For the previous season please see:
US FluView - Weekly Surveillance Flu report 2024/2025 season - for trend analysis
For Everyone
NOV. 14, 2025
KEY POINTS
Seasonal influenza activity remains low nationally but is increasing.


Key Points
U.S. virologic surveillance
Nationally and in HHS regions 1, 2, 3, 5, 6, 7, 8, and 10, the percentage of respiratory specimens testing positive for influenza virus in clinical laboratories increased (change of ≥ 0.5 percentage points) compared to the previous week. In regions 4 and 9, the percentage remained stable compared to the previous week but is trending upwards over the past several weeks. Percent positivity varied by region, ranging from 1.0% (Region 7) to 5.5% (Region 8). For regional and state level data and age group distribution, please visit FluView Interactive. Viruses known to be associated with recent receipt of live attenuated influenza vaccine (LAIV) or found upon further testing to be a vaccine virus are not included, as they are not circulating influenza viruses.
Clinical Laboratories
The results of tests performed by clinical laboratories nationwide are summarized below. Data from clinical laboratories (the percentage of specimens tested that are positive for influenza virus) are used to monitor whether influenza activity is increasing or decreasing.

Public Health Laboratories
The results of tests performed by public health laboratories nationwide are summarized below. Data from public health laboratories are used to monitor the proportion of circulating influenza viruses that belong to each influenza subtype/lineage.

Public Health Laboratories
The results of tests performed by public health laboratories nationwide are summarized below. Data from public health laboratories are used to monitor the proportion of circulating influenza viruses that belong to each influenza subtype/lineage.



*This graph reflects the number of specimens determined to be positive for influenza viruses at the public health lab (specimens tested is not the same as cases). It does not reflect specimens tested only at CDC and could include more than one specimen tested per person. Specimens tested as part of routine influenza surveillance as well as those tested as part of targeted testing for people exposed to avian influenza A(H5) are included.
Continued: https://www.cdc.gov/fluview/surveill...5-week-45.html
US FluView - Weekly Surveillance Flu report 2024/2025 season - for trend analysis
For Everyone
NOV. 14, 2025
KEY POINTS
Seasonal influenza activity remains low nationally but is increasing.
Key Points
- Seasonal influenza activity remains low nationally but is increasing, primarily among children.
- Percent positivity for influenza and the percentage of emergency department visits for influenza among pediatric age groups increased this week.
- The timing of the increasing activity is similar to several past seasons, including the 2024-2025 season.
- During Week 45, of the 72 influenza viruses reported by public health laboratories, 67 were influenza A and 5 were influenza B. Of the 53 influenza A viruses subtyped during Week 45, 15 (28.3%) were influenza A(H1N1)pdm09 and 38 (71.7%) were A(H3N2).
- No influenza-associated pediatric deaths occurring during the 2025-2026 season have been reported to CDC.
- CDC recommends that everyone ages 6 months and older get an annual influenza (flu) vaccine, anytime viruses are circulating.1 More than 121 million doses of influenza vaccine have been distributed in the United States this season.
- There are prescription flu antiviral drugs that can treat flu illness; those should be started as early as possible and are especially important for patients at higher risk for flu-related complications.2
- Influenza viruses are among several viruses contributing to respiratory disease activity. CDC has provided updated, integrated informationabout COVID-19, flu, and respiratory syncytial virus (RSV) activity on a weekly basis.
U.S. virologic surveillance
Nationally and in HHS regions 1, 2, 3, 5, 6, 7, 8, and 10, the percentage of respiratory specimens testing positive for influenza virus in clinical laboratories increased (change of ≥ 0.5 percentage points) compared to the previous week. In regions 4 and 9, the percentage remained stable compared to the previous week but is trending upwards over the past several weeks. Percent positivity varied by region, ranging from 1.0% (Region 7) to 5.5% (Region 8). For regional and state level data and age group distribution, please visit FluView Interactive. Viruses known to be associated with recent receipt of live attenuated influenza vaccine (LAIV) or found upon further testing to be a vaccine virus are not included, as they are not circulating influenza viruses.
Clinical Laboratories
The results of tests performed by clinical laboratories nationwide are summarized below. Data from clinical laboratories (the percentage of specimens tested that are positive for influenza virus) are used to monitor whether influenza activity is increasing or decreasing.
Public Health Laboratories
The results of tests performed by public health laboratories nationwide are summarized below. Data from public health laboratories are used to monitor the proportion of circulating influenza viruses that belong to each influenza subtype/lineage.
Public Health Laboratories
The results of tests performed by public health laboratories nationwide are summarized below. Data from public health laboratories are used to monitor the proportion of circulating influenza viruses that belong to each influenza subtype/lineage.
*This graph reflects the number of specimens determined to be positive for influenza viruses at the public health lab (specimens tested is not the same as cases). It does not reflect specimens tested only at CDC and could include more than one specimen tested per person. Specimens tested as part of routine influenza surveillance as well as those tested as part of targeted testing for people exposed to avian influenza A(H5) are included.
Continued: https://www.cdc.gov/fluview/surveill...5-week-45.html

 )
) )
) ) weekly hospitalization rate per 100,000 residents
) weekly hospitalization rate per 100,000 residents
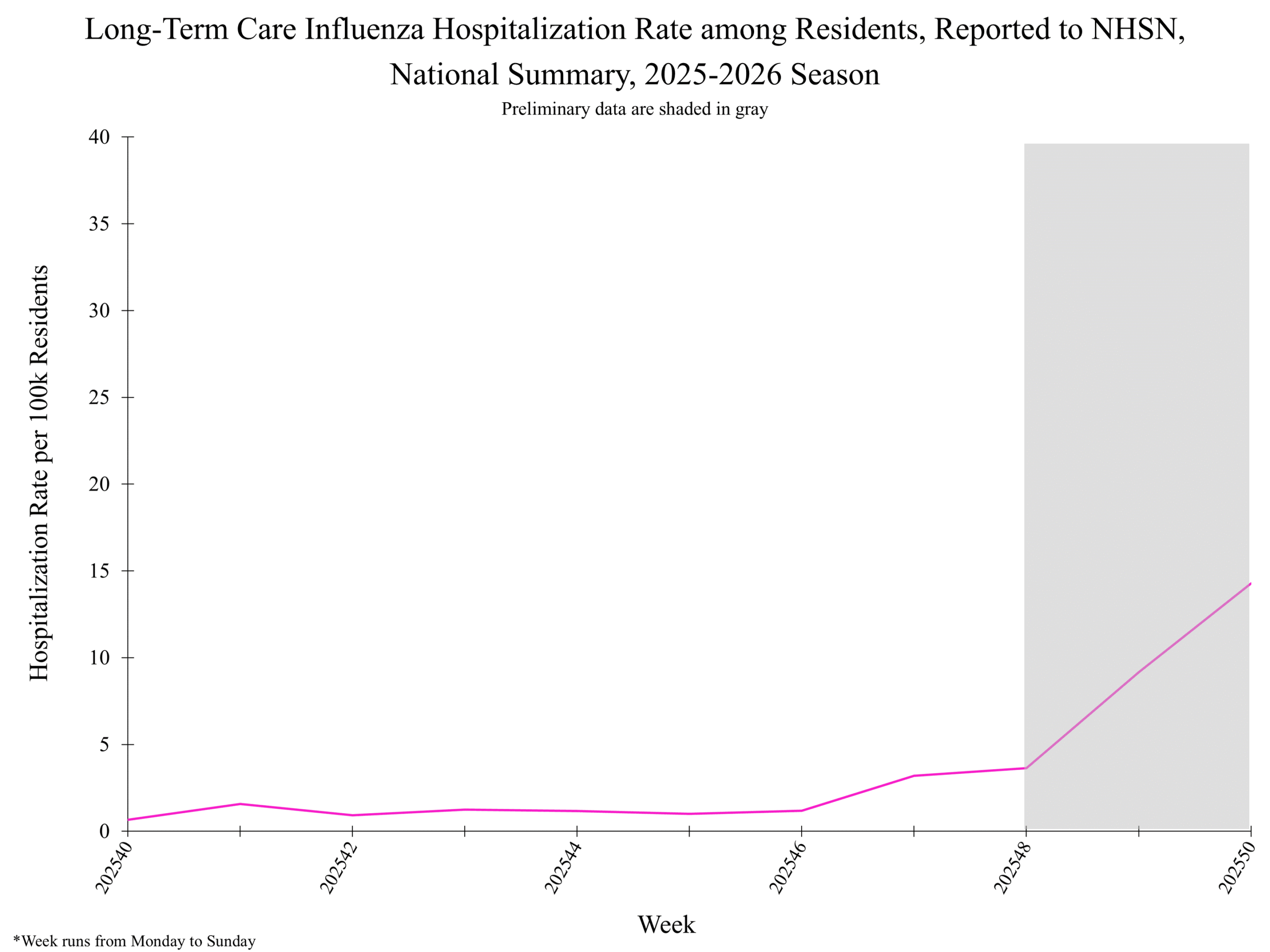 National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module
National Healthcare Safety Network (NHSN) Long-Term Care Respiratory Pathogens & Vaccination Module
Comment