Re: 7 dead, 91 sick from suspected fungal meningitis as outbreak worsens
Frequently Asked Questions: Multistate Meningitis Outbreak Investigation
October 6, 2012 8:00pm ET
Background
The Centers for Disease Control and Prevention (CDC) and the Food and Drug Administration (FDA) are currently coordinating a multistate investigation of fungal meningitis among patients who received an epidural steroid injection with a potentially contaminated product. Several of these patients also suffered strokes that are believed to have resulted from their infection.
How many cases have been reported?
Updates about the investigation, including case counts, are available at http://www.cdc.gov/hai/outbreaks/meningitis.html.
What is causing these infections?
The infections are caused by a fungus. At this point, the original source of the outbreak has not been determined; however, all infected patients identified thus far have received preservative-free (PF) methylprednisolone acetate (80mg/ml) from among the three lots voluntarily recalled by the New England Compounding Center in Framingham, Massachusetts, on September 26, 2012. These three lots are:
Are patients who did not receive an injection at risk?
No. Fungal meningitis is not transmitted from person to person. These infections are associated with a potentially contaminated medication that is injected into the body.
What should patients do?
What should physicians be doing?
Is there a role for prophylaxis?
Currently, CDC emphasizes the need for rapid clinical evaluation and diagnosis, if indicated. Prophylaxis is not recommended at this time.
Is there a role for lumbar puncture in asymptomatic patients?
At this time, CDC does not recommend performing lumbar puncture in exposed patients who are currently asymptomatic. These patients should be closely monitored for development of symptoms, with a low threshold for performing lumbar puncture if the patient should become symptomatic. The clinical investigation of patients associated with this outbreak is ongoing, and this recommendation may change as new information becomes available.
Were the three lots mentioned above only used for spinal injections?
No. These medications were used for other types of injections, including injections into the joint (e.g., knee). To date, CDC has only identified infections in patients who received epidural steroid injections with these medications. However, patients who received other types of injections with these products may also be at risk.
What states have received the potentially contaminated medication?
Twenty-three states have received medication from the New England Compounding Center:
The names of the facilities that have received medication from one of these lots are available at http://www.cdc.gov/hai/outbreaks/men...ities-map.html.
Are other medications from the New England Compounding Center located in Framingham, Massachusetts associated with infections?
To date, CDC has not received reports of infections linked to other products from the New England Compounding Center. However, out of an abundance of caution, CDC recommends that healthcare professional cease use of any product produced by the New England Compounding Center until further information is available.
What is fungal meningitis?
Fungal meningitis occurs when the protective membranes that cover the brain and spinal cord are infected with a fungus. Fungal meningitis can develop after a fungus spreads through the bloodstream from somewhere else in the body, as a result of the fungus being introduced directly into the central nervous system, or by direct extension from an infected body site next to the central nervous system.
Is fungal meningitis common after epidural injections?
Epidural injections are generally very safe procedures, and complications are rare. Fungal meningitis is an extremely rare type of meningitis overall, including after epidural injections. The type of epidural medication given to patients affected by this outbreak is not the same type of medication as that given to women during childbirth.
What are the symptoms of fungal meningitis?
Symptoms of fungal meningitis are similar to symptoms of other forms of meningitis; however, they often appear gradually and can be very mild at first. In addition to typical meningitis symptoms, like headache, fever, nausea, and stiffness of the neck, people with fungal meningitis may also experience confusion, dizziness, and discomfort from bright lights. Patients might have only one or two of these symptoms.
What is a compounding pharmacy? Why are these medications compounded when they are also commercially available?
Compounding pharmacies create special formulations of medications in order to fit patients’ healthcare needs. For example, they may change the dose or change the formulation of a medication from a solid to a liquid.
Where can I find updates and additional information on this outbreak?
For complete information and updates on this outbreak, visit www.cdc.gov/hai/outbreaks/meningitis/html.
Contact Us:
Frequently Asked Questions: Multistate Meningitis Outbreak Investigation
October 6, 2012 8:00pm ET
Background
The Centers for Disease Control and Prevention (CDC) and the Food and Drug Administration (FDA) are currently coordinating a multistate investigation of fungal meningitis among patients who received an epidural steroid injection with a potentially contaminated product. Several of these patients also suffered strokes that are believed to have resulted from their infection.
How many cases have been reported?
Updates about the investigation, including case counts, are available at http://www.cdc.gov/hai/outbreaks/meningitis.html.
What is causing these infections?
The infections are caused by a fungus. At this point, the original source of the outbreak has not been determined; however, all infected patients identified thus far have received preservative-free (PF) methylprednisolone acetate (80mg/ml) from among the three lots voluntarily recalled by the New England Compounding Center in Framingham, Massachusetts, on September 26, 2012. These three lots are:
- Methylprednisolone Acetate (PF) 80 mg/ml Injection, Lot #05212012@68, BUD 11/17/2012
- Methylprednisolone Acetate (PF) 80 mg/ml Injection, Lot #06292012@26, BUD 12/26/2012
- Methylprednisolone Acetate (PF) 80 mg/ml Injection, Lot #08102012@51, BUD 2/6/2013
Are patients who did not receive an injection at risk?
No. Fungal meningitis is not transmitted from person to person. These infections are associated with a potentially contaminated medication that is injected into the body.
What should patients do?
- Find out if you received a potentially contaminated medication. If patients are concerned about which product was used in their procedure, they should first contact the physician who performed their procedure.
The facilities who received one of the lots recalled on September 26, 2012, are actively contacting patients to find out if they are feeling well. The list of facilities that received medication from one of these three lots is available at http://www.cdc.gov/hai/outbreaks/men...ities-map.html.
- If you have received a potentially contaminated medication, seek medical attention if you have symptoms. It is important to note that infected patients have had very mild symptoms that are only slightly worse than usual. For example, many infected patients have had slight weakness, slightly worsened back pain, or even a mild headache. Patients have had symptoms generally starting from 1 to 4 weeks after their injection.
Patients who have had an epidural steroid injection since May 21, 2012, and have any of the following symptoms, should talk to their doctor as soon as possible:- New or worsening headache
- Fever
- Sensitivity to light
- Stiff neck
- New weakness or numbness in any part of your body
- Slurred speech
- Increased pain, redness or swelling at your injection site
What should physicians be doing?
- Find out if you have administered a potentially contaminated medication from New England Compounding Center. The three potentially contaminated lots are:
- Methylprednisolone Acetate (PF) 80 mg/ml Injection, Lot #05212012@68, BUD 11/17/2012
- Methylprednisolone Acetate (PF) 80 mg/ml Injection, Lot #06292012@26, BUD 12/26/2012
- Methylprednisolone Acetate (PF) 80 mg/ml Injection, Lot #08102012@51, BUD 2/6/2013
- Contact patients who have been exposed. All patients who have received medication from one of these three lots should be immediately contacted directly (i.e. called) to determine if they are having any symptoms.
It is important to note that infected patients have presented with mild symptoms, only slightly worse than baseline. - Refer symptomatic patients for a diagnostic procedure. For patients who received epidural injection with medication from the lots listed above and have any symptoms of meningitis or basilar stroke, a diagnostic lumbar puncture (LP) should be performed, if not contraindicated. Because presenting symptoms of some patients with meningitis have been mild and not classic for meningitis (e.g., new or worsening headache without fever or neck stiffness), physicians should have a low threshold for LP.
While CDC is only aware of infections occurring in patients who have received epidural steroid injections, patients who received other types of injection (e.g., joint injection) with potentially contaminated methylprednisolone acetate should also be contacted to assess for signs of infection (e.g., swelling, increasing pain, redness, warmth at the injection site) and should be encouraged to seek evaluation (e.g., arthrocentesis) if such symptoms exist.
Interim instructions regarding diagnostic testing and treatment options are available at http://www.cdc.gov/HAI/outbreaks/meningitis.html. - Report concerning cases to public health. Physicians should report suspected cases to their state health department.
Clinicians are also requested to report any suspected adverse events following use of these products to FDA's MedWatch program at 1-800-332-1088 or www.fda.gov/medwatch .
.
Is there a role for prophylaxis?
Currently, CDC emphasizes the need for rapid clinical evaluation and diagnosis, if indicated. Prophylaxis is not recommended at this time.
Is there a role for lumbar puncture in asymptomatic patients?
At this time, CDC does not recommend performing lumbar puncture in exposed patients who are currently asymptomatic. These patients should be closely monitored for development of symptoms, with a low threshold for performing lumbar puncture if the patient should become symptomatic. The clinical investigation of patients associated with this outbreak is ongoing, and this recommendation may change as new information becomes available.
Were the three lots mentioned above only used for spinal injections?
No. These medications were used for other types of injections, including injections into the joint (e.g., knee). To date, CDC has only identified infections in patients who received epidural steroid injections with these medications. However, patients who received other types of injections with these products may also be at risk.
What states have received the potentially contaminated medication?
Twenty-three states have received medication from the New England Compounding Center:
- California
- Connecticut
- Florida
- Georgia
- Idaho
- Illinois
- Indiana
- Maryland
- Michigan
- Minnesota
- North Carolina
- New Hampshire
- New Jersey
- Nevada
- New York
- Ohio
- Pennsylvania
- Rhode Island
- South Carolina
- Tennessee
- Virginia
- Texas
- West Virginia
The names of the facilities that have received medication from one of these lots are available at http://www.cdc.gov/hai/outbreaks/men...ities-map.html.
Are other medications from the New England Compounding Center located in Framingham, Massachusetts associated with infections?
To date, CDC has not received reports of infections linked to other products from the New England Compounding Center. However, out of an abundance of caution, CDC recommends that healthcare professional cease use of any product produced by the New England Compounding Center until further information is available.
What is fungal meningitis?
Fungal meningitis occurs when the protective membranes that cover the brain and spinal cord are infected with a fungus. Fungal meningitis can develop after a fungus spreads through the bloodstream from somewhere else in the body, as a result of the fungus being introduced directly into the central nervous system, or by direct extension from an infected body site next to the central nervous system.
Is fungal meningitis common after epidural injections?
Epidural injections are generally very safe procedures, and complications are rare. Fungal meningitis is an extremely rare type of meningitis overall, including after epidural injections. The type of epidural medication given to patients affected by this outbreak is not the same type of medication as that given to women during childbirth.
What are the symptoms of fungal meningitis?
Symptoms of fungal meningitis are similar to symptoms of other forms of meningitis; however, they often appear gradually and can be very mild at first. In addition to typical meningitis symptoms, like headache, fever, nausea, and stiffness of the neck, people with fungal meningitis may also experience confusion, dizziness, and discomfort from bright lights. Patients might have only one or two of these symptoms.
What is a compounding pharmacy? Why are these medications compounded when they are also commercially available?
Compounding pharmacies create special formulations of medications in order to fit patients’ healthcare needs. For example, they may change the dose or change the formulation of a medication from a solid to a liquid.
Where can I find updates and additional information on this outbreak?
For complete information and updates on this outbreak, visit www.cdc.gov/hai/outbreaks/meningitis/html.
Contact Us:
- Centers for Disease Control and Prevention
1600 Clifton Rd
Atlanta, GA 30333 - 800-CDC-INFO
(800-232-4636)
TTY: (888) 232-6348 - New Hours of Operation
8am-8pm ET/Monday-Friday
Closed Holidays - cdcinfo@cdc.gov
- Page last reviewed: October 6, 2012 8:00pm ET
- Page last updated: October 6, 2012 8:00pm ET
- Content source: Centers for Disease Control and Prevention
National Center for Emerging and Zoonotic Infectious Diseases (NCEZID)
Division of Healthcare Quality Promotion (DHQP)






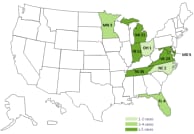 Click to view case count map.
Click to view case count map.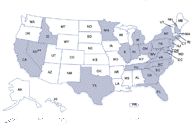 Click to view facilities map.
Click to view facilities map.
Comment