Translation Google
Introduction of the mpox virus to the islands of the Southwest Indian Ocean
Olivia Vong
February 25, 2026

Introduction of the mpox virus to the islands of the Southwest Indian Ocean
Ambroise Mercier 1,2 , Mélissa Bérot 3 , Sylvain Brisse 4 , Marie-Pierre Moiton 5 , Olivier Belmonte 1 , Laurent Souply 1 , Guillaume Miltgen 1,2,6
Introduction
Mpox, or mpox B, is a zoonotic disease caused by the mpox virus ( mpox virus [MPV], formerly monkeypox virus ). This virus belongs to the genus Orthopoxvirus (family Poxviridae ), which includes other members such as smallpox virus (variola virus [VARV]), vaccinia virus ( vaccinia virus [VACV]), and cowpox virus (CPV) (1). This zoonosis, less severe than smallpox, affects rodents (fungiur rats, giant pouched rats, dormice, etc. ) and primates (including humans) (2). Since the eradication of smallpox in 1977 through vaccination, MPV and CPV are the most studied orthopoxviruses due to their high epidemic potential (3).
MPV, initially distributed in Africa, has been responsible for numerous mycobacterium tuberculosis epidemics worldwide. This virus was first discovered in a pet shop in Copenhagen in 1958 in primates (4). It was not until 1970 that it was described in humans, in the Democratic Republic of Congo (5). Since then, other cases have been reported in Africa. The first extra-African epidemic occurred in 2003 in the United States, where domestic prairie dogs were infected through contact with infected savannah worms from Ghana (6). In 2022, a new global epidemic occurred, characterized by human-to-human transmission via direct contact with the skin and mucous membrane lesions of infected patients, primarily men who have sex with men (MSM) (7).
Phylogenetically, MPV is divided into two clades based on genomic sequences, with clade I from Central Africa exhibiting higher severity and mortality than clade II from West Africa (8). The epidemiological and clinical characteristics of the different clades are summarized in Table 1 (9).
The epidemic caused by MPV clade Ib emerged in 2023 in the Democratic Republic of Congo and surrounding countries (10). In 2024–2025, cases were reported outside of Africa, such as in the USA (11) and in Europe, with cases observed in travelers returning from endemic areas (12), as well as in autochthonous (13) and nosocomial (14) cases. Cases of MPV clade Ib infection were reported from December 2025 onwards in the territories of the Southwest Indian Ocean, originating from the coastal region of Mahajanga in Madagascar, where more than two hundred cases were confirmed (Figure 1, 15) . Other cases of measles were also reported outside of Madagascar, notably in the Comoros, where 16 cases were confirmed, primarily on Grande Comore (data as of 19/02/2026, 16).
Table 1. Epidemiology of MPOX virus clades (adapted from Beiras et al. , 2025, 9)
* Men having sex with men.
On January 8, 2026, the first case was diagnosed in Mayotte. Nine additional cases have been confirmed since then (15). On January 22, 2026, the first case of medullary pulmonary encephalopathy (MPE) caused by clade Ib was confirmed in Réunion (17) . This patient, a resident of Réunion, was referred to the University Hospital of Réunion by his general practitioner after a stay in Madagascar. This introduction of MPV into the Indian Ocean territories was the subject of a Public Health Information Message (PHI) from the French Directorate General of Health (DGS) on February 5, 2026, describing a rapidly evolving epidemiological situation in Madagascar that could lead to imported cases throughout the country (18).
Documented cases in Mayotte and Réunion
In Mayotte
Between January 8 and February 12, 2026, ten confirmed cases of medullary tuberculosis (MPO) were identified in Mayotte. Clade Ib was confirmed in the first cases by the National Reference Center (CNR) for Orthopoxviruses (French Armed Forces Biomedical Research Institute, Brétigny-sur-Orge). All cases involved adults, and a history of sexual contact was reported in all cases. The individuals presented with polymorphic skin or mucocutaneous rashes. No severe cases were observed.
Among the initial cases, three were linked to recent stays in Madagascar and were classified as imported. These introductions resulted in four secondary transmissions, including sexual and/or intrafamilial transmission. Furthermore, a transmission chain was identified from a secondary case, confirming local tertiary transmission. One autochthonous case, without a formally confirmed epidemiological link, was also identified.
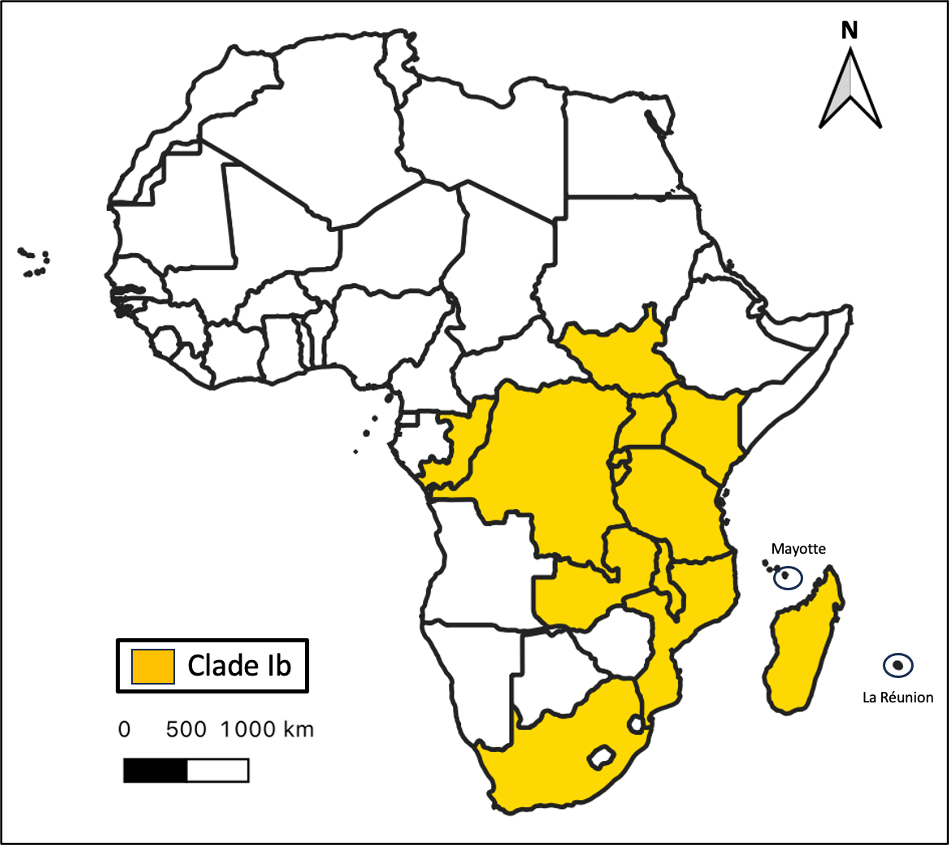
Figure 1. Geographic distribution of countries in which cases of autochthonous MPV clade Ib infections have been reported in the last six weeks. Data from 19/02/2026 from the World Health Organization (WHO, 15).
Investigations were conducted around each case to identify at-risk contacts and implement control measures, including isolation and post-exposure vaccination, which was administered to thirteen people. A targeted preventive vaccination campaign, in line with the recommendations of the High Council for Public Health (HCSP), was also deployed across the country.
In Reunion
Four cases of pyoderma pigmentosum (PPP) were identified in Réunion as of February 20, 2026. The first case involved an individual returning from Madagascar, where they reported unprotected sex. The diagnosis was confirmed after the appearance of genital lesions suggestive of primary syphilis or MPV infection. The clinical course was favorable with symptomatic treatment and dermatological monitoring (teleconsultation). The second case also involved a patient returning from Madagascar, presenting with perineal skin lesions consistent with MPV infection. This second case generated two secondary contacts. No complications were observed in any of the four patients. All additional tests for sexually transmitted infections (STIs) were negative. The four cases of infection were confirmed as MPV clade Ib by the Microbiology Laboratory of the University Hospital of Réunion.
These cases led to the implementation of the usual control measures, including the isolation of the index case and secondary prophylaxis of contacts.
In the laboratory
The handling of samples for virological diagnostic purposes for the search for MPV is described in the operational sampling procedure of the Operational Coordination for Epidemic and Biological Risk (COREB) (19).
Suspected patients presenting with clinical signs suggestive of MPV infection with high-risk exposure (return from travel to an endemic area, multiple sexual partners, MSM) are systematically tested. It is recommended to perform swabbing and/or biopsy of skin lesions on mucous membranes (20). For probable cases, testing is not systematic, but a mandatory reporting form (DO) must be submitted to the Regional Health Agency (ARS).
MPV is a class 3 biological agent (listed in the Microorganisms and Toxins or MOT list, 21), requiring decontamination of samples and transport in triple packaging (category B biological substance, UN3373 standard, packing instruction P650) (20). Virological diagnosis is performed by nucleic acid amplification tests (NAATs) based on Orthopoxvirus -type PCR followed by confirmation by MPV-specific PCR or directly by MPV-specific PCR. If positive, typing (clade determination) by PCR is performed to discriminate the clade responsible for the lesions (Ia, Ib, or II), by targeting specific sequences of the viral genes G2R, C3L, and C1B (22).
The risk of contamination from biological samples varies depending on the type of sample, as well as the transport and analysis conditions. There is a significant risk of contamination from skin and mucous membrane (SEM) and ENT (ear, nose, and throat) samples, whereas the risk is considered low for blood and urine samples that undergo standard handling procedures.
Control, treatment and prevention strategies
In France, the mpox response strategy is based on prevention (vaccination), detection and confirmation of cases by molecular biology (NAAT and sequencing), reporting and management of the index case and contacts (18).
Vesicular eruptions that may suggest MPV infection should prompt consideration of other differential diagnoses such as hand-foot-and-mouth disease ( enterovirus ), chickenpox, herpes, secondary syphilis, and other non-infectious bullous dermatoses (24). Furthermore, systematic STI screening should be performed in patients who have unprotected sex (HIV, HBV, HCV, and syphilis serology, and Neisseria gonorrhoeae and Chlamydia trachomatis PCR on first - void urine) (25).
Patient treatment is based on symptomatic management (level 1 to 3 analgesics) and, depending on infectious advice, specific treatment may be considered (tecovirimat, brincidofovir, cidofovir, immunoglobulins) depending on the patient's comorbidities (26).
A prophylactic vaccine against MPV exists and consists of a third - generation smallpox vaccine (8). Pre-exposure prophylaxis against MPV is indicated for men who have sex with men (MSM) and transgender individuals with multiple sexual partners, sex workers, professionals in sexual activity settings, and people living in the same household as at-risk individuals. The vaccine has demonstrated good efficacy in children who may develop severe forms of the disease (27). Post-exposure prophylaxis is also recommended around cases (26). Furthermore, the HCSP recommends vaccination for travelers to endemic areas (including Madagascar), namely healthcare workers, humanitarian workers at risk of exposure, people from areas of active transmission, and expatriates (depending on living conditions in the country) (28). For pregnant women, children, and immunocompromised patients, travel should be postponed, or if travel is maintained, vaccination should be offered to immunocompromised individuals, but based on current knowledge, it is not recommended for pregnant women and children. Due to the incomplete effectiveness of vaccination, other preventive measures rely on hand hygiene, isolation of cases, limiting contact with individuals likely to be infected with MPV, and avoiding sharing bedding, objects, or linens with these individuals (18).
Conclusions and perspectives
Numerous cases of pyoderma gangrenosum (PGB) linked to MPV clade Ib have recently been confirmed in the Southwest Indian Ocean region, requiring increased clinical vigilance and surveillance. This epidemiological situation is constantly evolving; to date (February 20, 2026), ten cases have been confirmed in Mayotte and four in Réunion. MPV infections are notifiable diseases that must be reported without delay to the Regional Health Agencies (ARS), without waiting for typing (23).
The spread of the measles epidemic to these satellite islands of the African continent, located in the southwestern Indian Ocean, underscores the interconnectedness of these territories, linked to population flows facilitated in particular by air traffic. It is essential to organize a coordinated response in the area, in conjunction with local, regional (Regional Health Agencies - ARS), national (Directorate General of Health/Ministry of Health), and international (WHO/IOC or Indian Ocean Commission) health authorities. The regional epidemiological situation, combined with the epidemic potential of this virus, necessitates taking this progression of the epidemic into account in order to prevent its potential spread to mainland France.
Bibliography
Introduction of the mpox virus to the islands of the Southwest Indian Ocean
Olivia Vong
February 25, 2026

Introduction of the mpox virus to the islands of the Southwest Indian Ocean
Ambroise Mercier 1,2 , Mélissa Bérot 3 , Sylvain Brisse 4 , Marie-Pierre Moiton 5 , Olivier Belmonte 1 , Laurent Souply 1 , Guillaume Miltgen 1,2,6
- Bacteriology Laboratory, Félix Guyon University Hospital, Saint-Denis, La Réunion, France.
- UMR Infectious Processes in Tropical Island Environments (PIMIT), CNRS 9192, INSERM U1187, IRD 249, University of Reunion Island, Saint-Denis, Reunion Island, France.
- Department of Security and Health Emergencies, Regional Health Agency of Mayotte, Mamoudzou, France.
- Biodiversity and Epidemiology of Pathogenic Bacteria Unit, Pasteur Institute, Paris, France.
- Infectious Diseases Department, University Hospital of Reunion Island, Saint-Denis, Reunion Island, France.
- Regional Centre for Antibiotic Therapy (CRAtb) of Reunion Island, Saint-Pierre, France.
Introduction
Mpox, or mpox B, is a zoonotic disease caused by the mpox virus ( mpox virus [MPV], formerly monkeypox virus ). This virus belongs to the genus Orthopoxvirus (family Poxviridae ), which includes other members such as smallpox virus (variola virus [VARV]), vaccinia virus ( vaccinia virus [VACV]), and cowpox virus (CPV) (1). This zoonosis, less severe than smallpox, affects rodents (fungiur rats, giant pouched rats, dormice, etc. ) and primates (including humans) (2). Since the eradication of smallpox in 1977 through vaccination, MPV and CPV are the most studied orthopoxviruses due to their high epidemic potential (3).
MPV, initially distributed in Africa, has been responsible for numerous mycobacterium tuberculosis epidemics worldwide. This virus was first discovered in a pet shop in Copenhagen in 1958 in primates (4). It was not until 1970 that it was described in humans, in the Democratic Republic of Congo (5). Since then, other cases have been reported in Africa. The first extra-African epidemic occurred in 2003 in the United States, where domestic prairie dogs were infected through contact with infected savannah worms from Ghana (6). In 2022, a new global epidemic occurred, characterized by human-to-human transmission via direct contact with the skin and mucous membrane lesions of infected patients, primarily men who have sex with men (MSM) (7).
Phylogenetically, MPV is divided into two clades based on genomic sequences, with clade I from Central Africa exhibiting higher severity and mortality than clade II from West Africa (8). The epidemiological and clinical characteristics of the different clades are summarized in Table 1 (9).
The epidemic caused by MPV clade Ib emerged in 2023 in the Democratic Republic of Congo and surrounding countries (10). In 2024–2025, cases were reported outside of Africa, such as in the USA (11) and in Europe, with cases observed in travelers returning from endemic areas (12), as well as in autochthonous (13) and nosocomial (14) cases. Cases of MPV clade Ib infection were reported from December 2025 onwards in the territories of the Southwest Indian Ocean, originating from the coastal region of Mahajanga in Madagascar, where more than two hundred cases were confirmed (Figure 1, 15) . Other cases of measles were also reported outside of Madagascar, notably in the Comoros, where 16 cases were confirmed, primarily on Grande Comore (data as of 19/02/2026, 16).
Table 1. Epidemiology of MPOX virus clades (adapted from Beiras et al. , 2025, 9)
| Clade | Period | Distribution | Transmission | Epidemics | Population |
| Ia | 1970-2024 | Central Africa | Zoonotics | Low incidence | Children |
| Ib | 2023 | DRC | Interhuman | Emergence in 2023 | Adults and children |
| IIa | 1970-2018 | West Africa | Zoonotics | Endemic in the USA in 2003 | Adults and children |
| IIb line A | 2017-2024 | Nigeria | Zoonototics + interhuman | Nigeria Epidemic 2017 | Adults |
| IIb line B | 2022-2023 | Worldwide distribution | Sexual | Global pandemic 2022 | MSH* |
On January 8, 2026, the first case was diagnosed in Mayotte. Nine additional cases have been confirmed since then (15). On January 22, 2026, the first case of medullary pulmonary encephalopathy (MPE) caused by clade Ib was confirmed in Réunion (17) . This patient, a resident of Réunion, was referred to the University Hospital of Réunion by his general practitioner after a stay in Madagascar. This introduction of MPV into the Indian Ocean territories was the subject of a Public Health Information Message (PHI) from the French Directorate General of Health (DGS) on February 5, 2026, describing a rapidly evolving epidemiological situation in Madagascar that could lead to imported cases throughout the country (18).
Documented cases in Mayotte and Réunion
In Mayotte
Between January 8 and February 12, 2026, ten confirmed cases of medullary tuberculosis (MPO) were identified in Mayotte. Clade Ib was confirmed in the first cases by the National Reference Center (CNR) for Orthopoxviruses (French Armed Forces Biomedical Research Institute, Brétigny-sur-Orge). All cases involved adults, and a history of sexual contact was reported in all cases. The individuals presented with polymorphic skin or mucocutaneous rashes. No severe cases were observed.
Among the initial cases, three were linked to recent stays in Madagascar and were classified as imported. These introductions resulted in four secondary transmissions, including sexual and/or intrafamilial transmission. Furthermore, a transmission chain was identified from a secondary case, confirming local tertiary transmission. One autochthonous case, without a formally confirmed epidemiological link, was also identified.

Figure 1. Geographic distribution of countries in which cases of autochthonous MPV clade Ib infections have been reported in the last six weeks. Data from 19/02/2026 from the World Health Organization (WHO, 15).
Investigations were conducted around each case to identify at-risk contacts and implement control measures, including isolation and post-exposure vaccination, which was administered to thirteen people. A targeted preventive vaccination campaign, in line with the recommendations of the High Council for Public Health (HCSP), was also deployed across the country.
In Reunion
Four cases of pyoderma pigmentosum (PPP) were identified in Réunion as of February 20, 2026. The first case involved an individual returning from Madagascar, where they reported unprotected sex. The diagnosis was confirmed after the appearance of genital lesions suggestive of primary syphilis or MPV infection. The clinical course was favorable with symptomatic treatment and dermatological monitoring (teleconsultation). The second case also involved a patient returning from Madagascar, presenting with perineal skin lesions consistent with MPV infection. This second case generated two secondary contacts. No complications were observed in any of the four patients. All additional tests for sexually transmitted infections (STIs) were negative. The four cases of infection were confirmed as MPV clade Ib by the Microbiology Laboratory of the University Hospital of Réunion.
These cases led to the implementation of the usual control measures, including the isolation of the index case and secondary prophylaxis of contacts.
In the laboratory
The handling of samples for virological diagnostic purposes for the search for MPV is described in the operational sampling procedure of the Operational Coordination for Epidemic and Biological Risk (COREB) (19).
Suspected patients presenting with clinical signs suggestive of MPV infection with high-risk exposure (return from travel to an endemic area, multiple sexual partners, MSM) are systematically tested. It is recommended to perform swabbing and/or biopsy of skin lesions on mucous membranes (20). For probable cases, testing is not systematic, but a mandatory reporting form (DO) must be submitted to the Regional Health Agency (ARS).
MPV is a class 3 biological agent (listed in the Microorganisms and Toxins or MOT list, 21), requiring decontamination of samples and transport in triple packaging (category B biological substance, UN3373 standard, packing instruction P650) (20). Virological diagnosis is performed by nucleic acid amplification tests (NAATs) based on Orthopoxvirus -type PCR followed by confirmation by MPV-specific PCR or directly by MPV-specific PCR. If positive, typing (clade determination) by PCR is performed to discriminate the clade responsible for the lesions (Ia, Ib, or II), by targeting specific sequences of the viral genes G2R, C3L, and C1B (22).
The risk of contamination from biological samples varies depending on the type of sample, as well as the transport and analysis conditions. There is a significant risk of contamination from skin and mucous membrane (SEM) and ENT (ear, nose, and throat) samples, whereas the risk is considered low for blood and urine samples that undergo standard handling procedures.
Control, treatment and prevention strategies
In France, the mpox response strategy is based on prevention (vaccination), detection and confirmation of cases by molecular biology (NAAT and sequencing), reporting and management of the index case and contacts (18).
Vesicular eruptions that may suggest MPV infection should prompt consideration of other differential diagnoses such as hand-foot-and-mouth disease ( enterovirus ), chickenpox, herpes, secondary syphilis, and other non-infectious bullous dermatoses (24). Furthermore, systematic STI screening should be performed in patients who have unprotected sex (HIV, HBV, HCV, and syphilis serology, and Neisseria gonorrhoeae and Chlamydia trachomatis PCR on first - void urine) (25).
Patient treatment is based on symptomatic management (level 1 to 3 analgesics) and, depending on infectious advice, specific treatment may be considered (tecovirimat, brincidofovir, cidofovir, immunoglobulins) depending on the patient's comorbidities (26).
A prophylactic vaccine against MPV exists and consists of a third - generation smallpox vaccine (8). Pre-exposure prophylaxis against MPV is indicated for men who have sex with men (MSM) and transgender individuals with multiple sexual partners, sex workers, professionals in sexual activity settings, and people living in the same household as at-risk individuals. The vaccine has demonstrated good efficacy in children who may develop severe forms of the disease (27). Post-exposure prophylaxis is also recommended around cases (26). Furthermore, the HCSP recommends vaccination for travelers to endemic areas (including Madagascar), namely healthcare workers, humanitarian workers at risk of exposure, people from areas of active transmission, and expatriates (depending on living conditions in the country) (28). For pregnant women, children, and immunocompromised patients, travel should be postponed, or if travel is maintained, vaccination should be offered to immunocompromised individuals, but based on current knowledge, it is not recommended for pregnant women and children. Due to the incomplete effectiveness of vaccination, other preventive measures rely on hand hygiene, isolation of cases, limiting contact with individuals likely to be infected with MPV, and avoiding sharing bedding, objects, or linens with these individuals (18).
Conclusions and perspectives
Numerous cases of pyoderma gangrenosum (PGB) linked to MPV clade Ib have recently been confirmed in the Southwest Indian Ocean region, requiring increased clinical vigilance and surveillance. This epidemiological situation is constantly evolving; to date (February 20, 2026), ten cases have been confirmed in Mayotte and four in Réunion. MPV infections are notifiable diseases that must be reported without delay to the Regional Health Agencies (ARS), without waiting for typing (23).
The spread of the measles epidemic to these satellite islands of the African continent, located in the southwestern Indian Ocean, underscores the interconnectedness of these territories, linked to population flows facilitated in particular by air traffic. It is essential to organize a coordinated response in the area, in conjunction with local, regional (Regional Health Agencies - ARS), national (Directorate General of Health/Ministry of Health), and international (WHO/IOC or Indian Ocean Commission) health authorities. The regional epidemiological situation, combined with the epidemic potential of this virus, necessitates taking this progression of the epidemic into account in order to prevent its potential spread to mainland France.
Bibliography
- Alakunle E, Kolawole D, Diaz-Cánova D, Alele F, Adegboye O, Moens U, et al. A comprehensive review of monkeypox virus and mpox characteristics. Front Cell Infect Microbiol. 2024;14:1360586.
- Doty JB, Malekani JM, Kalemba LN, Stanley WT, Monroe BP, Nakazawa YU, et al. Assessing Monkeypox Virus Prevalence in Small Mammals at the Human-Animal Interface in the Democratic Republic of the Congo. Viruses. 2017;9(10):283.
- Yahya G, Mohamed NH, Wadan AHS, Castro EM, Kamel A, Abdelmoaty AA, et al. The Emerging Threat of Monkeypox: An Updated Overview. Viruses. 2026;18(1):69.
- Parker S, Buller RM. A review of experimental and natural infections of animals with monkeypox virus between 1958 and 2012. Future Virol. 2013;8(2):129–57.
- Bunge EM, Hoet B, Chen L, Lienert F, Weidenthaler H, Baer LR, et al. The changing epidemiology of human monkeypox-A potential threat? A systematic review. PLoS Negl Too Say. 2022;16(2):e0010141.
- Reed KD, Melski JW, Graham MB, Regnery RL, Sotir MJ, Wegner MV, et al. The detection of monkeypox in humans in the Western Hemisphere. N Engl J Med. 2004;350(4):342–50.
- Suñer C, Ubals M, Tarín-Vicente EJ, Mendoza A, Alemany A, Hernández-Rodríguez Á, et al. Viral dynamics in patients with monkeypox infection: a prospective cohort study in Spain. Lancet Infect Dis. 2023;23(4):445–53.
- Elsayed S, Bondy L, Hanage WP. Monkeypox Virus Infections in Humans. Clin Microbiol Rev. 2022;35(4):e0009222.
- Beiras CG, Malembi E, Escrig-Sarreta R, Ahuka S, Mbala P, Mavoko HM, et al. Competitor outbreaks of mpox in Africa—an update. The Lancet. 2025;405(10472):86–96.
- Satheshkumar PS, Gigante CM, Mbala-Kingebeni P, Nakazawa Y, Anderson M, Balinandi S, et al. Emergence of Clade Ib Monkeypox Virus—Current State of Evidence. Emerg Infect Dis. 2025;31(8):1516–25.
- Tang EC, Haw M, Kulkarni S, Johnson E, Gigante CM, Balanji N, et al. Detection of Community Transmission of Clade Ib Mpox Virus in the United States. NEJM Evid. 2026;5(2):EVIDpha2500319.
- Treutiger CJ, Filén F, Rehn M, Aarum J, Jacks A, Gisslén M, et al. First case of mpox with monkeypox virus clade Ib outside Africa in a returning traveler, Sweden, August 2024: public health measures. Euro Surveillance 2024;29(48):2400740.
- Elsinga J, van de Ven C, de Vries A, Coppoolse H, Petrignani M, van Cleef B, et al. First detection and autochthonous transmission of monkeypox virus clade Ib in the Netherlands, October to November, 2025. Euro Surveill. 2026;31(3):2500958.
- McLoughlin M, Fahey L, Carr M, Caceda B, Igoe D, Dean J, et al. Nosocomial transmission in a monkeypox virus clade Ib outbreak, Ireland, August to October 2025. Euro Surveill. 2025;30(50):2500926.
- Global Mpox Trends [Internet]. World Health Organization. 2026. Available from: https://worldhealthorg.shinyapps.io/mpx_global/
- Monkeypox in the Comoros: 10 confirmed cases, six recovered patients, and 63 identified contacts. Data from the Ministry of Health, Union of the Comoros. Available from: https://la1ere.franceinfo.fr/reunion...s-1668309.html
- Smallpox (mpox): First case identified in Réunion. Réunion Regional Health Agency. 2026. Available from: https://www.lareunion.ars.sante.fr/v...cas-la-reunion
- Circulation of smallpox B in the Indian Ocean. MISP No. 2026-01. Directorate General of Health. Ministry of Health, Family, Autonomy and Persons with Disabilities [Internet]. 2026. Available from: https://sante.gouv.fr/professionnels/article/dgs-urgent
- Mpox virus infection: operational procedure for sampling. COREB practical guide. 2024. Available from: https://www.coreb.infectiologie.com/...pratiques.html
- Fact sheet: Management of biological samples from a patient suspected or confirmed to have monkeypox virus (MPV) infection. Version 4. French Society for Microbiology. 2024. Available from: https://www.sfm-microbiologie.org/ac...keypox-fiches/
- Order of 26 April 2023 establishing the list of microorganisms and toxins provided for in Article L. 5139-1 of the Public Health Code. Official Journal No. 0123 of 28 May 2023. Available from: https://www.legifrance.gouv.fr/jorf/...XT000047597611
- Schuele L, Masirika LM, Udahemuka JC, Siangoli FB, Mbiribindi JB, Ndishimye P, et al. Real-time PCR assay to detect the novel Clade Ib monkeypox virus, September 2023 to May 2024. Euro Surveill. 2024;29(32):2400486.
- List of notifiable diseases. Public Health France. Available from: https://www.santepubliquefrance.fr/m...on-obligatoire
- MONKEYPOX – Aid in dermatological diagnosis and symptomatic treatment. COREB fact sheets. 2022. Available from: https://www.coreb.infectiologie.com/...pratiques.html
- New treatment recommendations for bacterial sexually transmitted infections. French National Authority for Health (HAS). 2025. Available from: https://anrs.fr/actualites/actualite...harge-des-ist/
- Advice on updated preventive measures against monkeypox virus infection for people traveling to the epidemic zone (travelers and healthcare professionals on humanitarian missions). High Council for Public Health. 2024. Available from: file:///C:/Users/143762/Downloads/hcspa20240902_mesdeprvpoulesvoyvisvisdumpo.pdf
- Ladhani SN, Dowell AC, Jones S, Hicks B, Rowe C, Begum J, et al. Early evaluation of the safety, reactogenicity, and immune response after a single dose of modified vaccinia Ankara–Bavaria Nordic vaccine against mpox in children: a national outbreak response. Lancet Infect Dis. 2023;23(9):1042–50.
- Health recommendations for travelers in 2025. High Council for Public Health. Available from: https://www.hcsp.fr/Explore.cgi/Avis...ine?clefr=1439