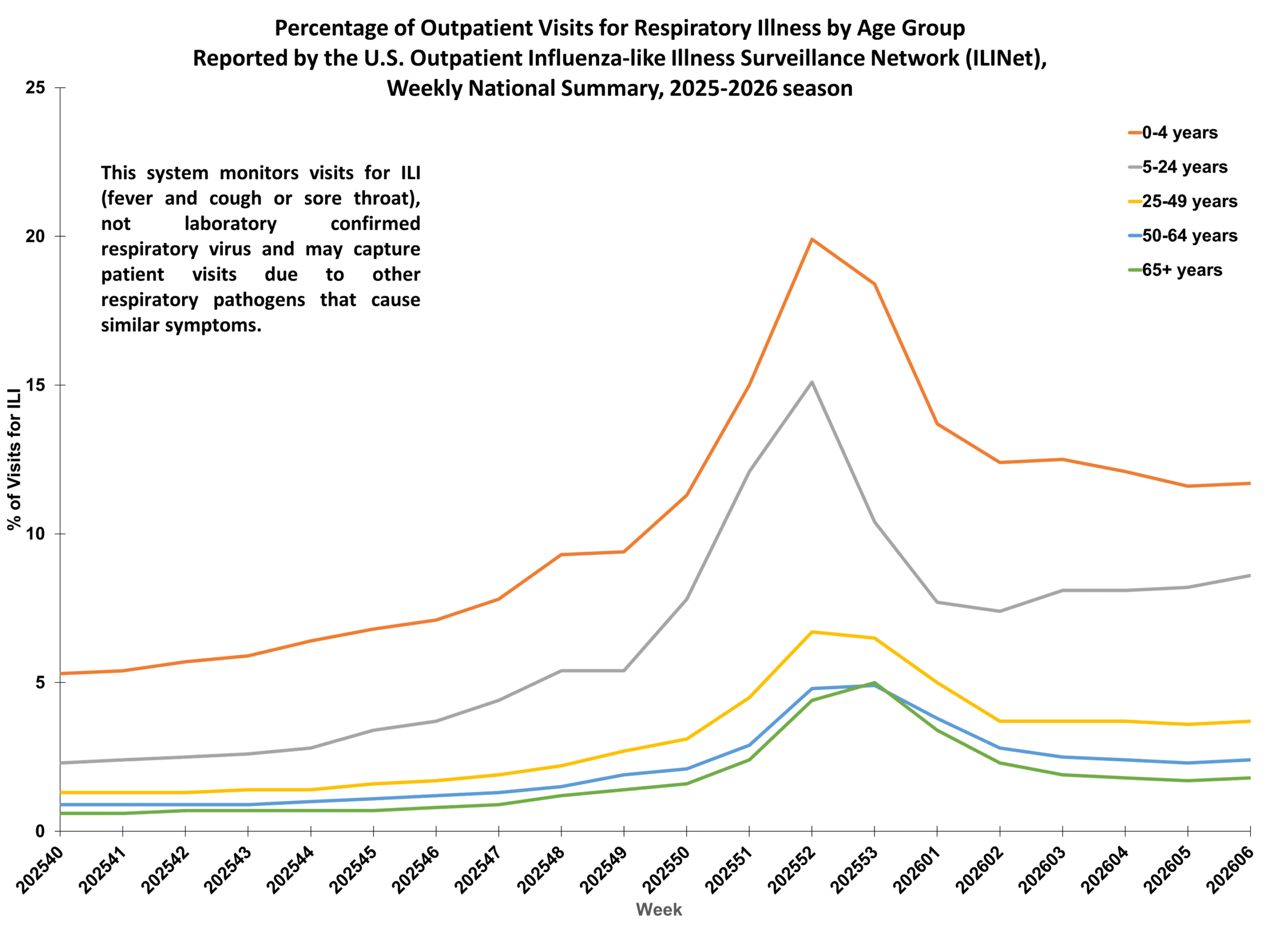 Percent of Outpatient Visits for Respiratory Illness by Age Group. Reported by the U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet)
Percent of Outpatient Visits for Respiratory Illness by Age Group. Reported by the U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet)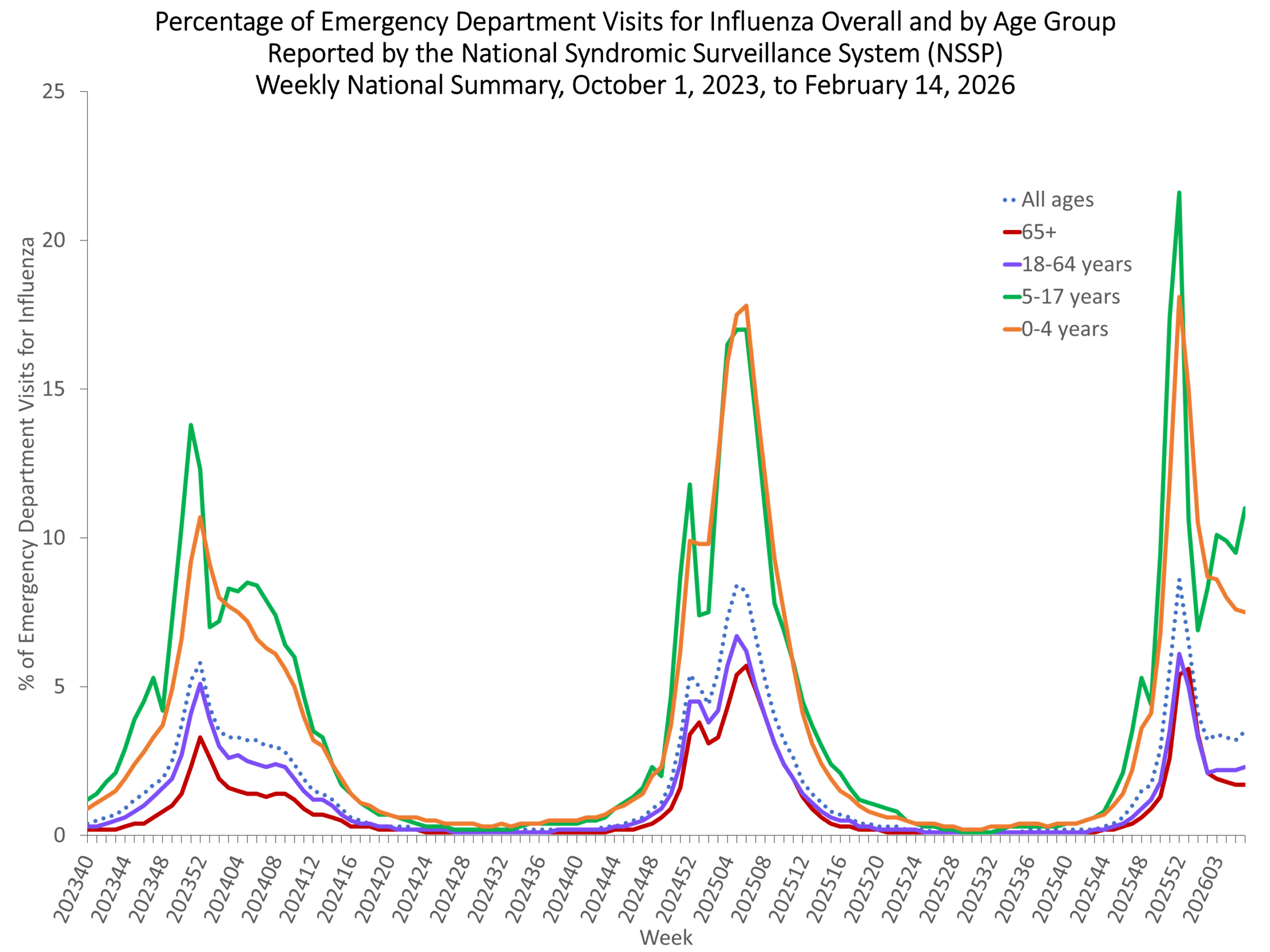 NSSP week 6 Additional information about emergency department visits for flu for current and past seasons:
NSSP week 6 Additional information about emergency department visits for flu for current and past seasons: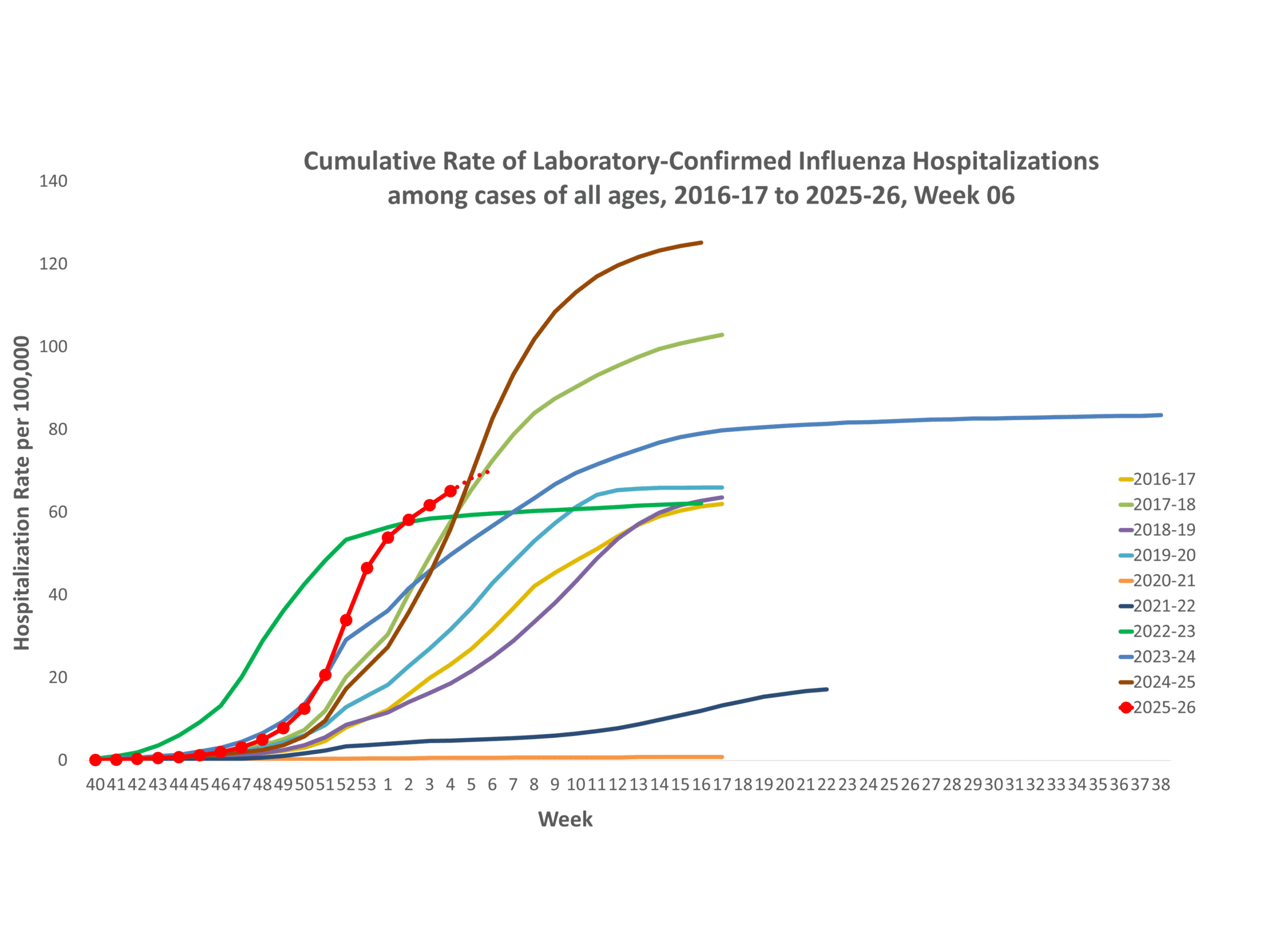 **In this figure, weekly rates for all seasons prior to the 2025-2026 season reflect end-of-season rates. For the 2025-2026 season, rates for recent hospital admissions are subject to reporting delays and are shown as a dashed line for the current se... Show More
**In this figure, weekly rates for all seasons prior to the 2025-2026 season reflect end-of-season rates. For the 2025-2026 season, rates for recent hospital admissions are subject to reporting delays and are shown as a dashed line for the current se... Show More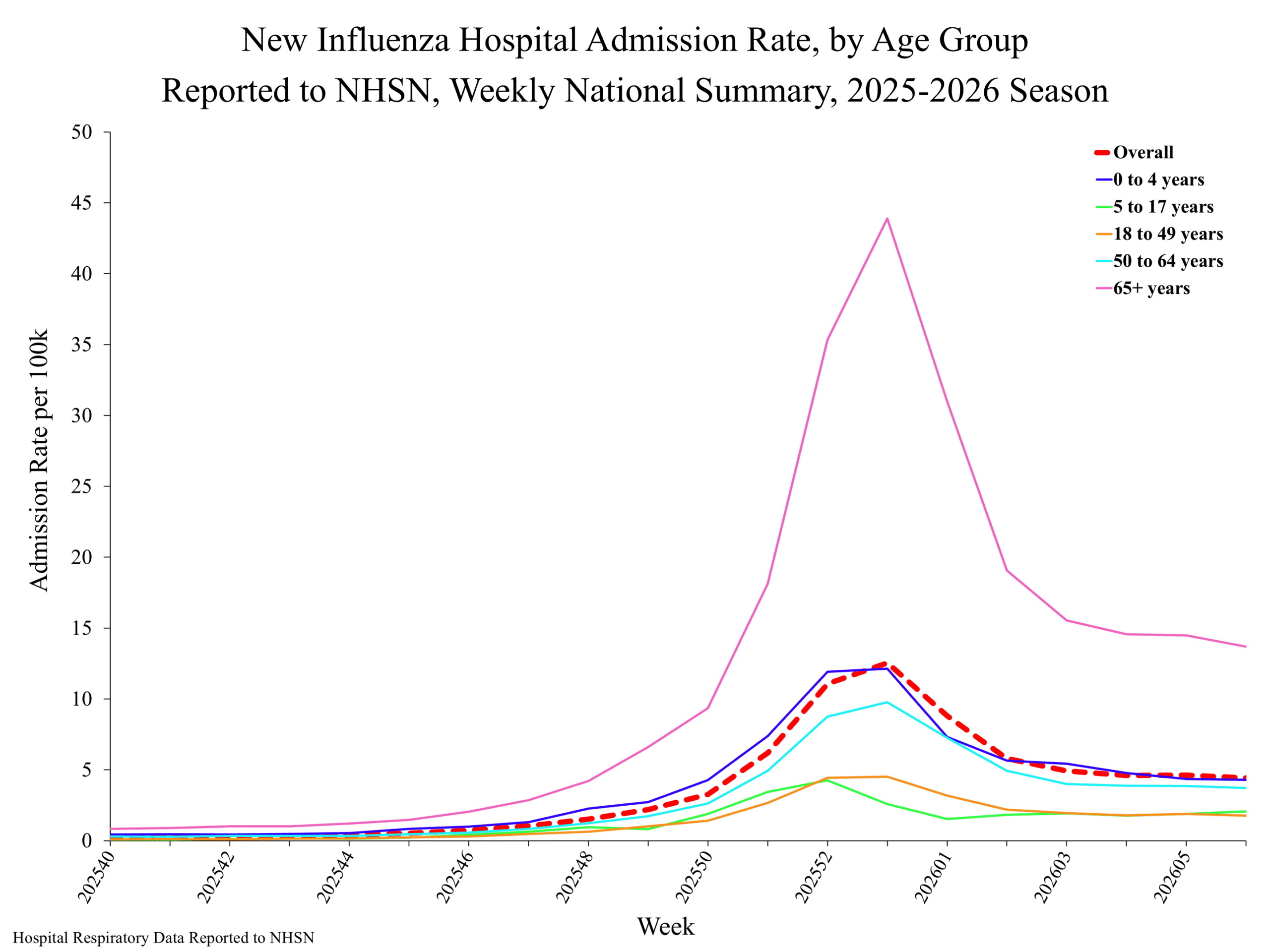 NHSN week 6
NHSN week 6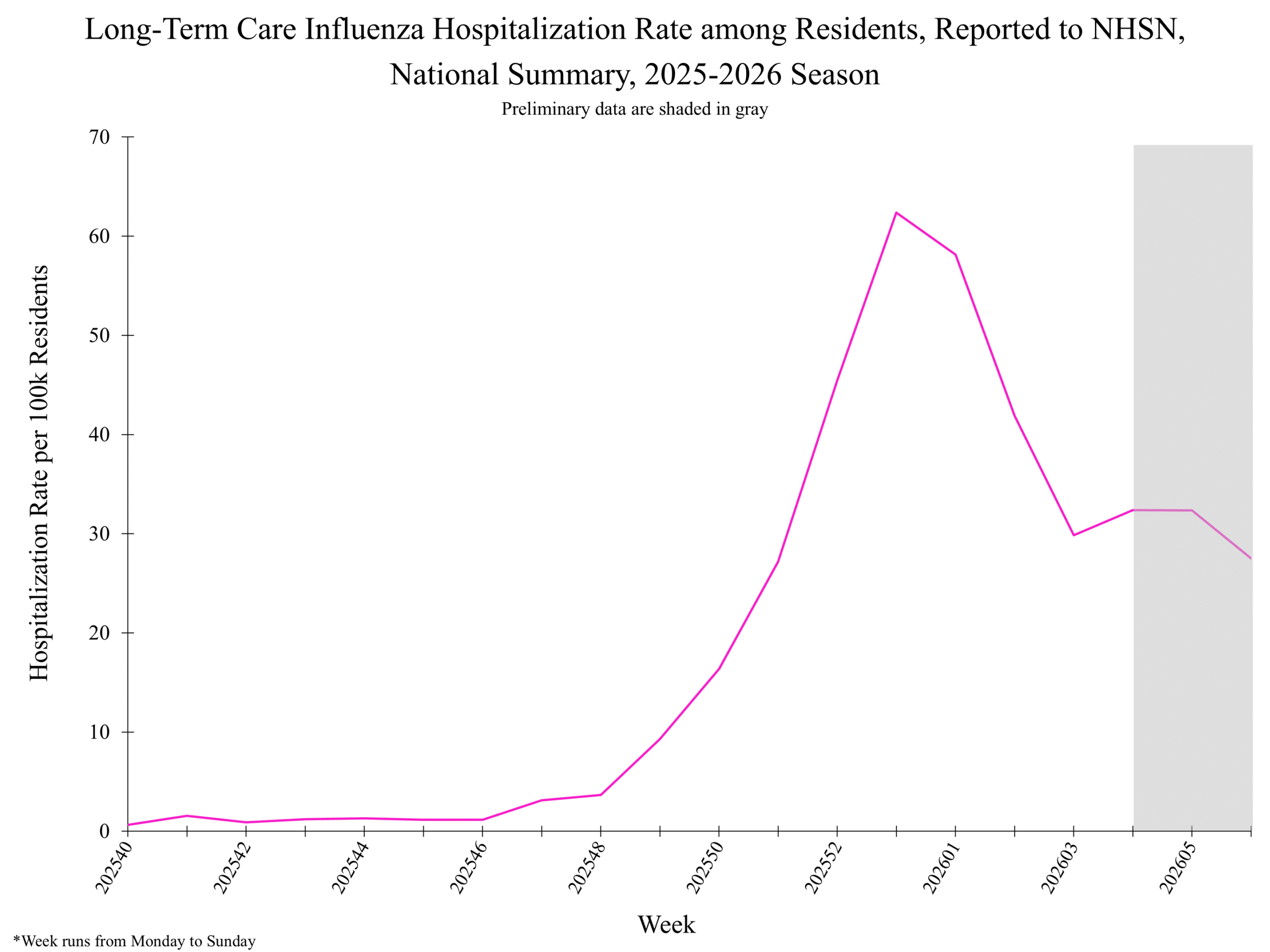 National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module Mortality surveillance
National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module Mortality surveillance Influenza Mortality from the National Center for Health Statistics Mortality Surveillance System
Influenza Mortality from the National Center for Health Statistics Mortality Surveillance System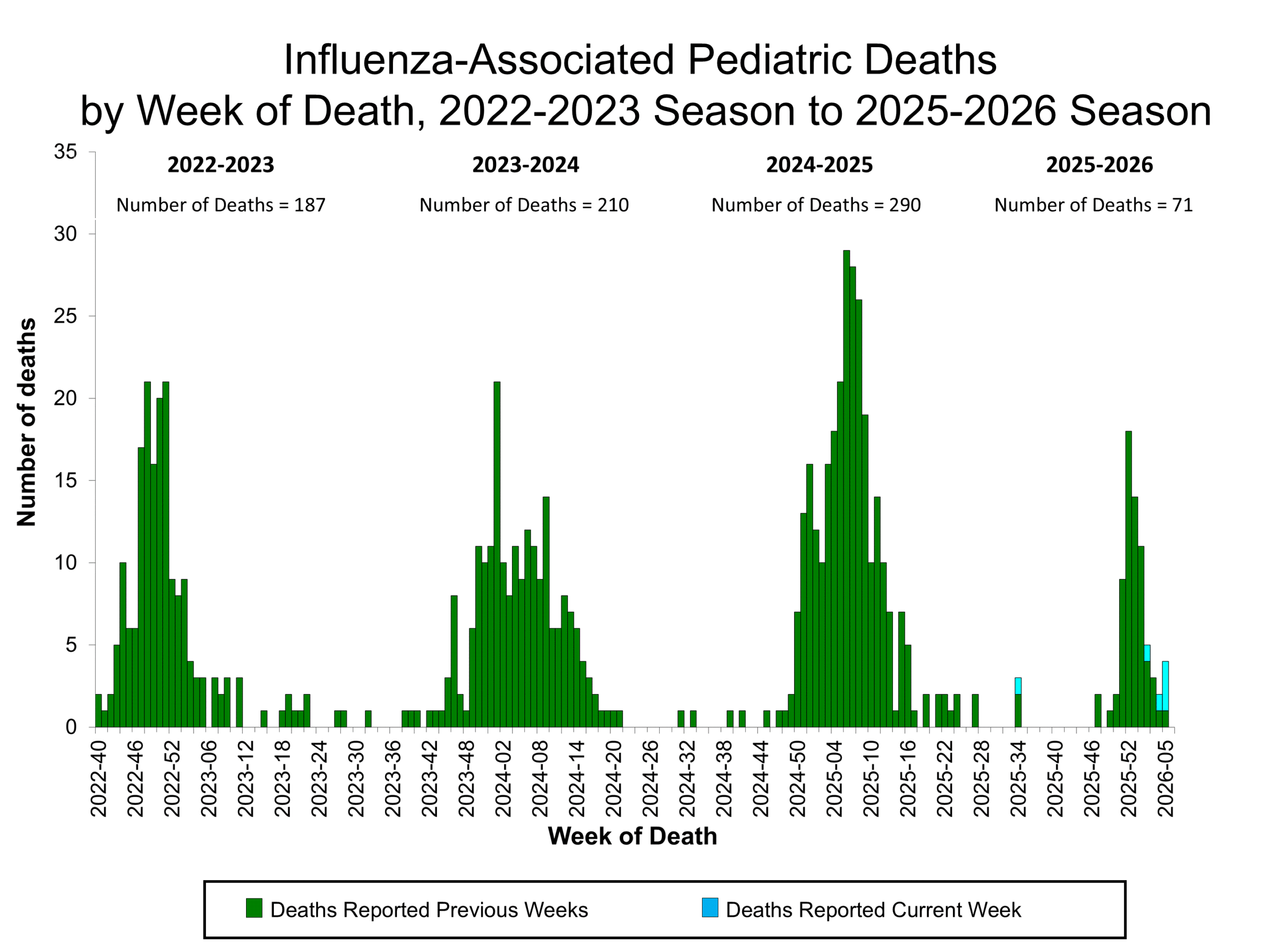 Influenza-Associated Pediatric Deaths by Week of Death, 2022-23 season to 2025-26 season Additional pediatric mortality surveillance information for current and past seasons:
Influenza-Associated Pediatric Deaths by Week of Death, 2022-23 season to 2025-26 season Additional pediatric mortality surveillance information for current and past seasons: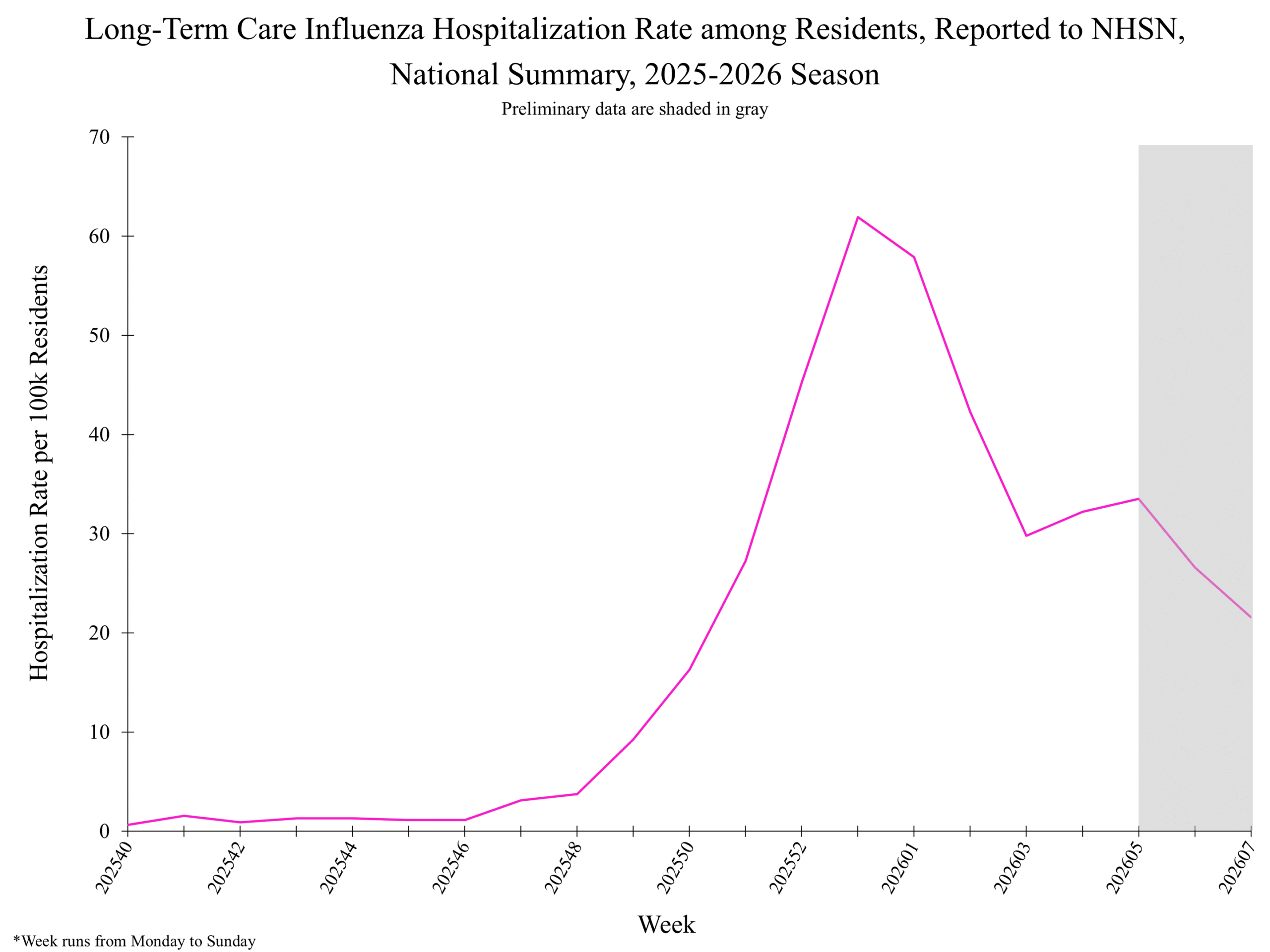 National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module
National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module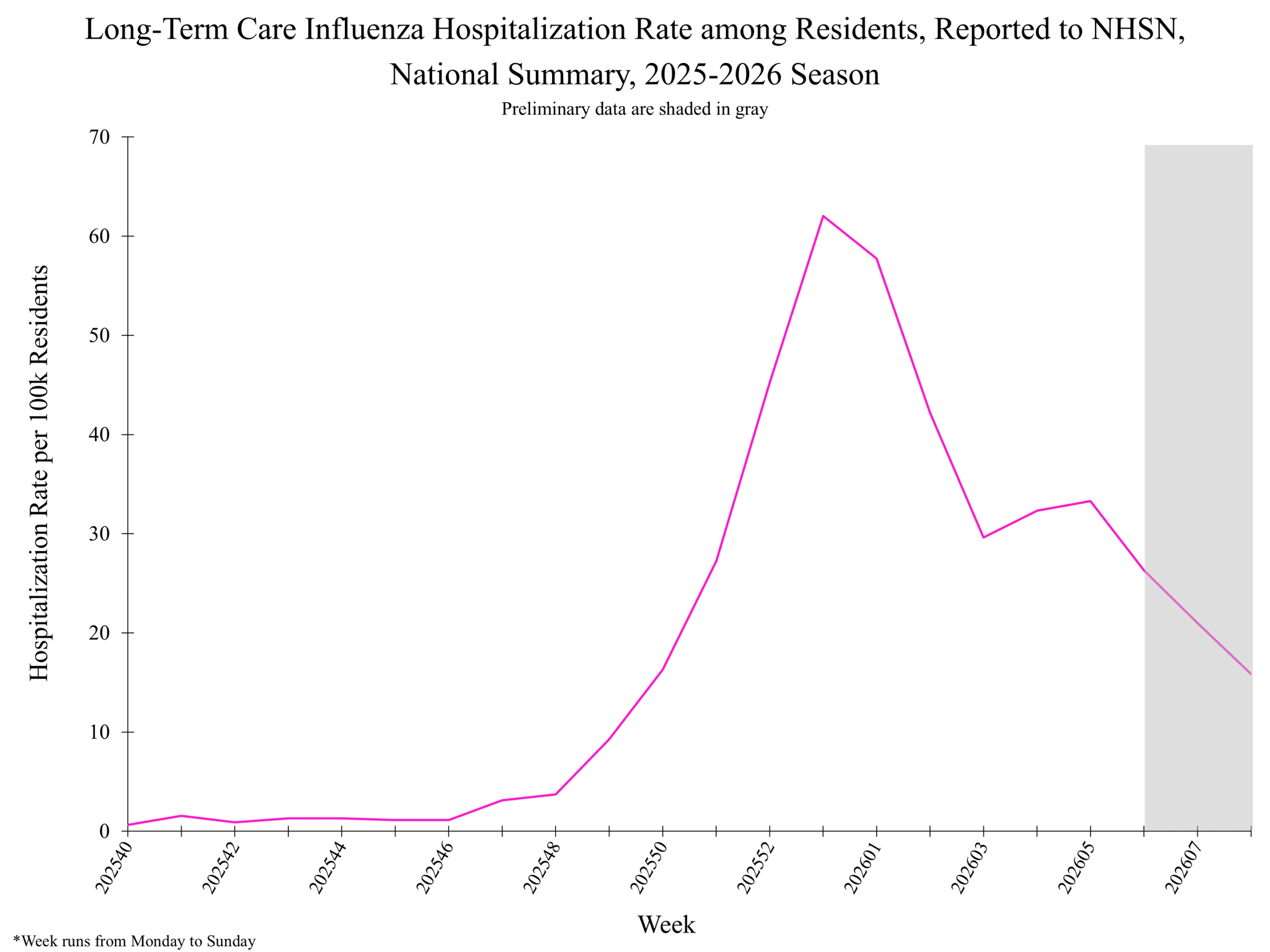 National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module
National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module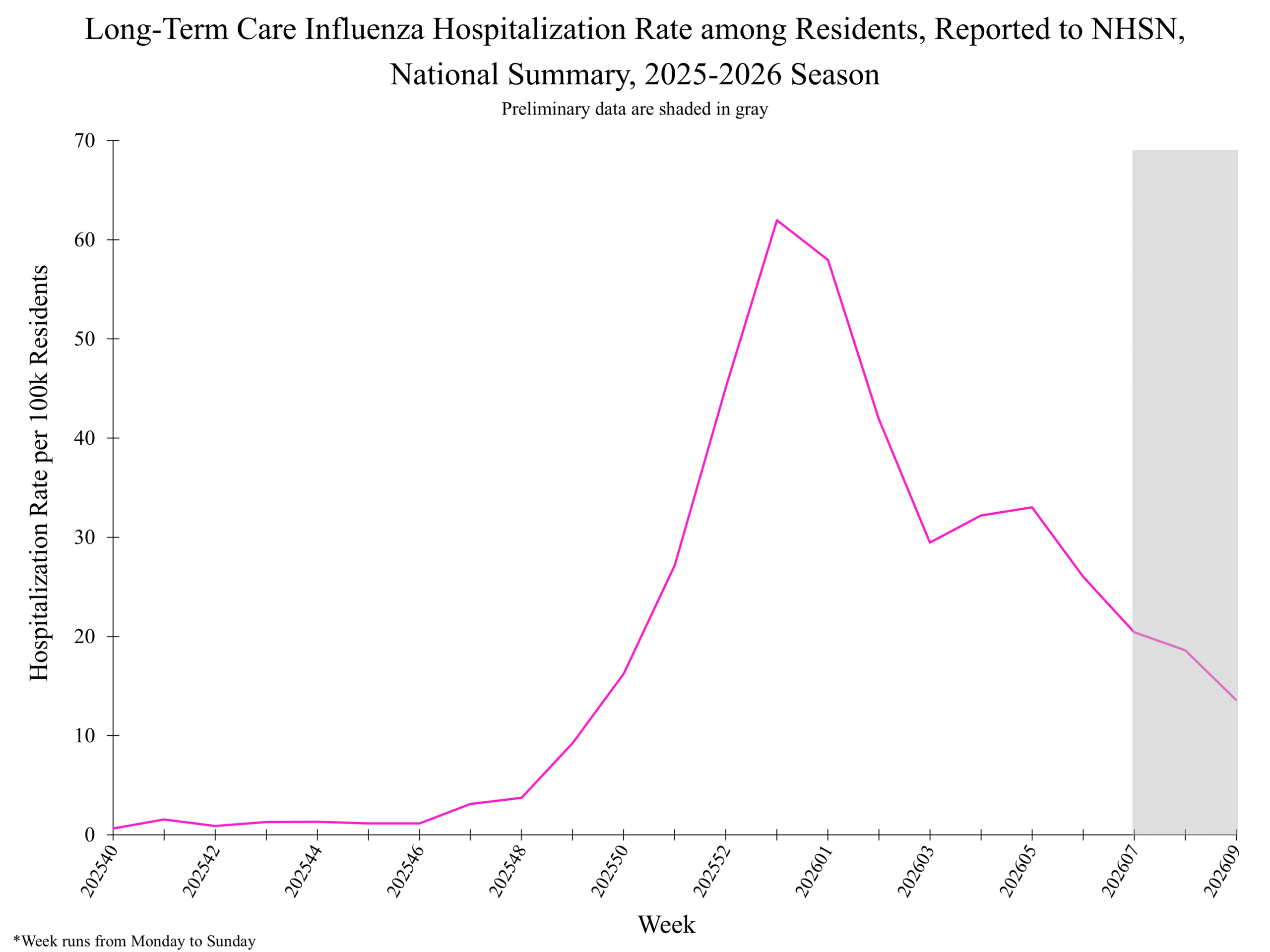 National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module
National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module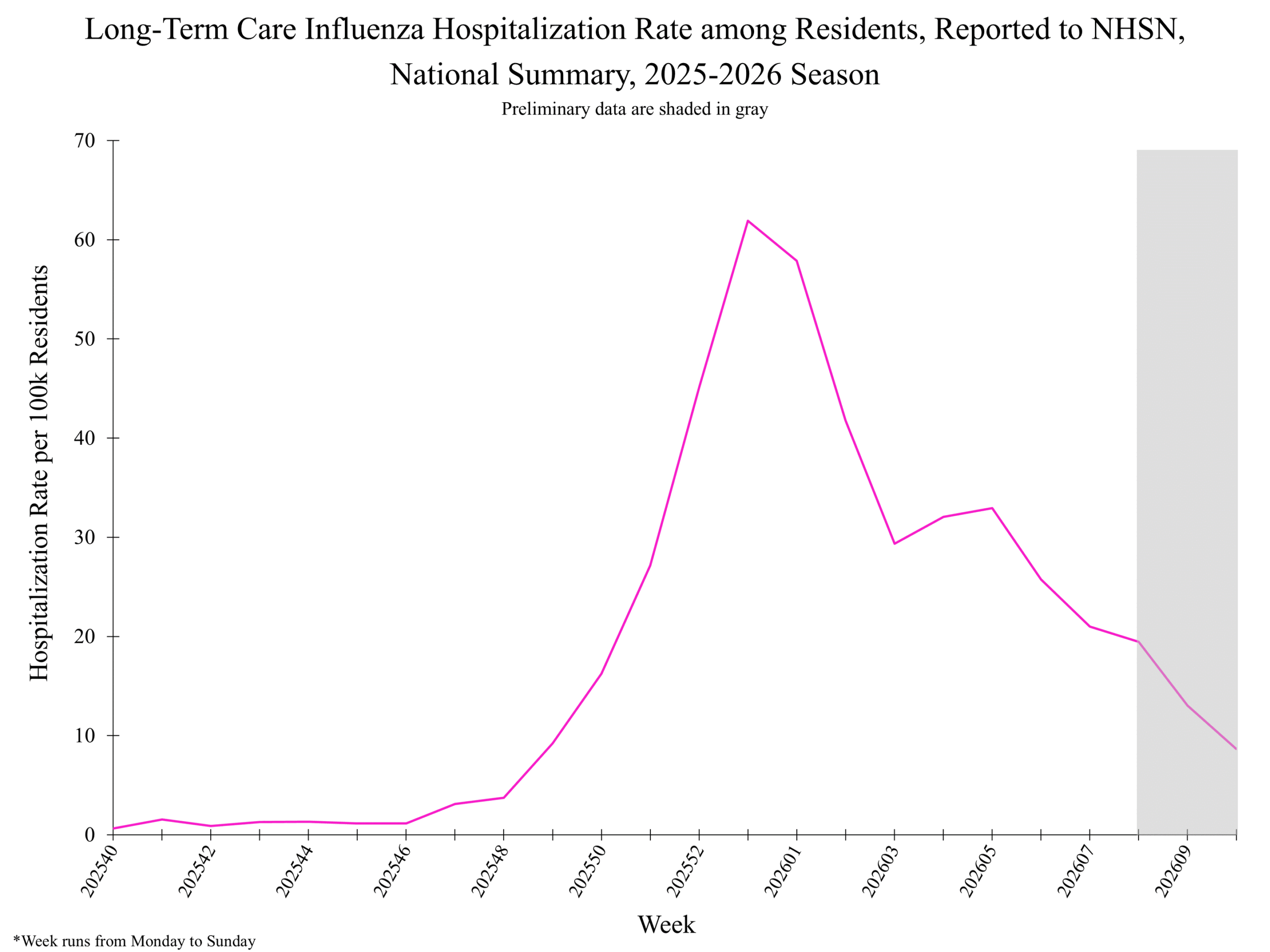 National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module
National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module
Comment