Previous thread FluView thread here. Sorry for the spotty thread, but there was very little flu reported for 2020/2021 season.
Weekly U.S. Influenza Surveillance Report

Note: CDC is tracking the COVID-19 pandemic in a weekly publication called COVID Data Tracker Weekly Review.
2020-2021 Influenza Season for Week 39, ending October 2, 2021
All data are preliminary and may change as more reports are received.
A description of the CDC influenza surveillance system, including methodology and detailed descriptions of each data component is available on the surveillance methods page.
Additional information on the current and previous influenza seasons for each surveillance component are available on FluView Interactive.
U.S. Virologic Surveillance:
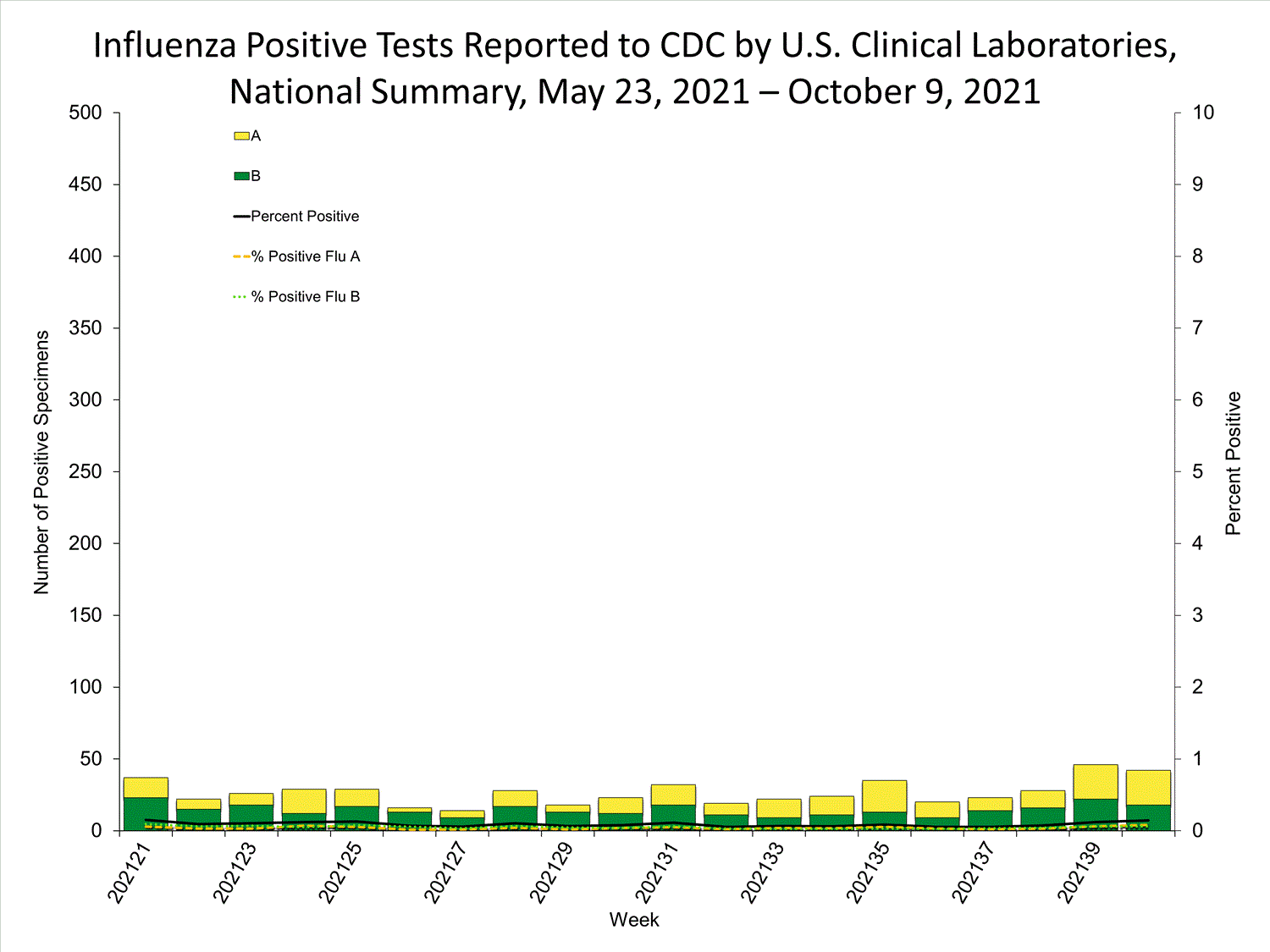
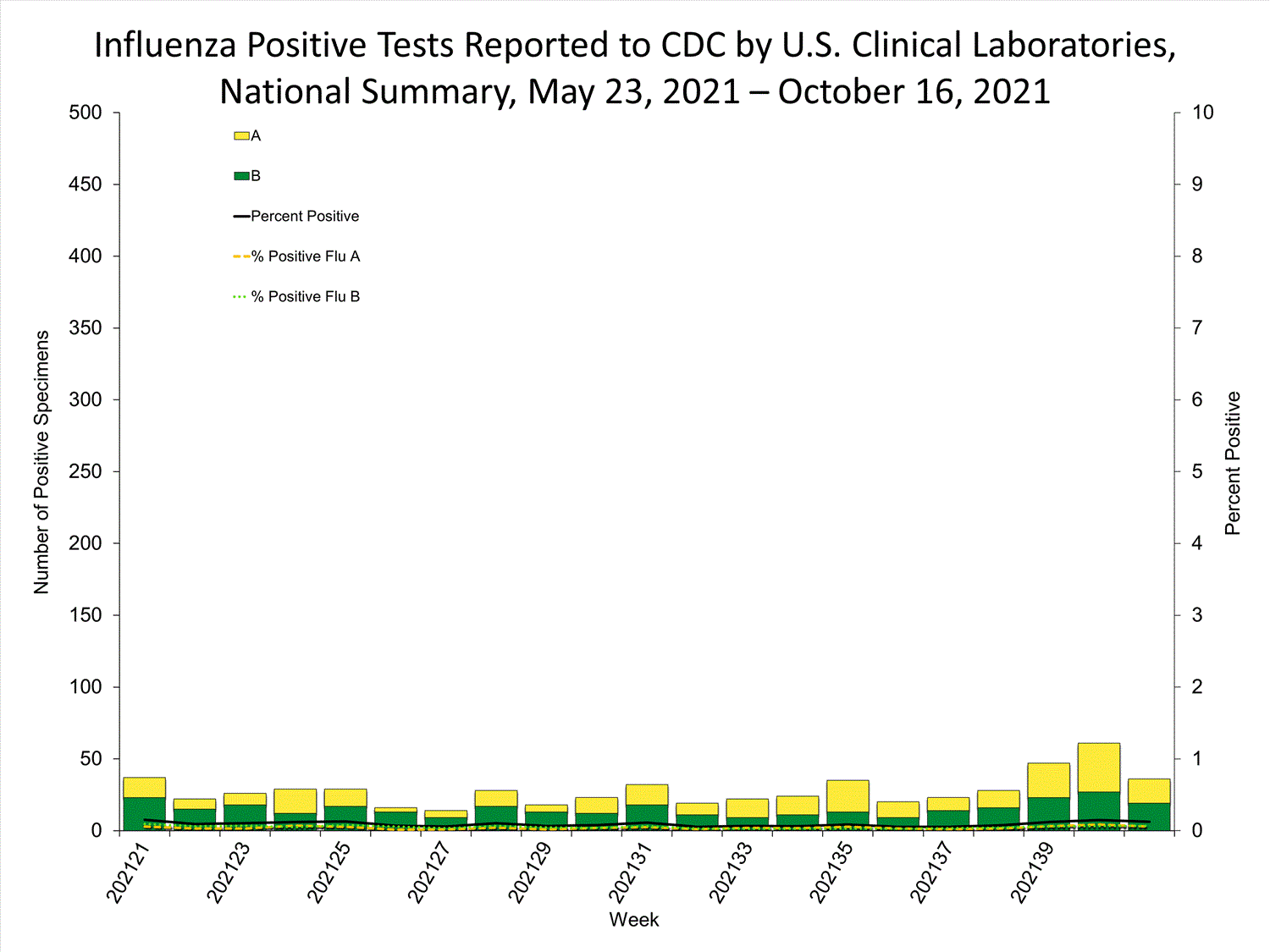
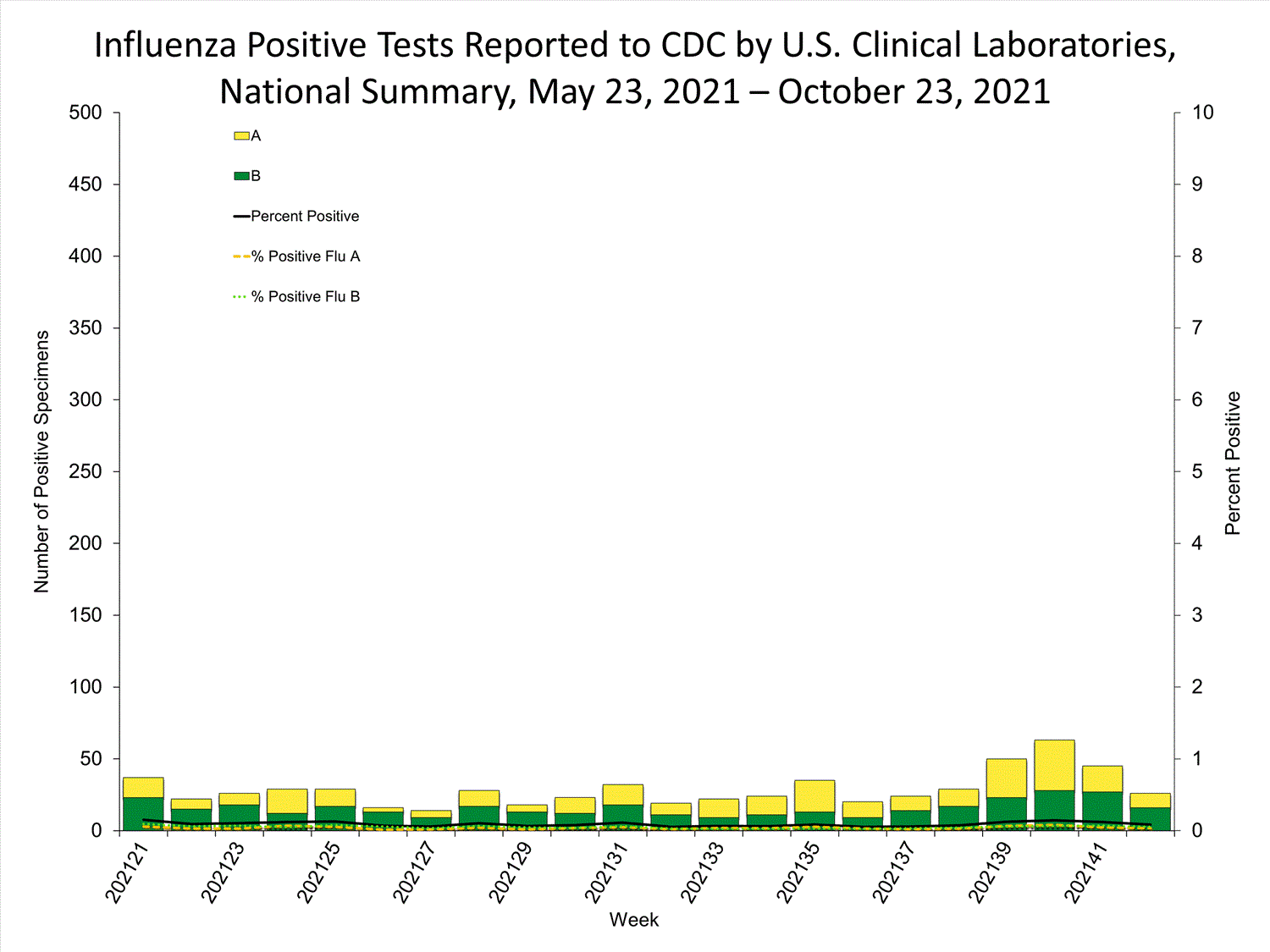
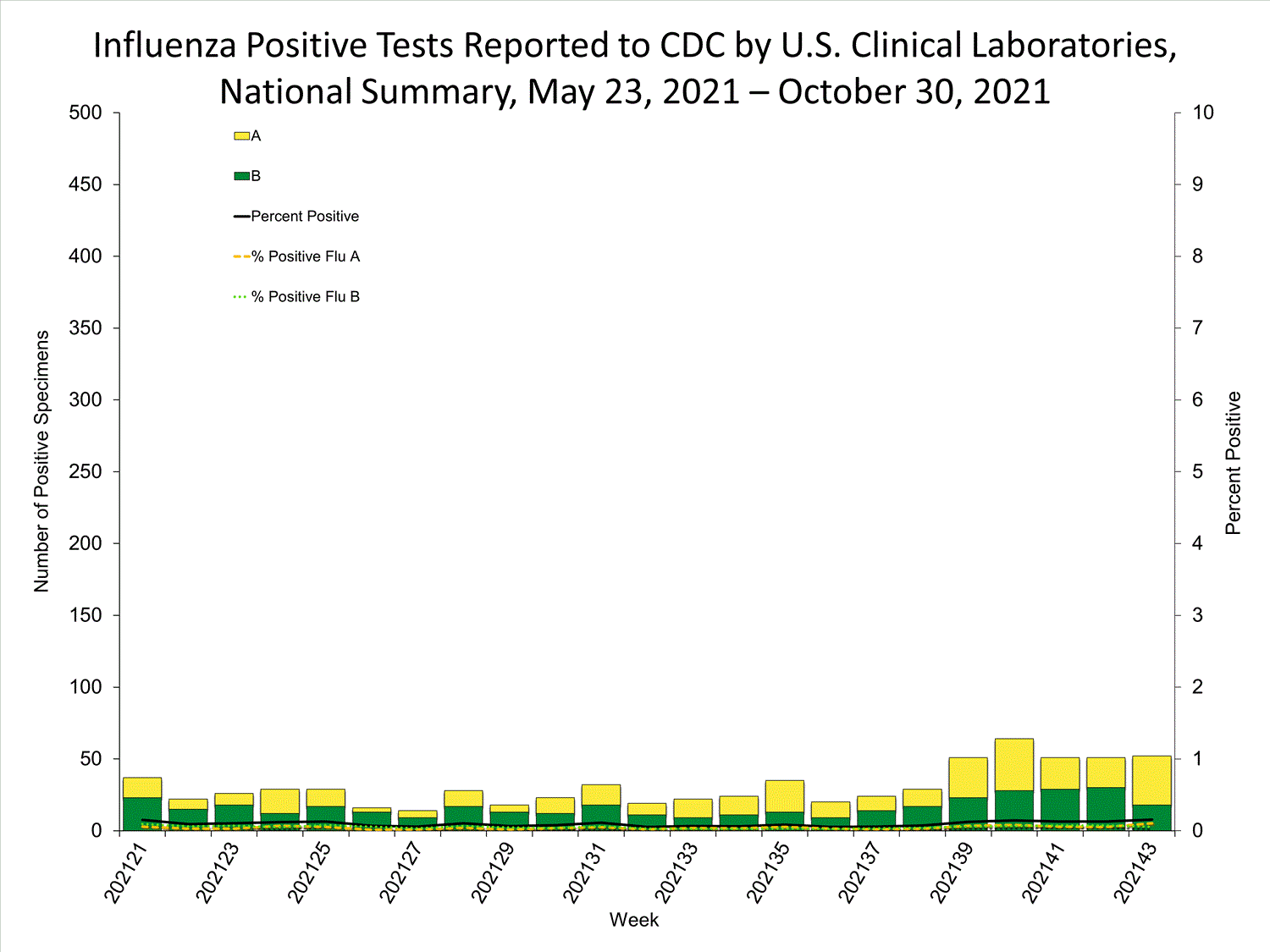
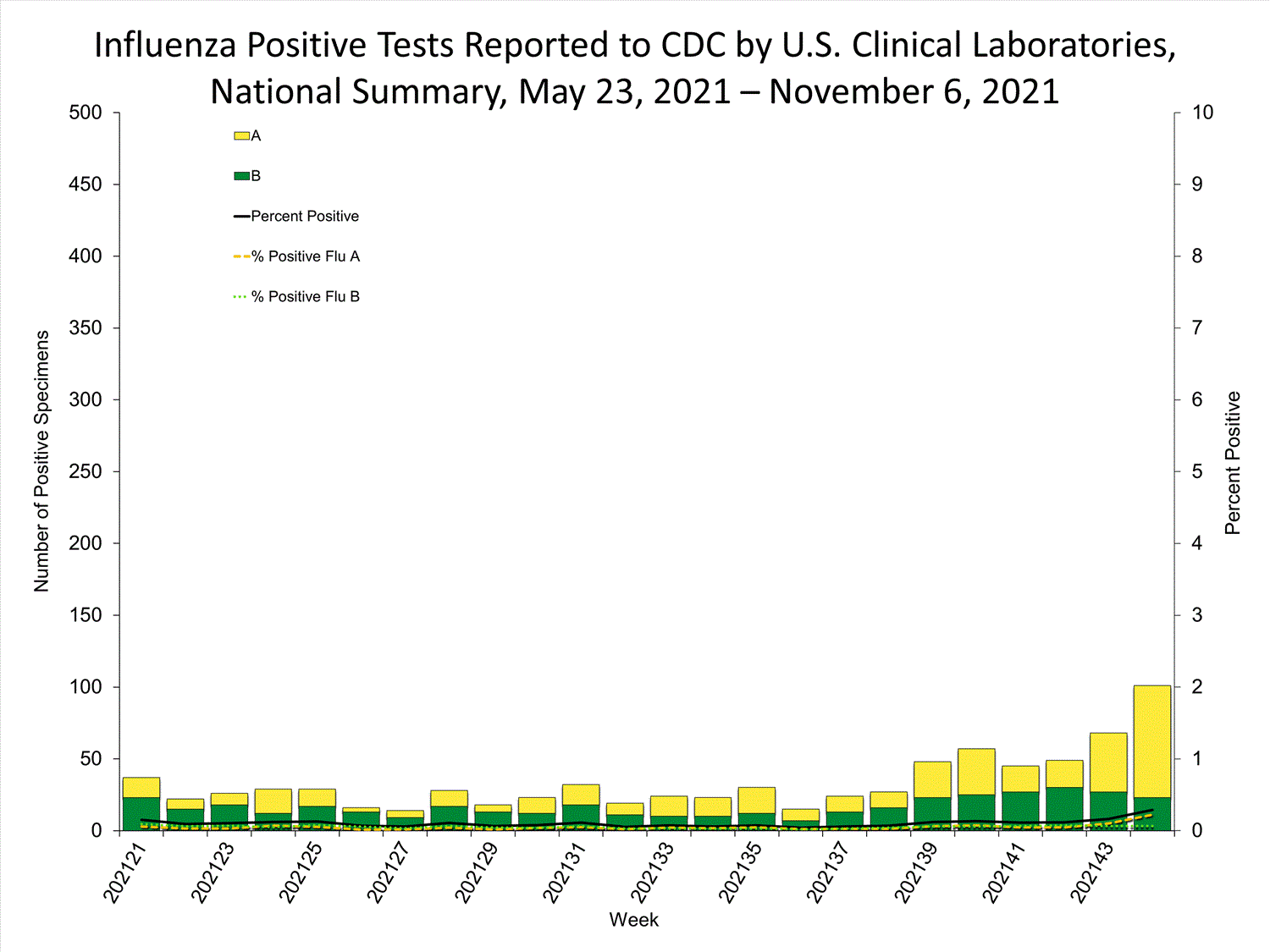
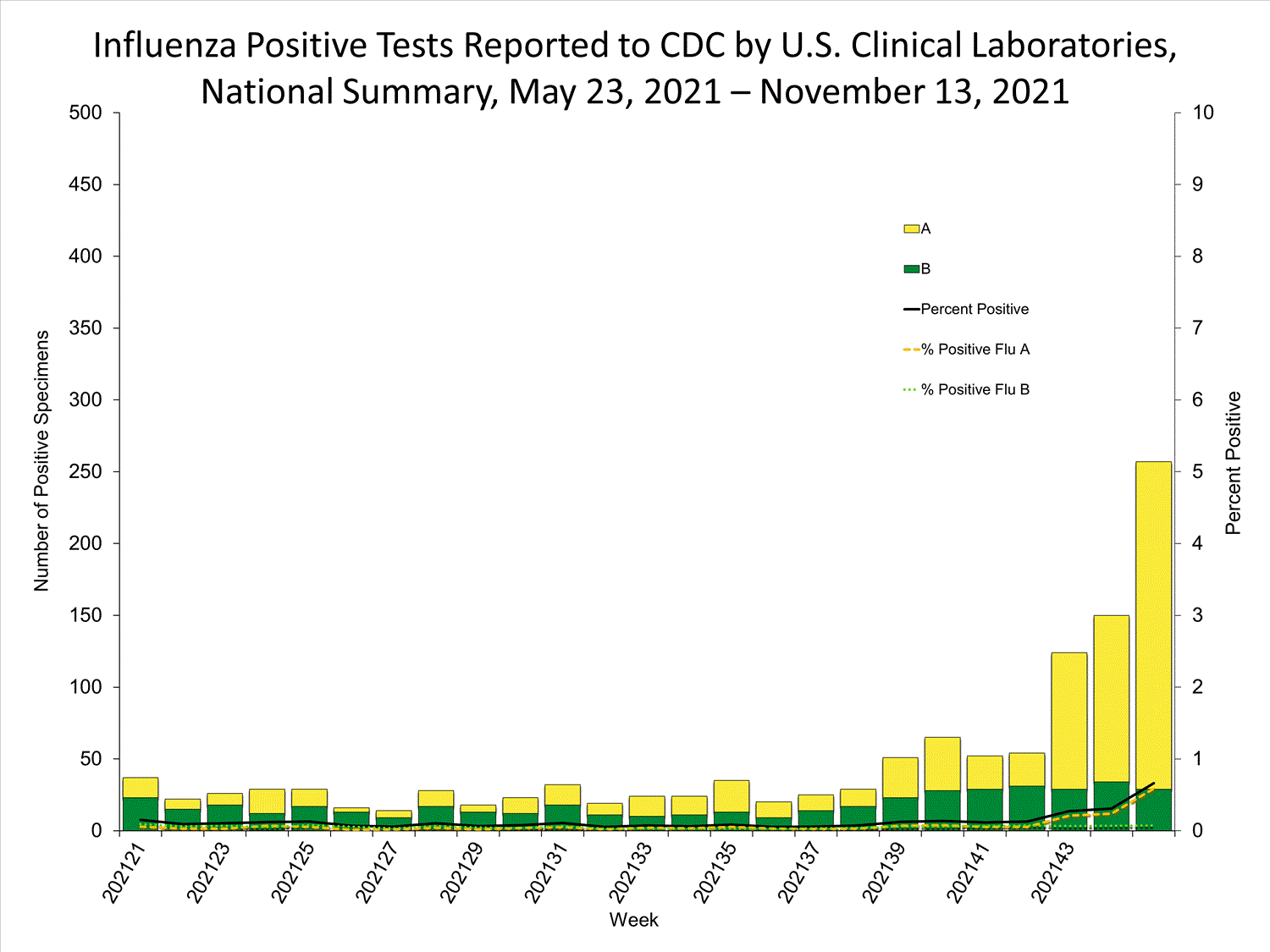
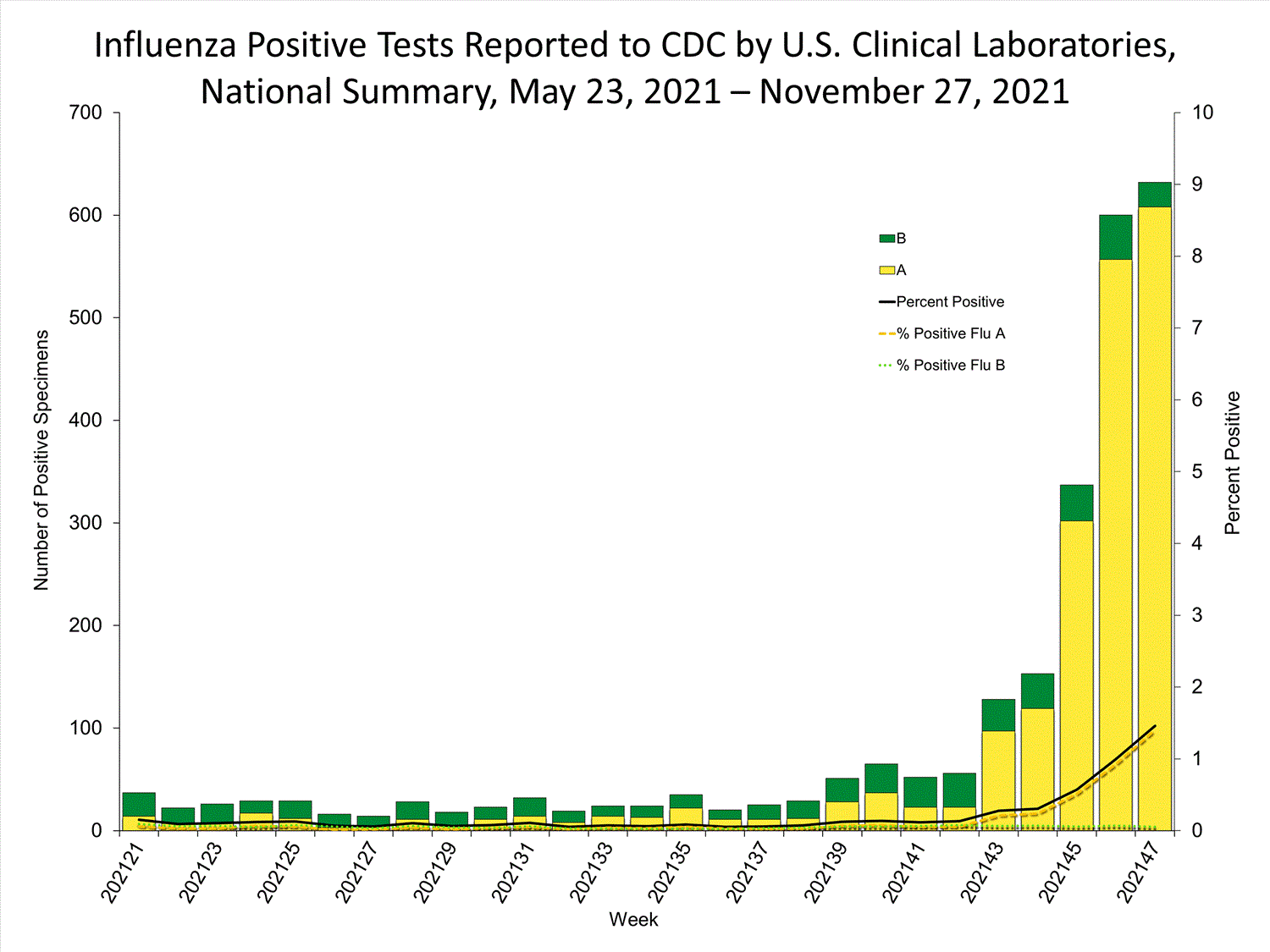
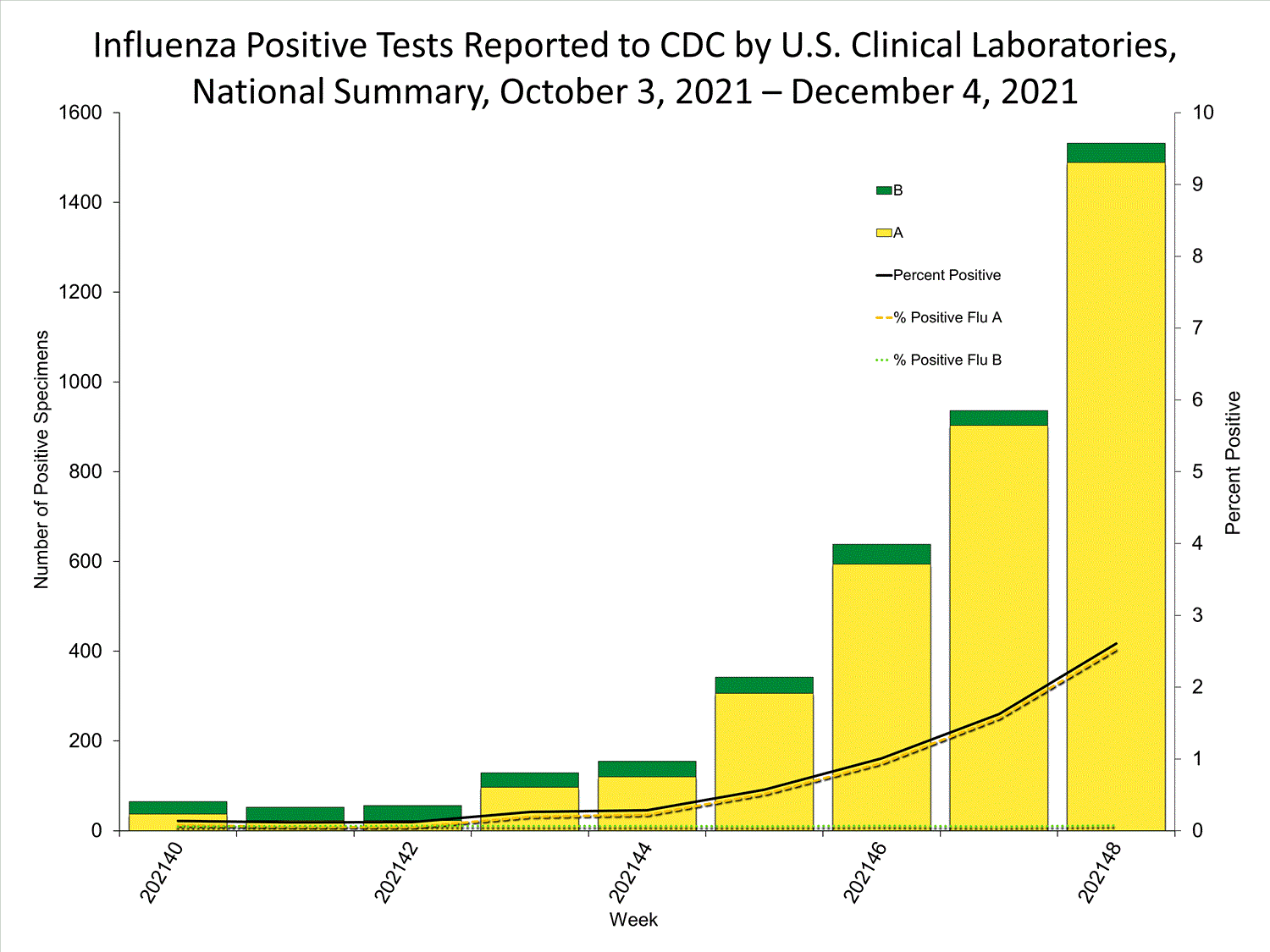
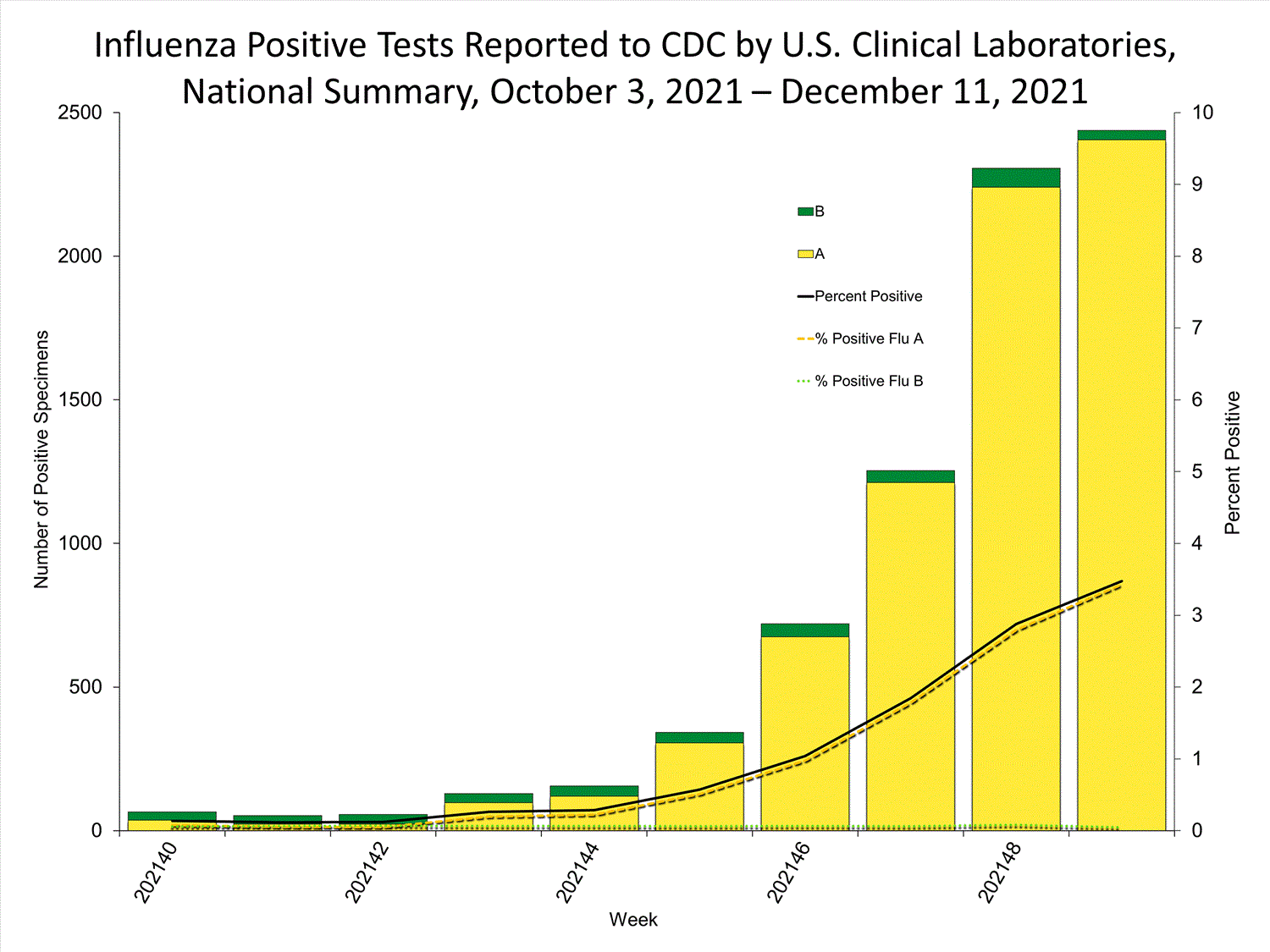
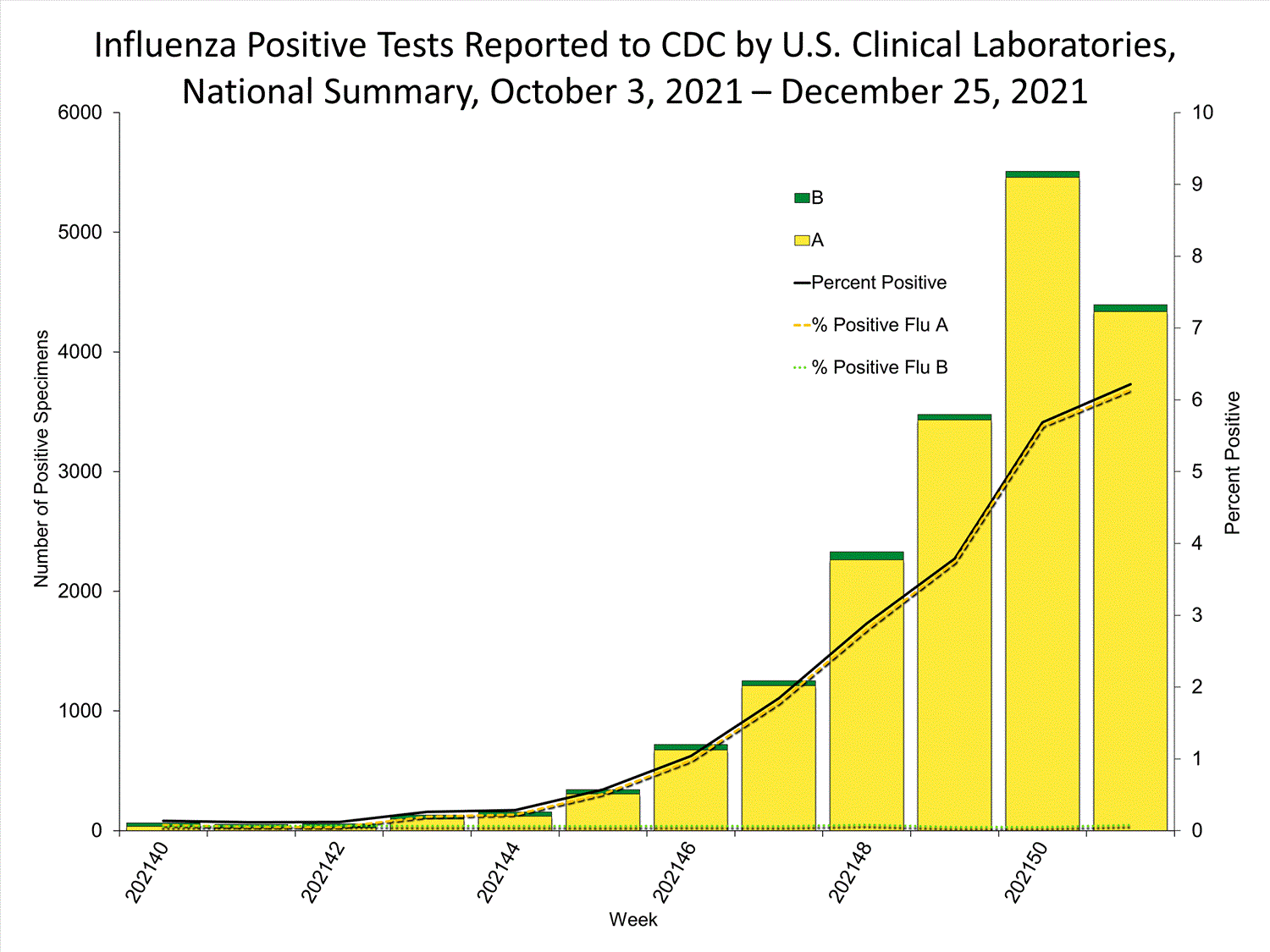
Clinical Laboratories
Data from clinical laboratories (the percentage of specimens tested that are positive for influenza) are used to monitor whether influenza activity is increasing or decreasing.
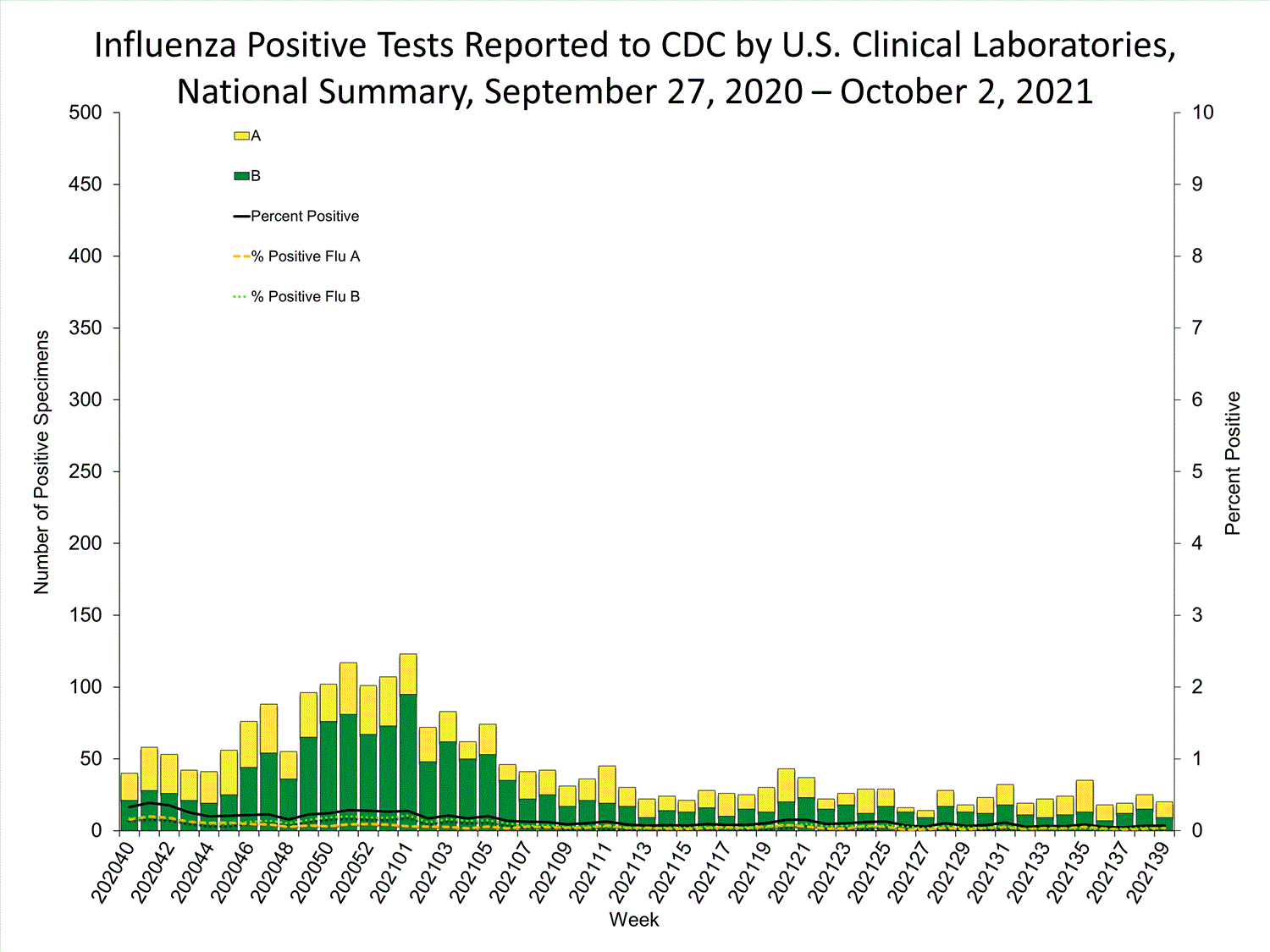
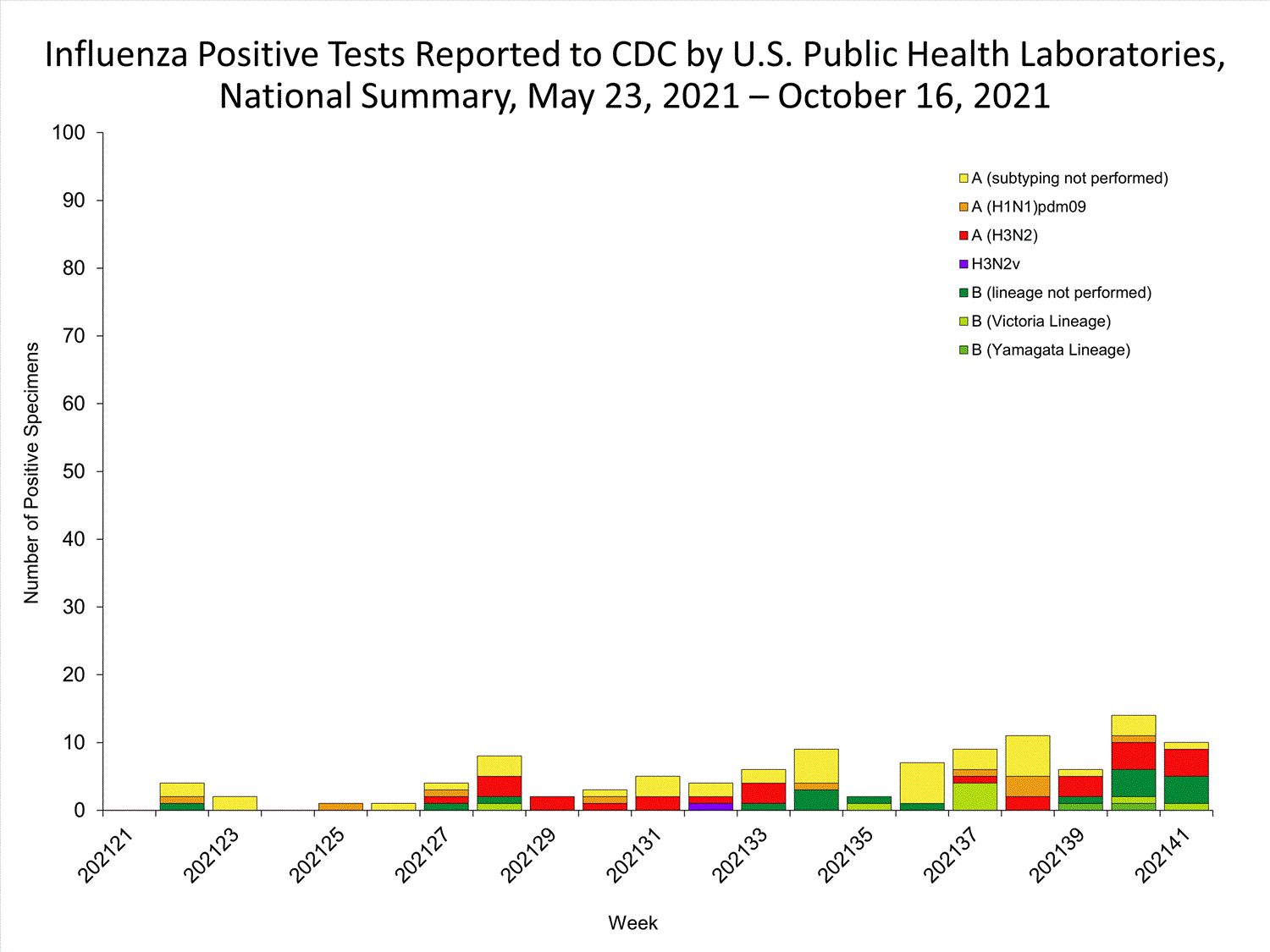
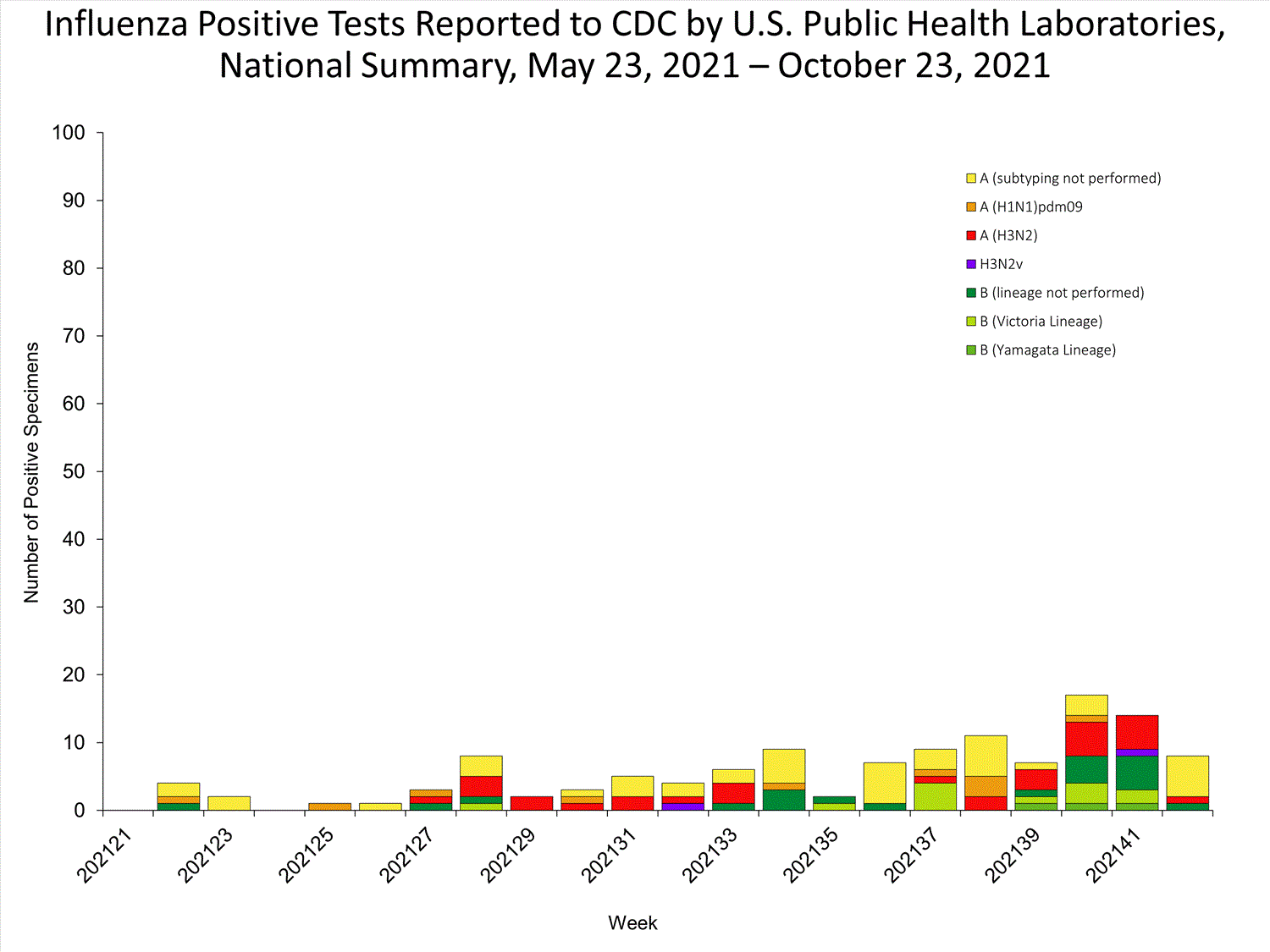
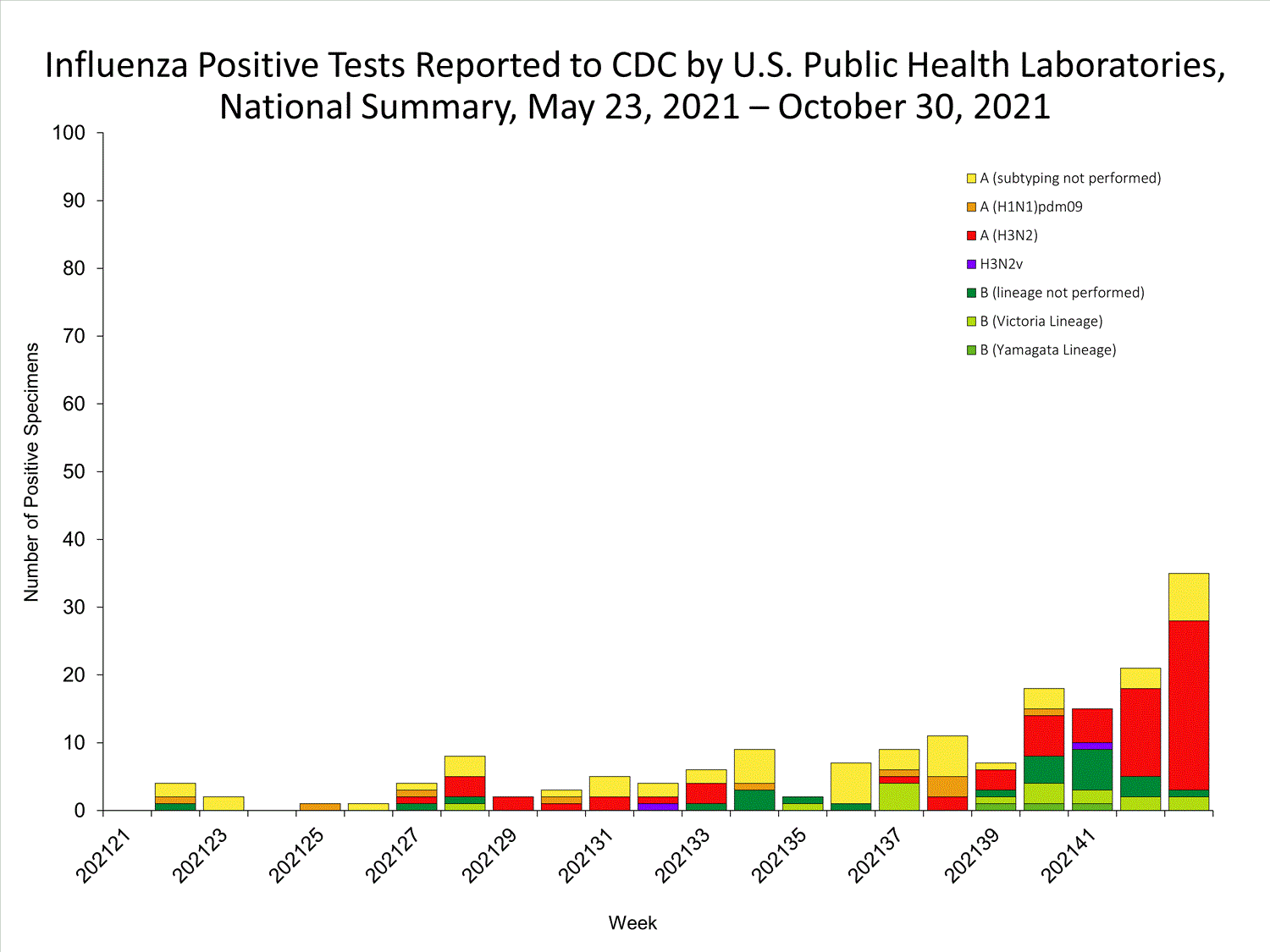
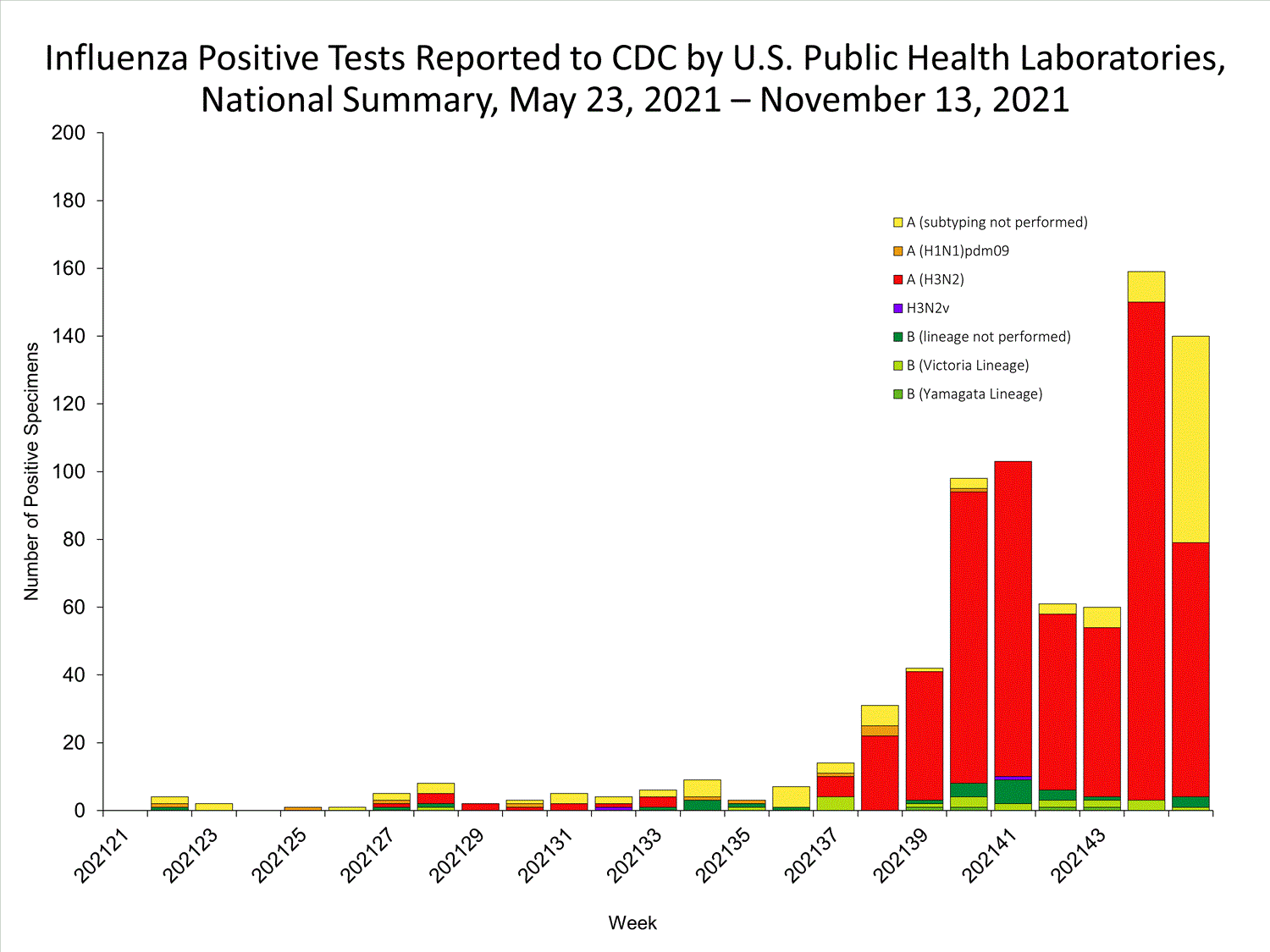
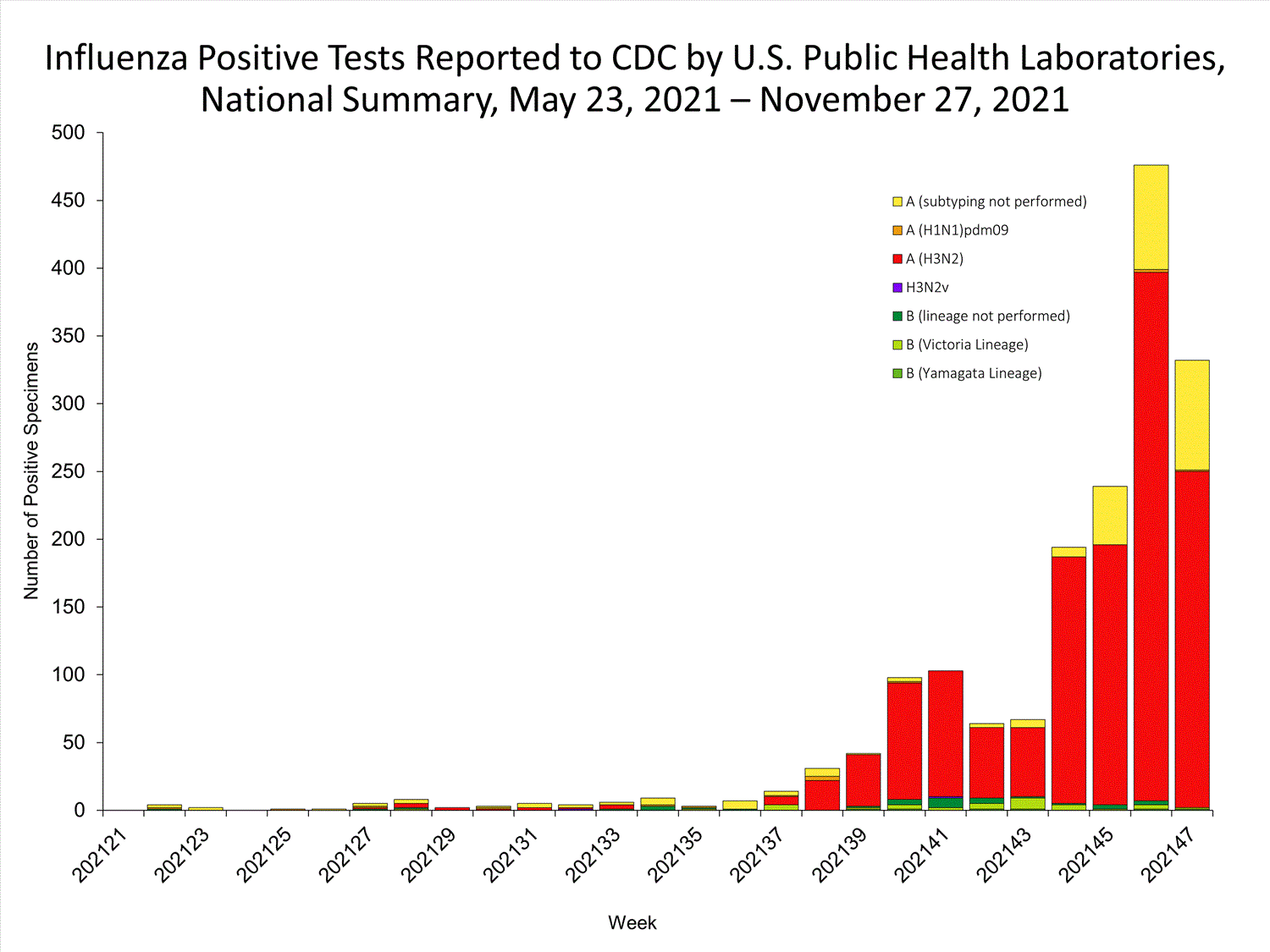
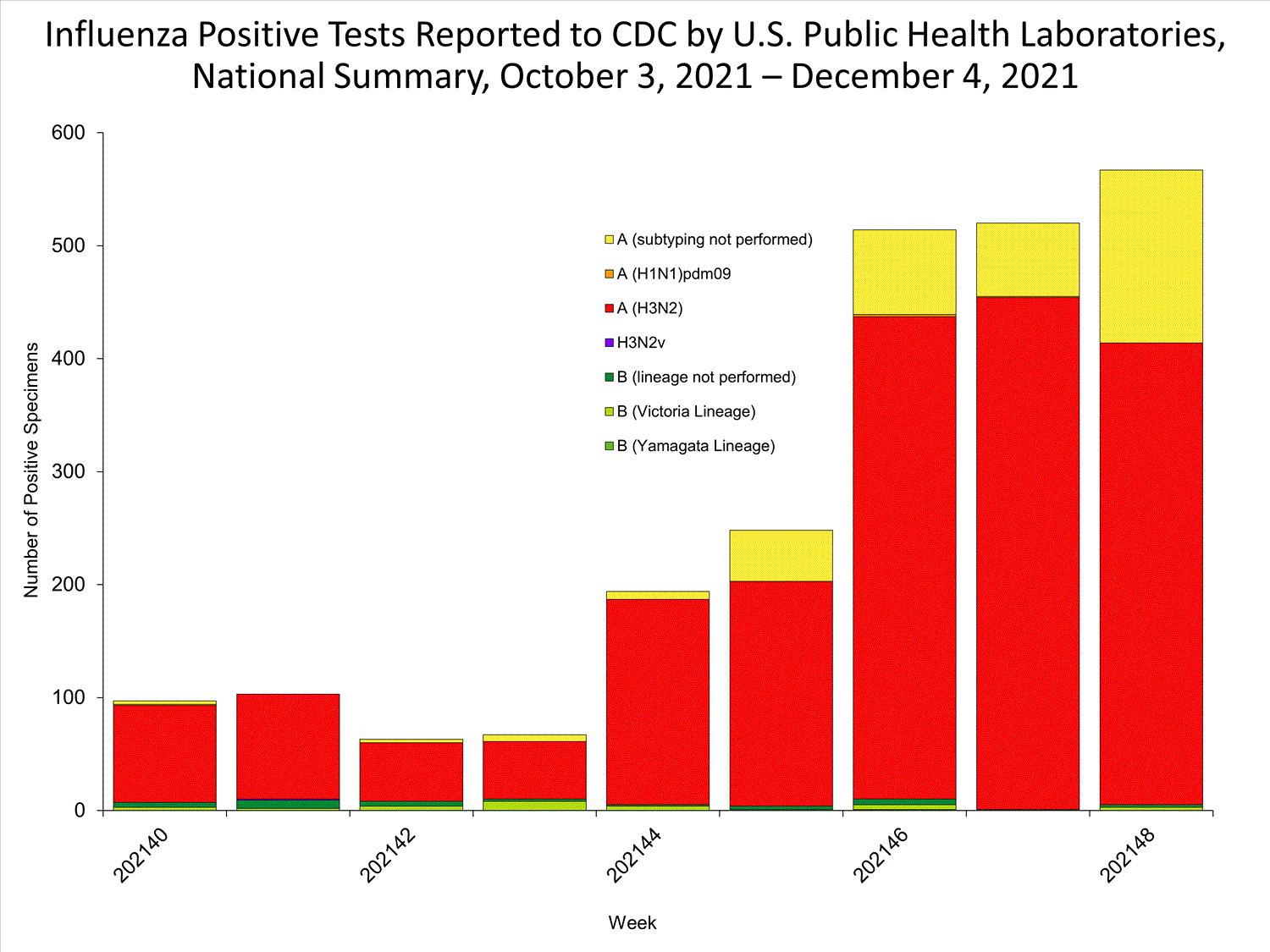
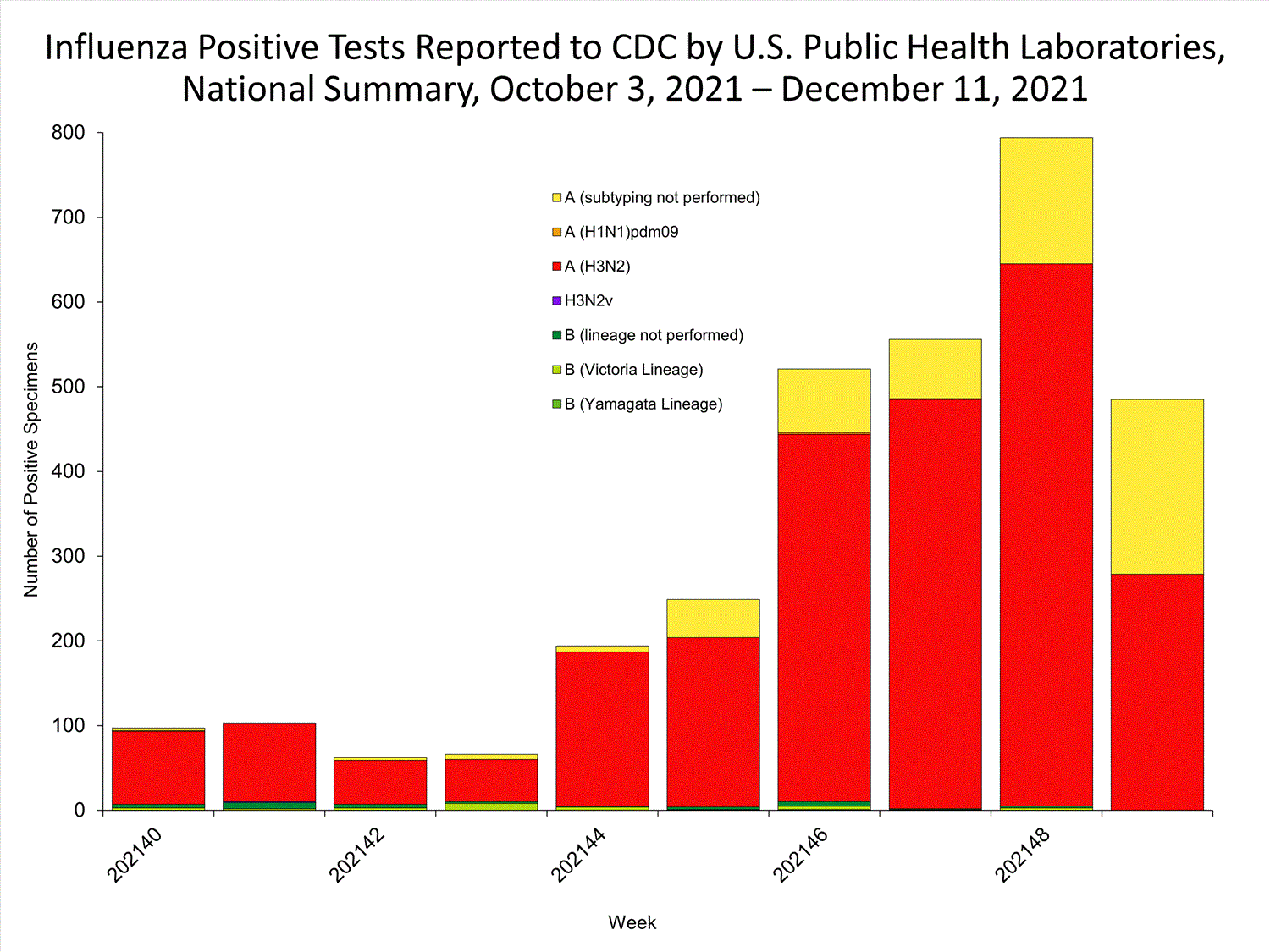
View Chart Data | View Full ScreenPublic Health Laboratories
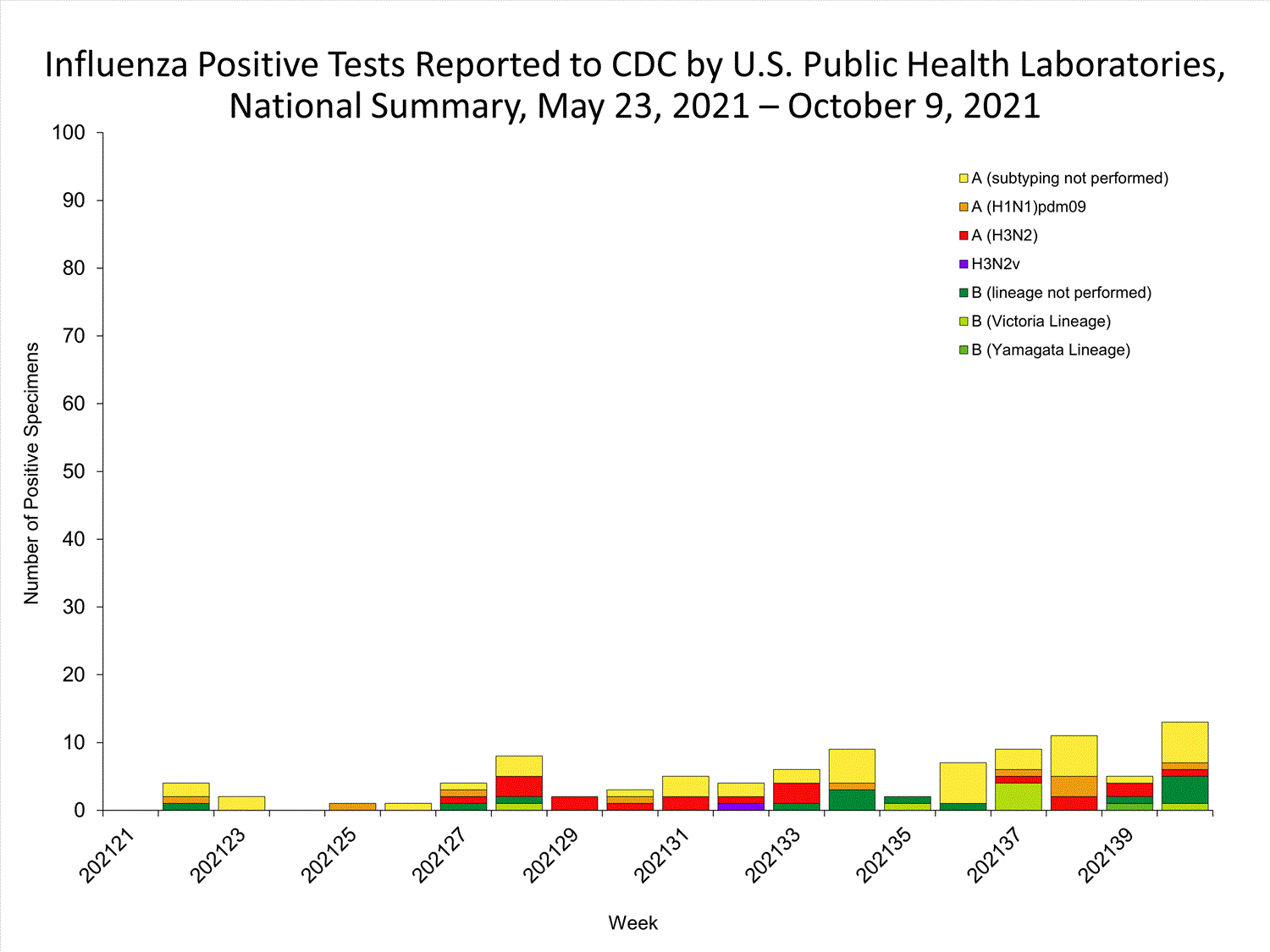
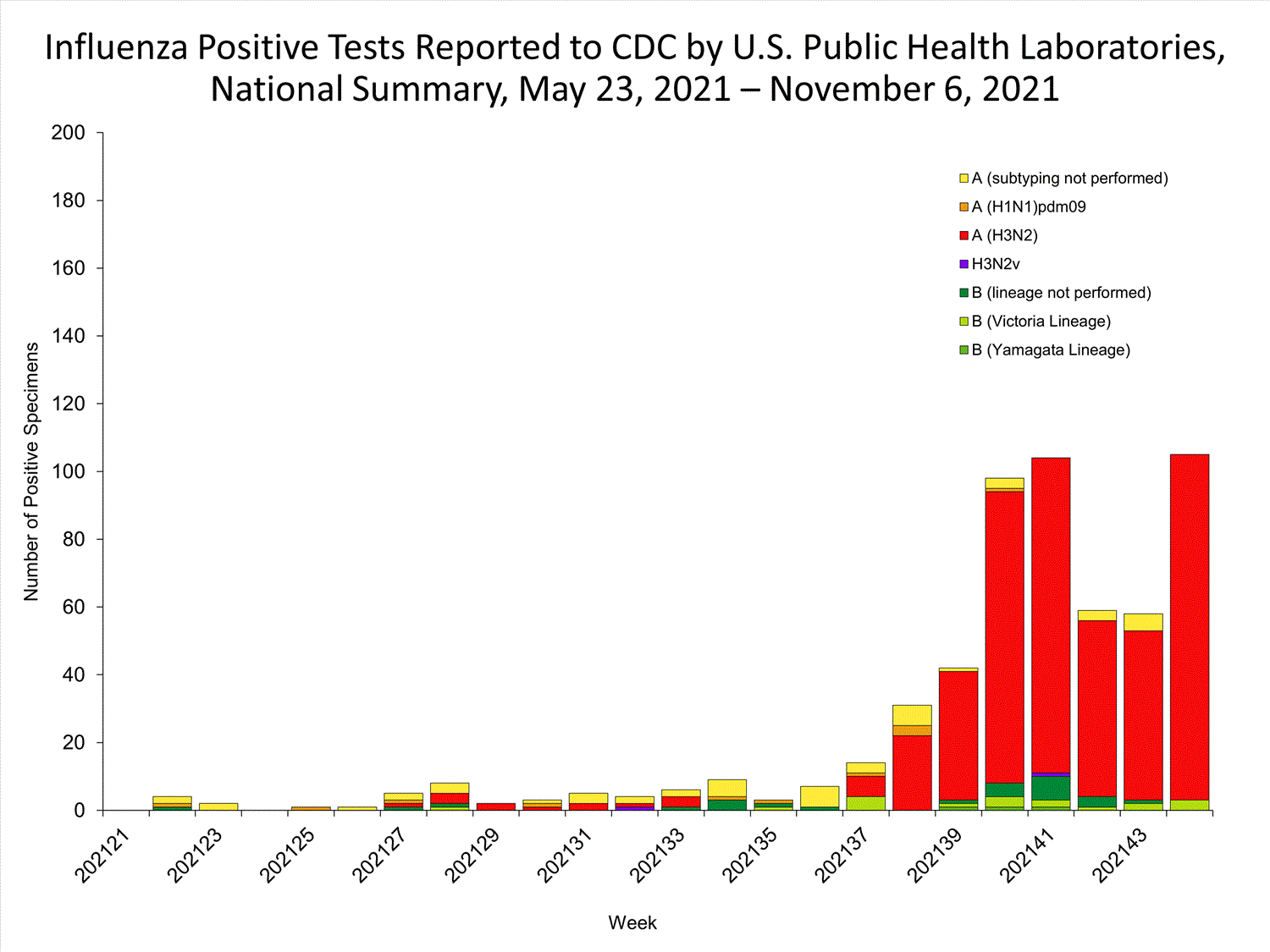
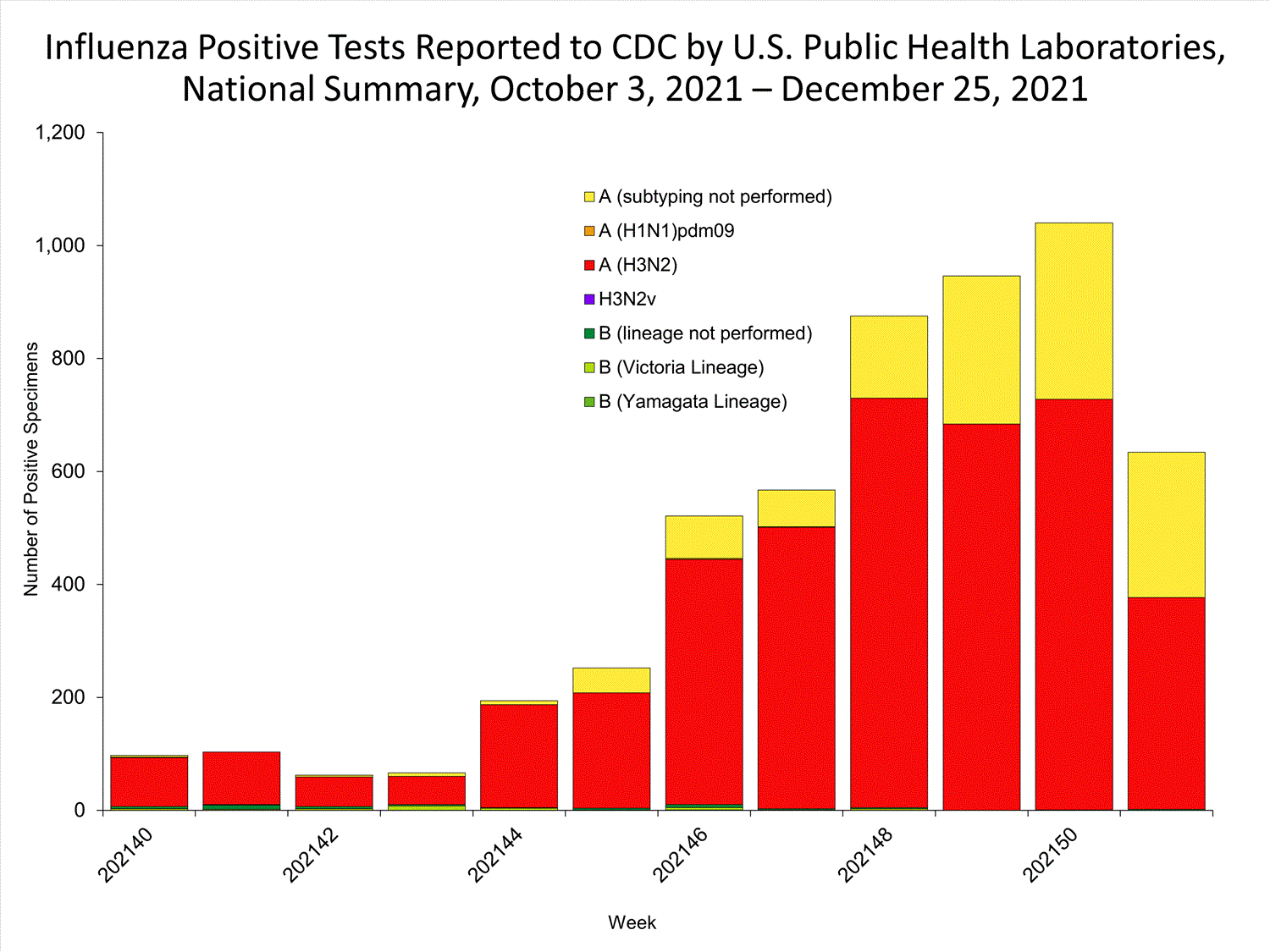
Data from public health laboratories are used to monitor the proportion of circulating viruses that belong to each influenza subtype/lineage.
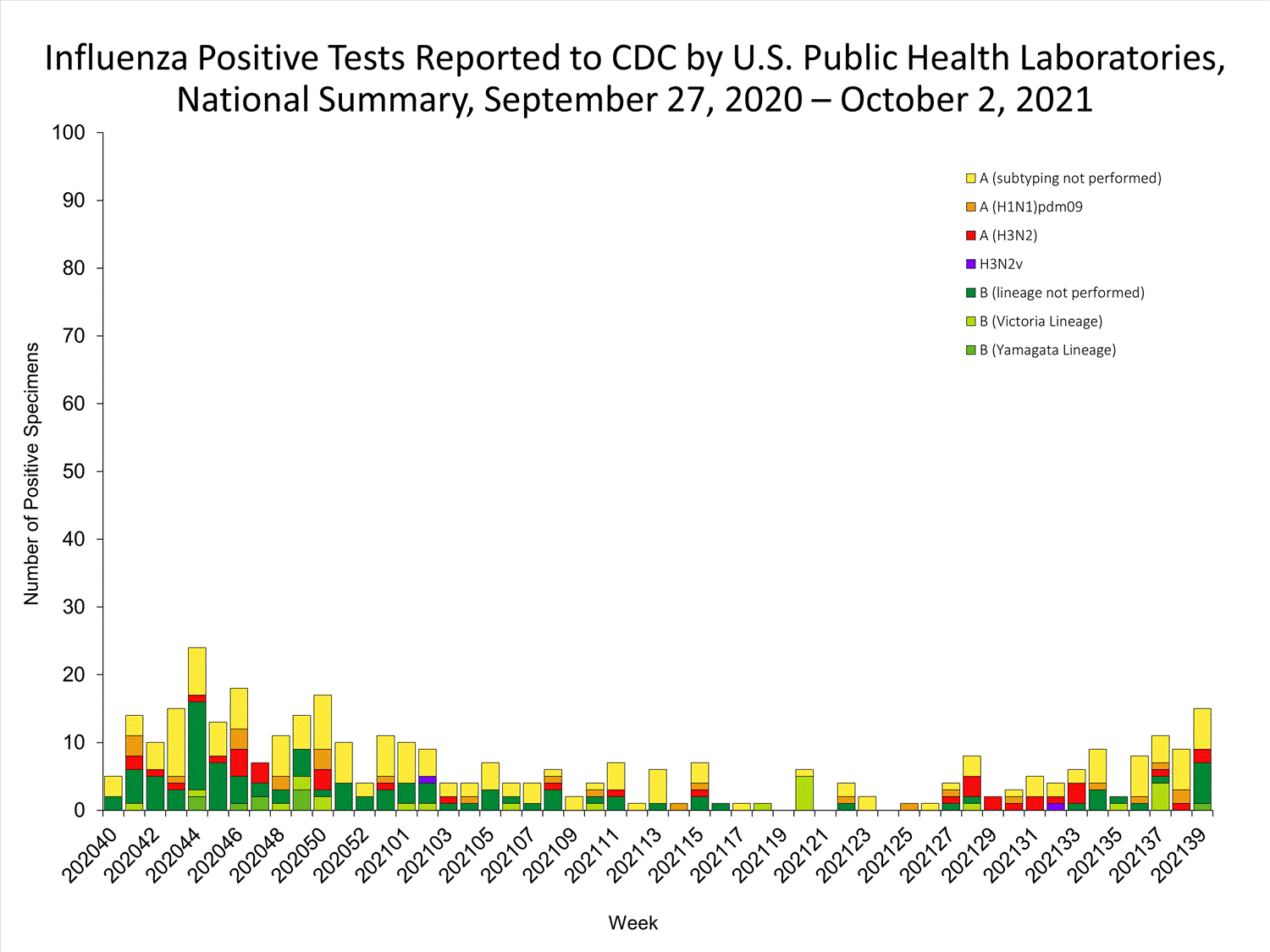
View Chart Data | View Full Screen
Additional information about medically attended visits for ILI for current and past seasons:
Surveillance Methods | FluView Interactive: National, Regional, and State Data or ILI Activity Map
Outpatient Illness Surveillance
Please note, the U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet) monitors outpatient visits for influenza-like illness (ILI), not laboratory-confirmed influenza, and will capture visits due to other respiratory pathogens, such as SARS-CoV-2, that present with similar symptoms. In addition, health care-seeking behaviors have changed dramatically during the COVID-19 pandemic. Many people are accessing the health care system in alternative settings, which may or may not be captured as a part of ILINet. Therefore, ILI data, including ILI activity levels, should be interpreted with caution. It is particularly important at this time to evaluate syndromic surveillance data, including that from ILINet, in the context of other sources of surveillance data to obtain a complete and accurate picture of influenza, COVID-19, and other respiratory virus activity. CDC is tracking the COVID-19 pandemic in a weekly publication called COVID Data Tracker Weekly Review. Information about other respiratory virus activity can be found on CDC’s National Respiratory and Enteric Virus Surveillance System (NREVSS) website.
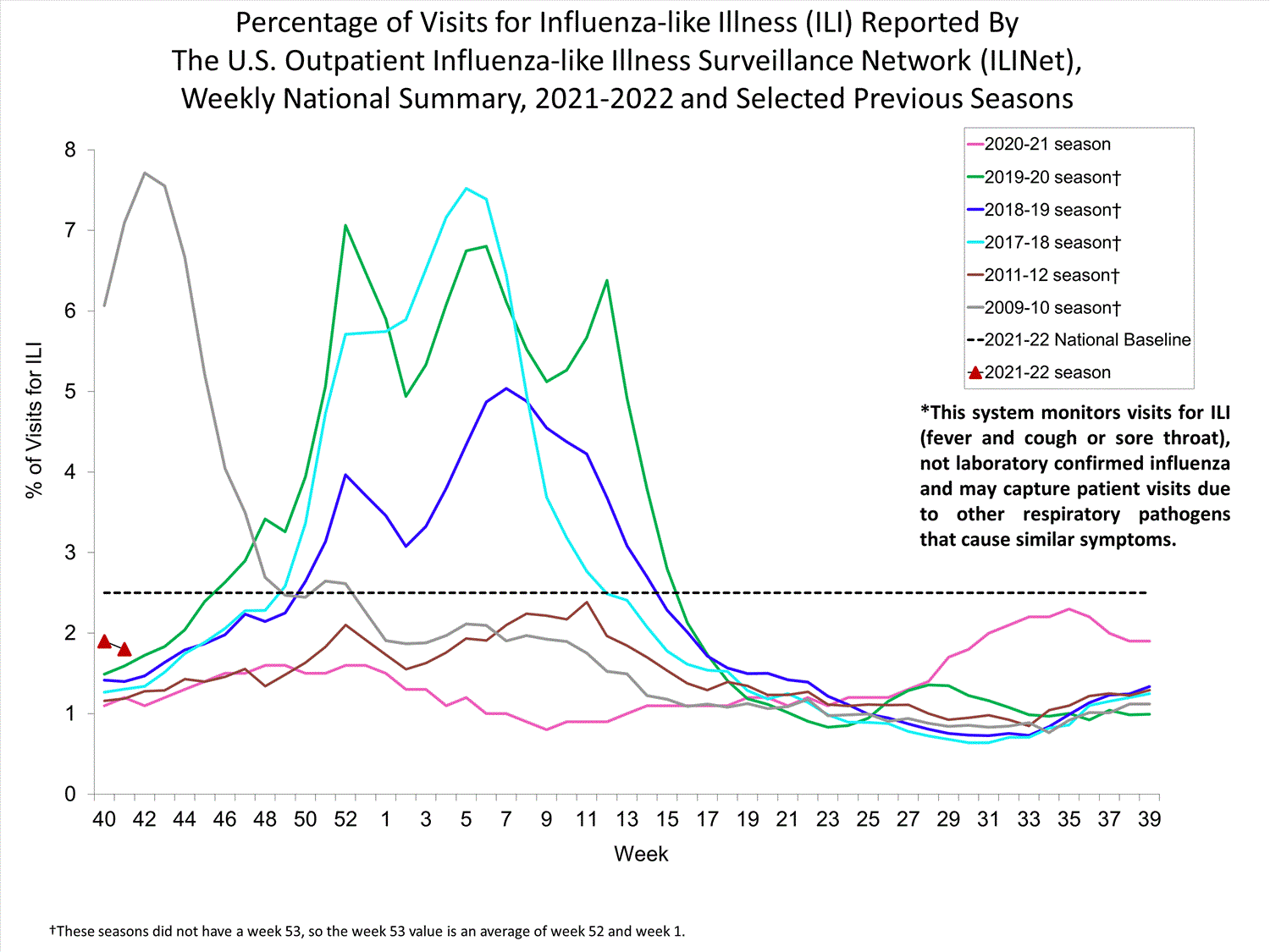
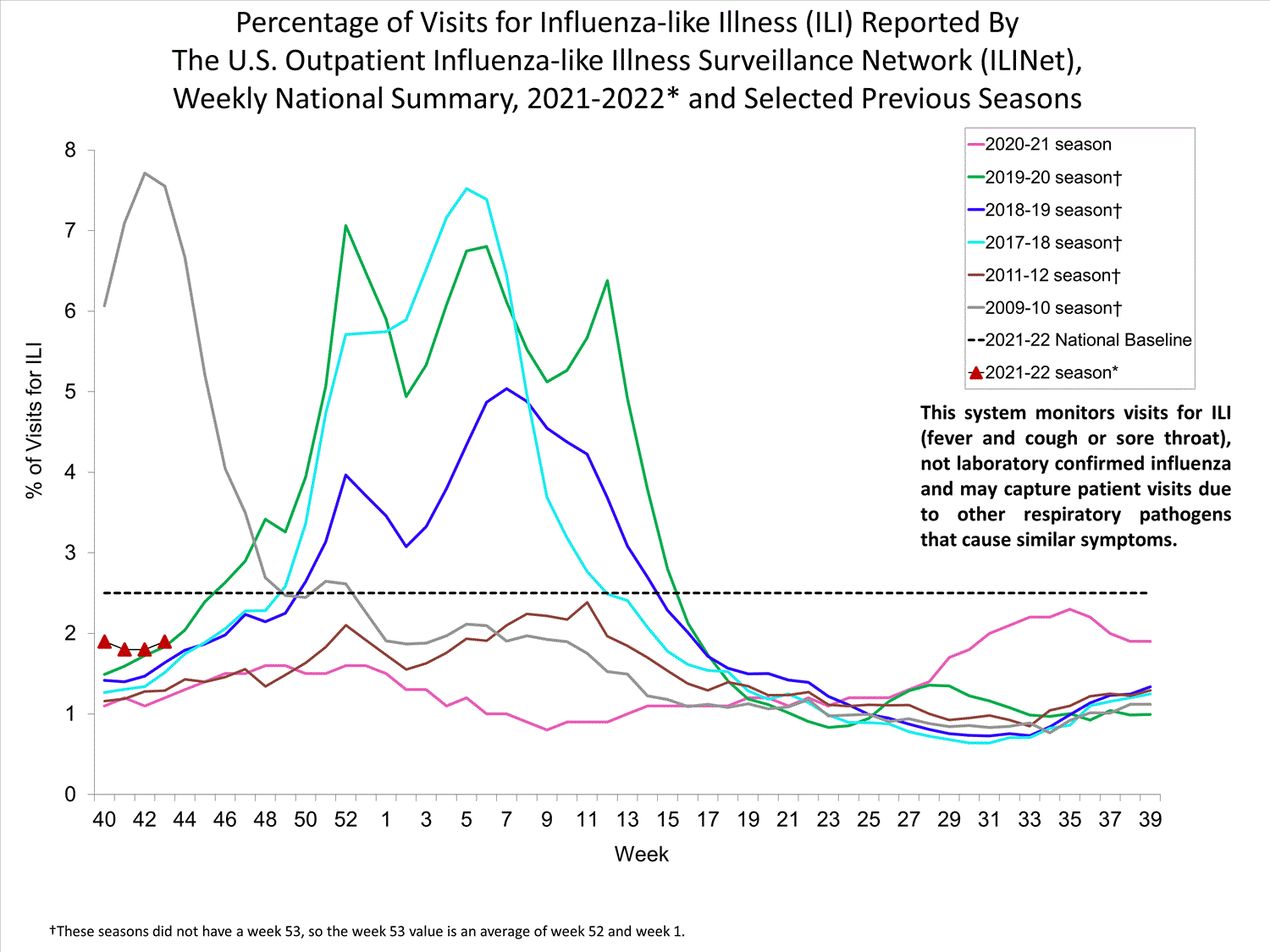
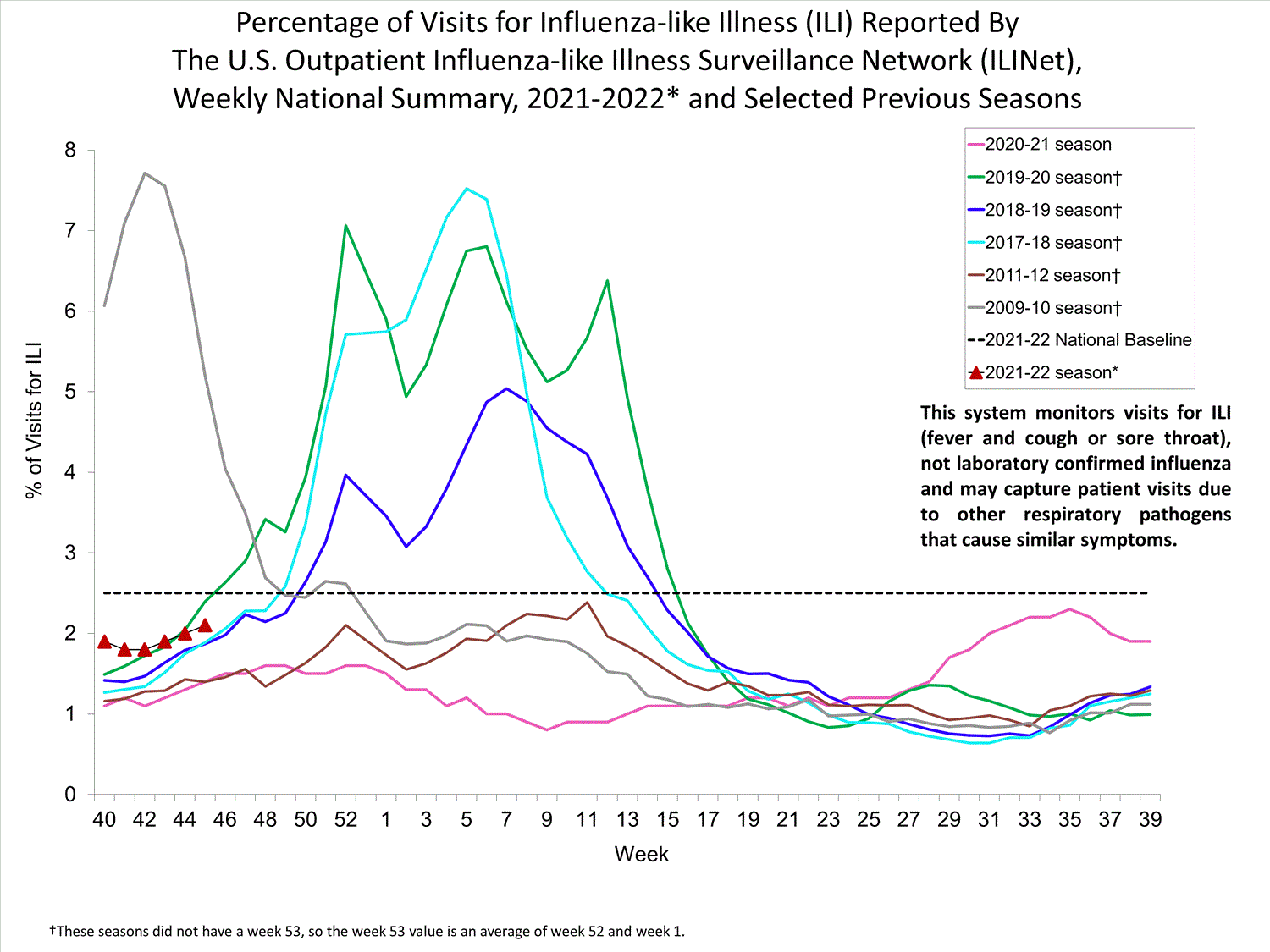
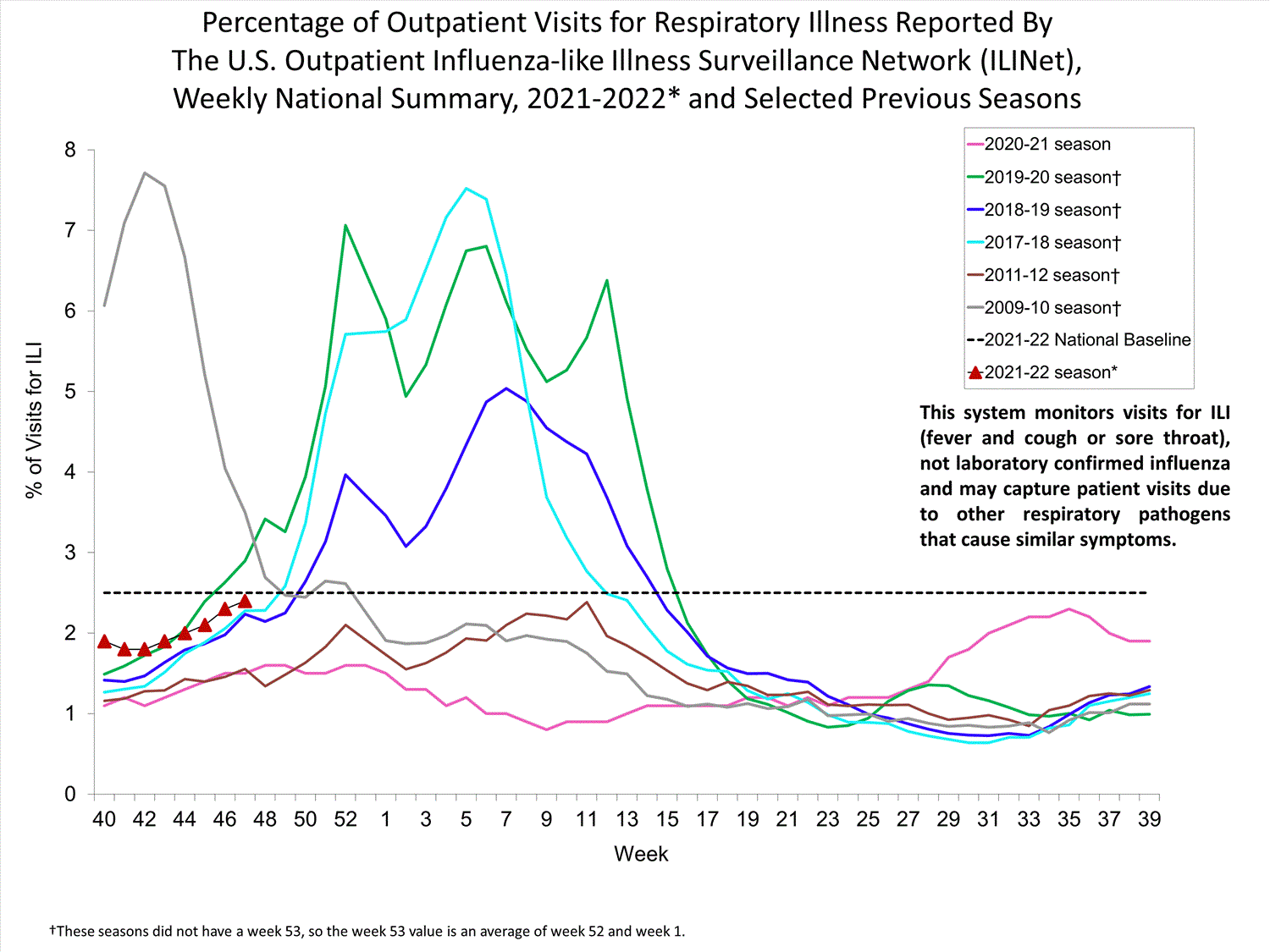
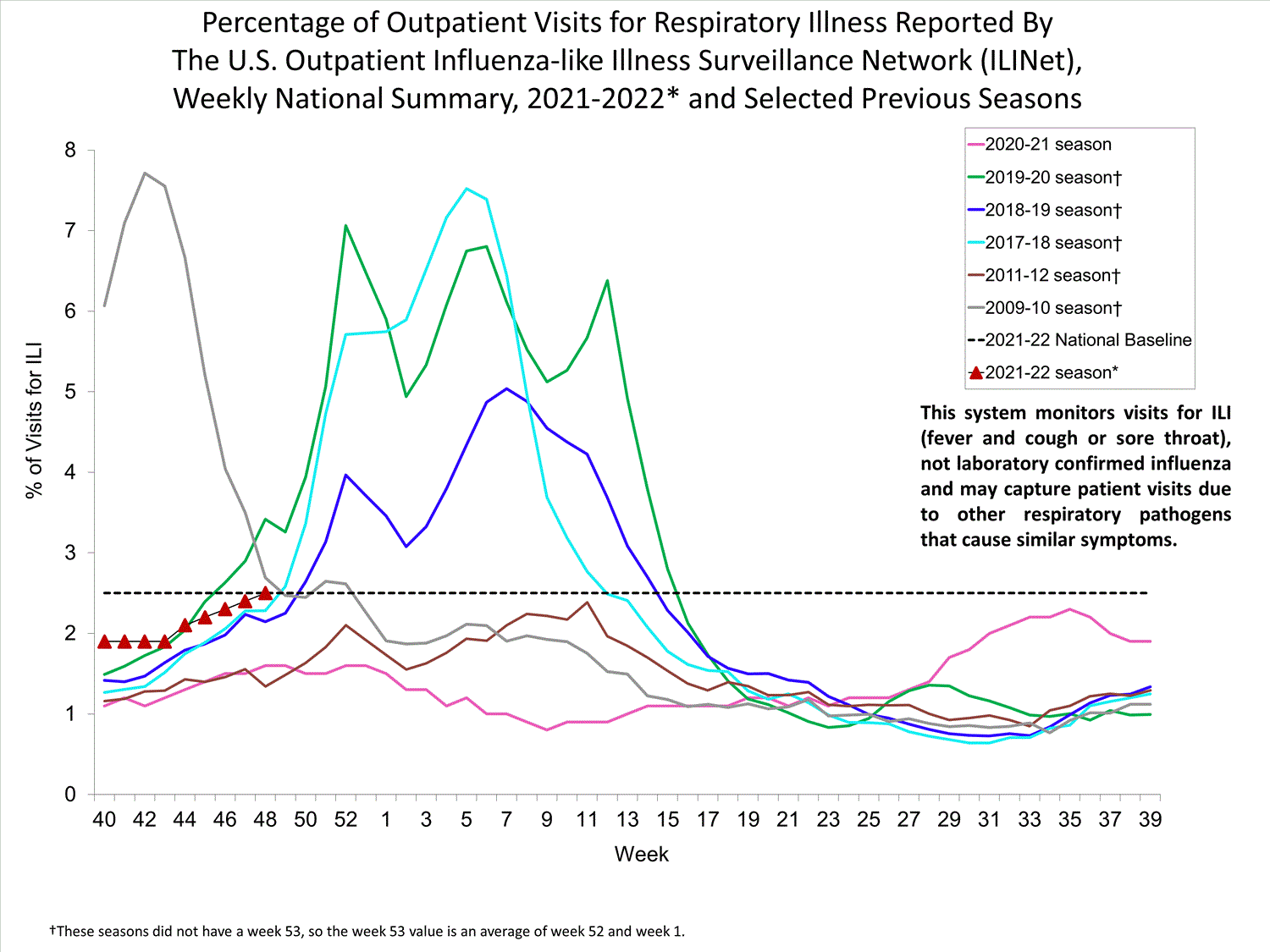
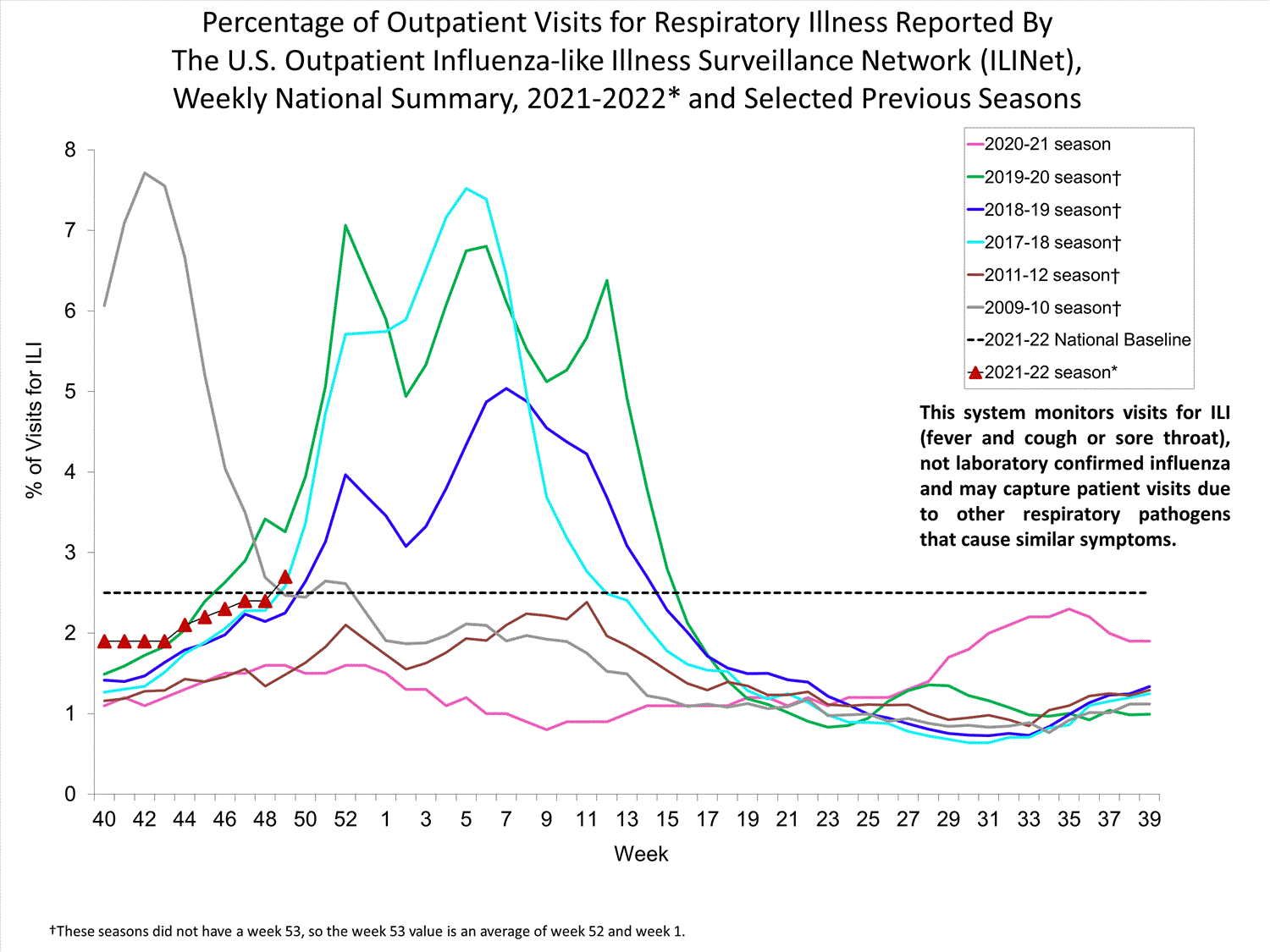
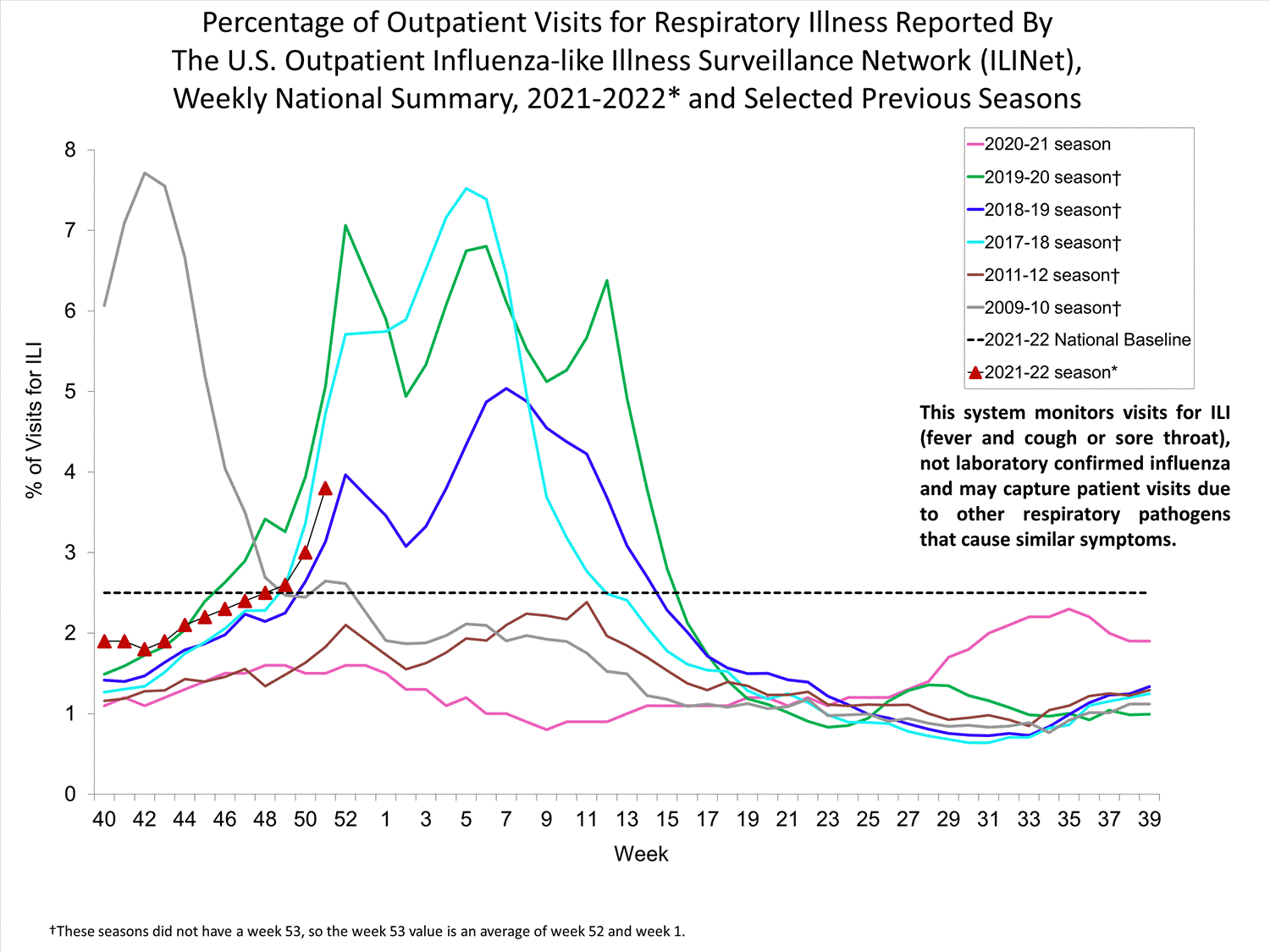
ILINet
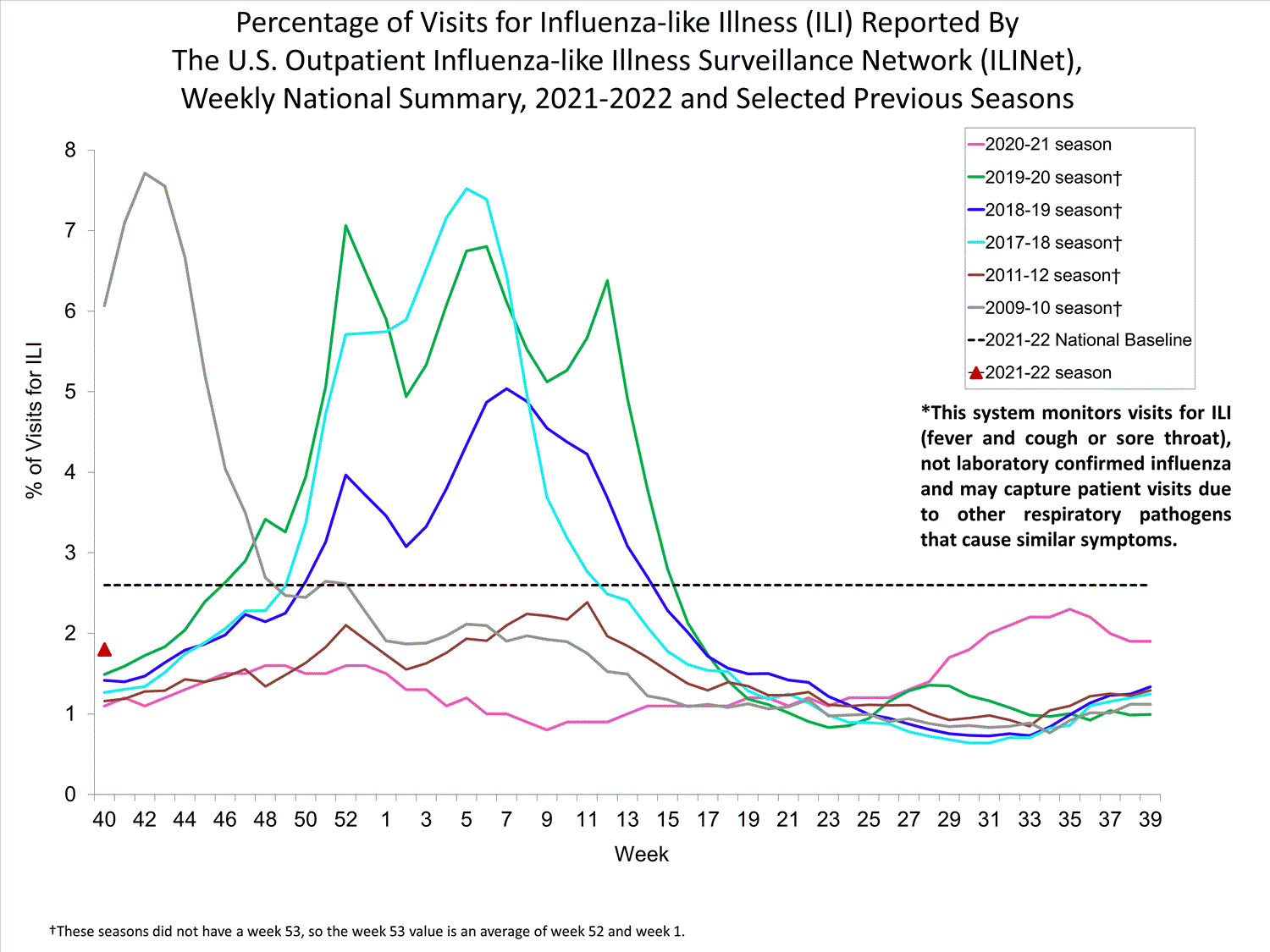
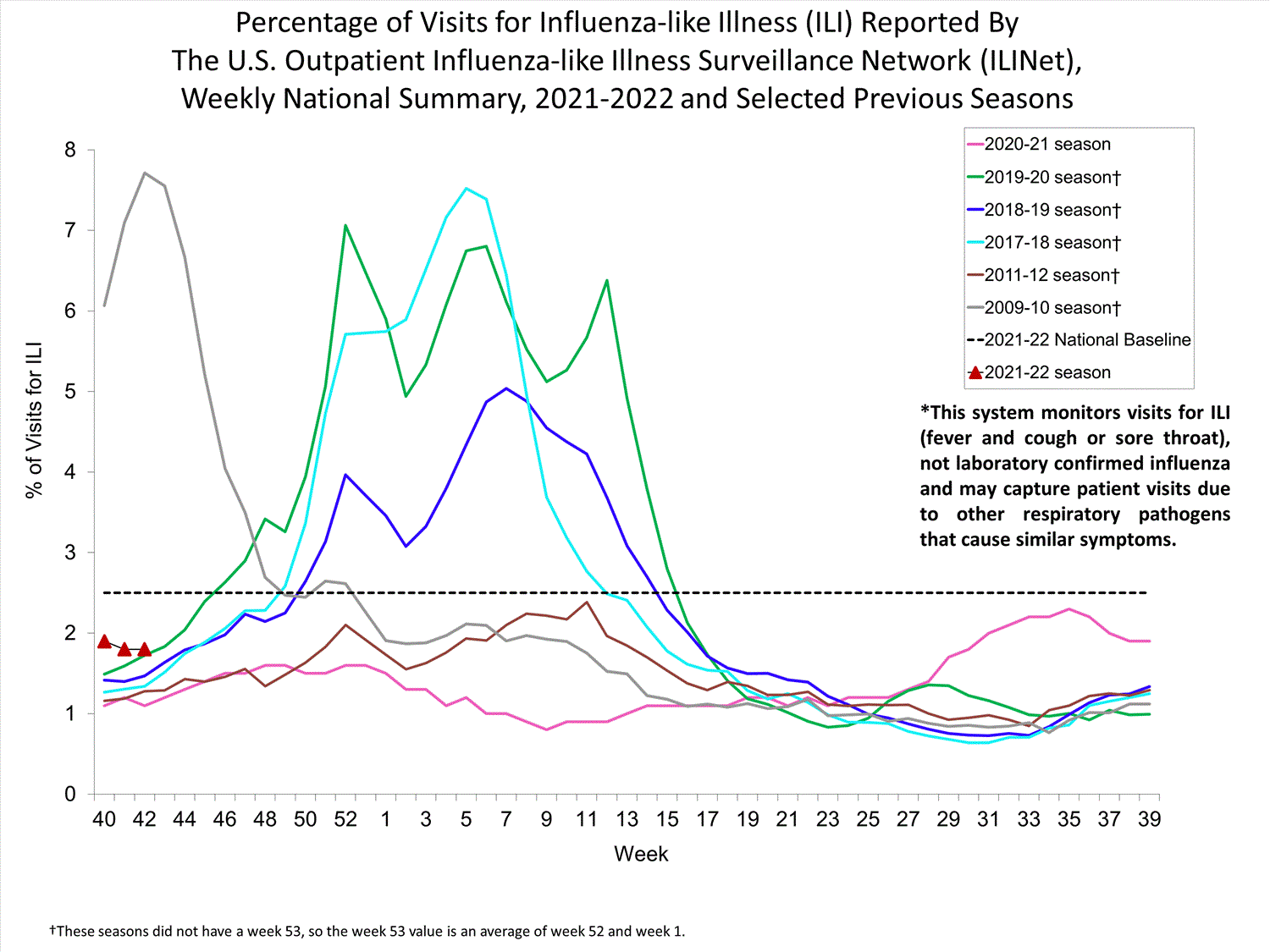
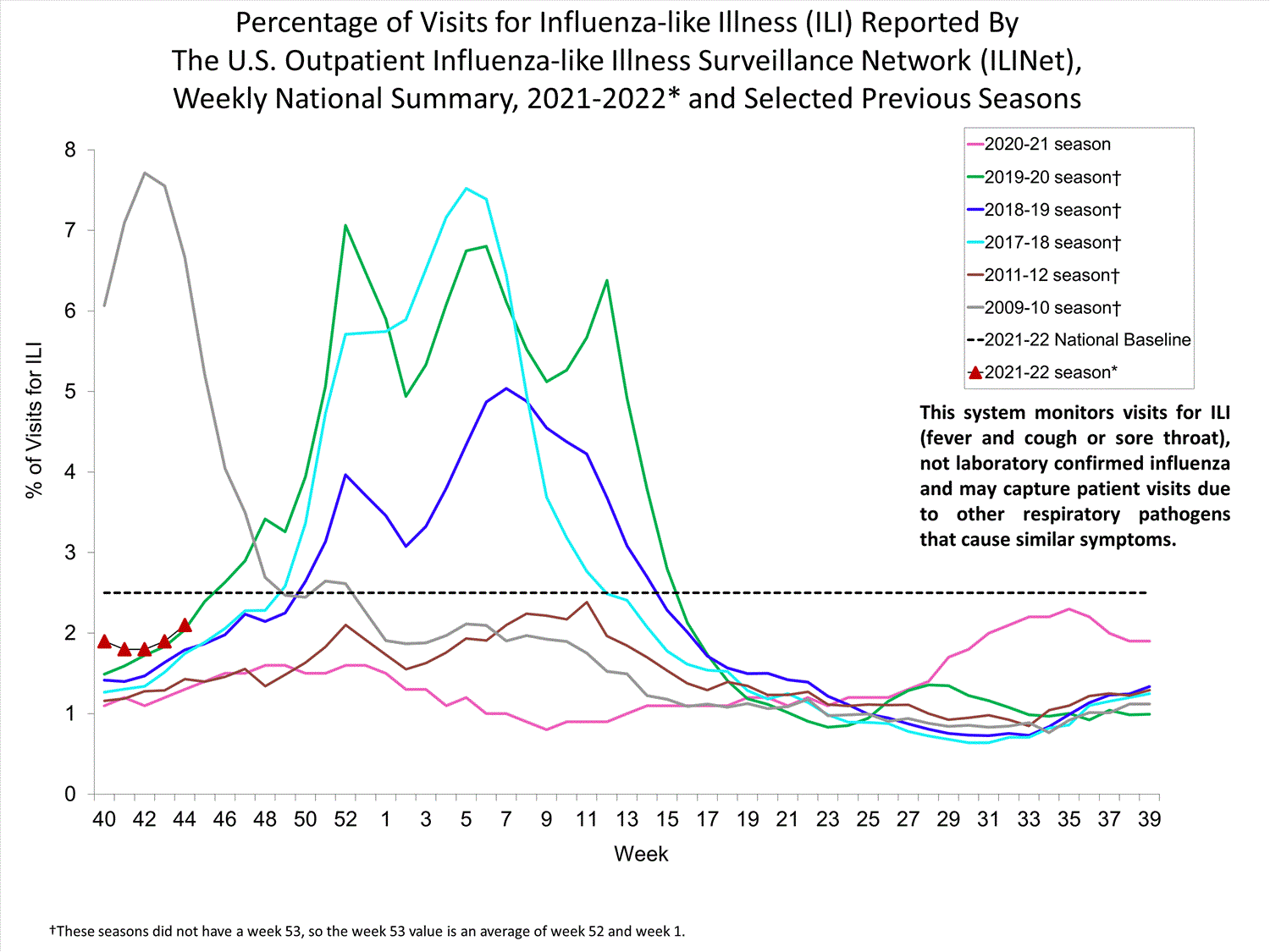
Nationwide during week 39, 1.9% of patient visits reported through ILINet were due to ILI. The percentage of patient visits for ILI remains below the baseline of 2.6% nationally. All ten regions are below their region-specific baselines.
Influenza virus circulation remains low; therefore, increases in ILI activity are likely due to increased circulation of other respiratory viruses.
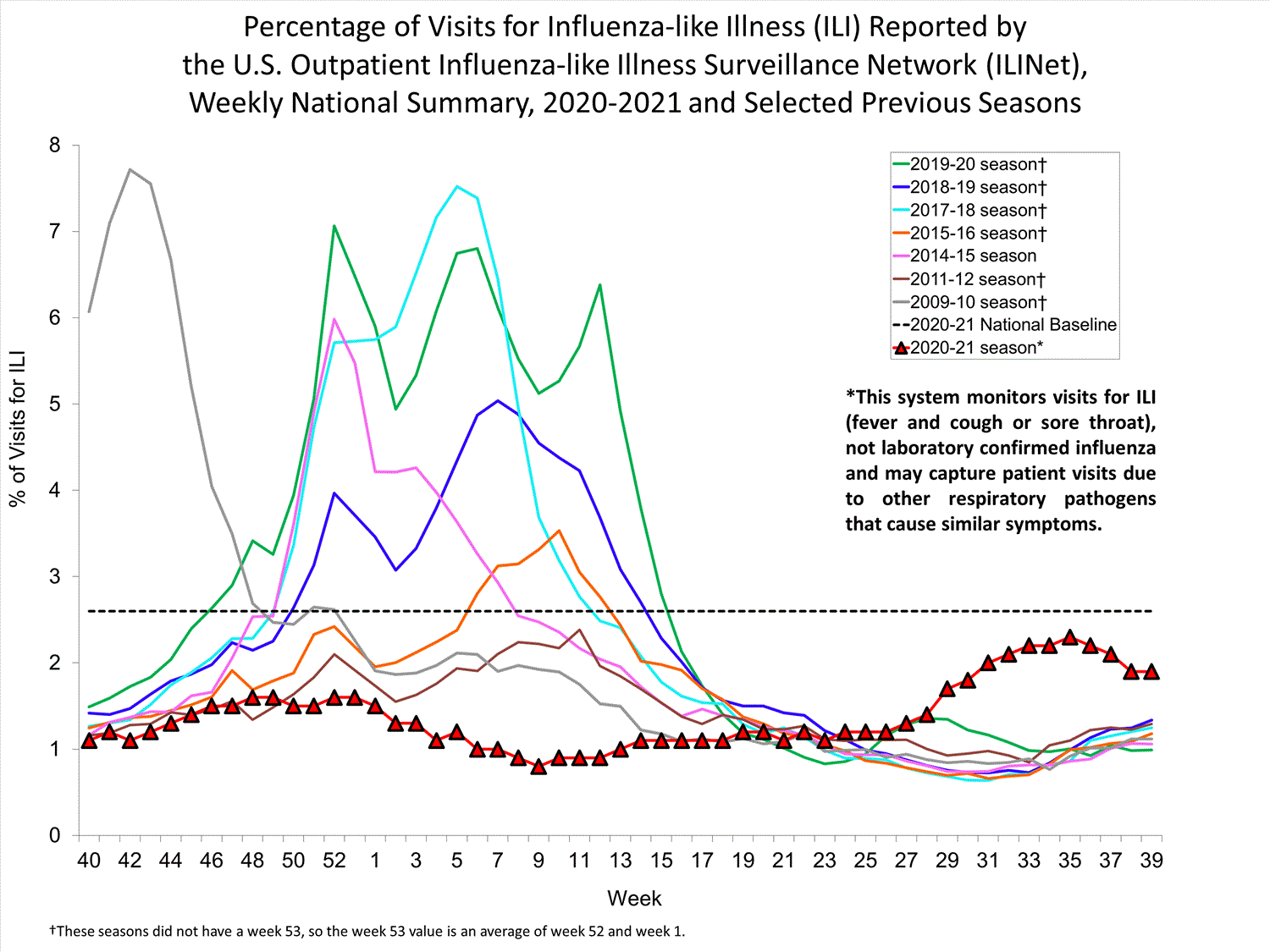
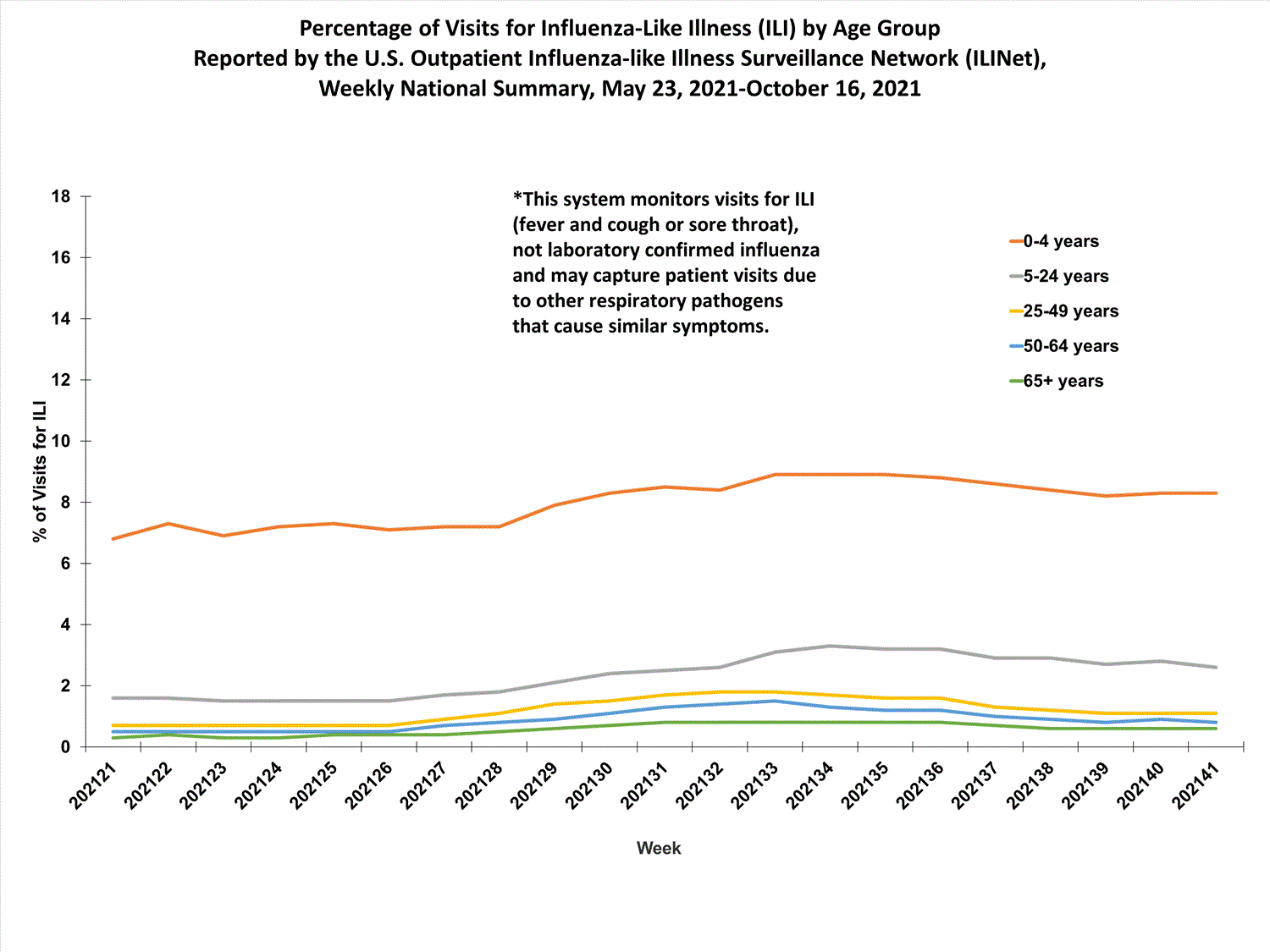
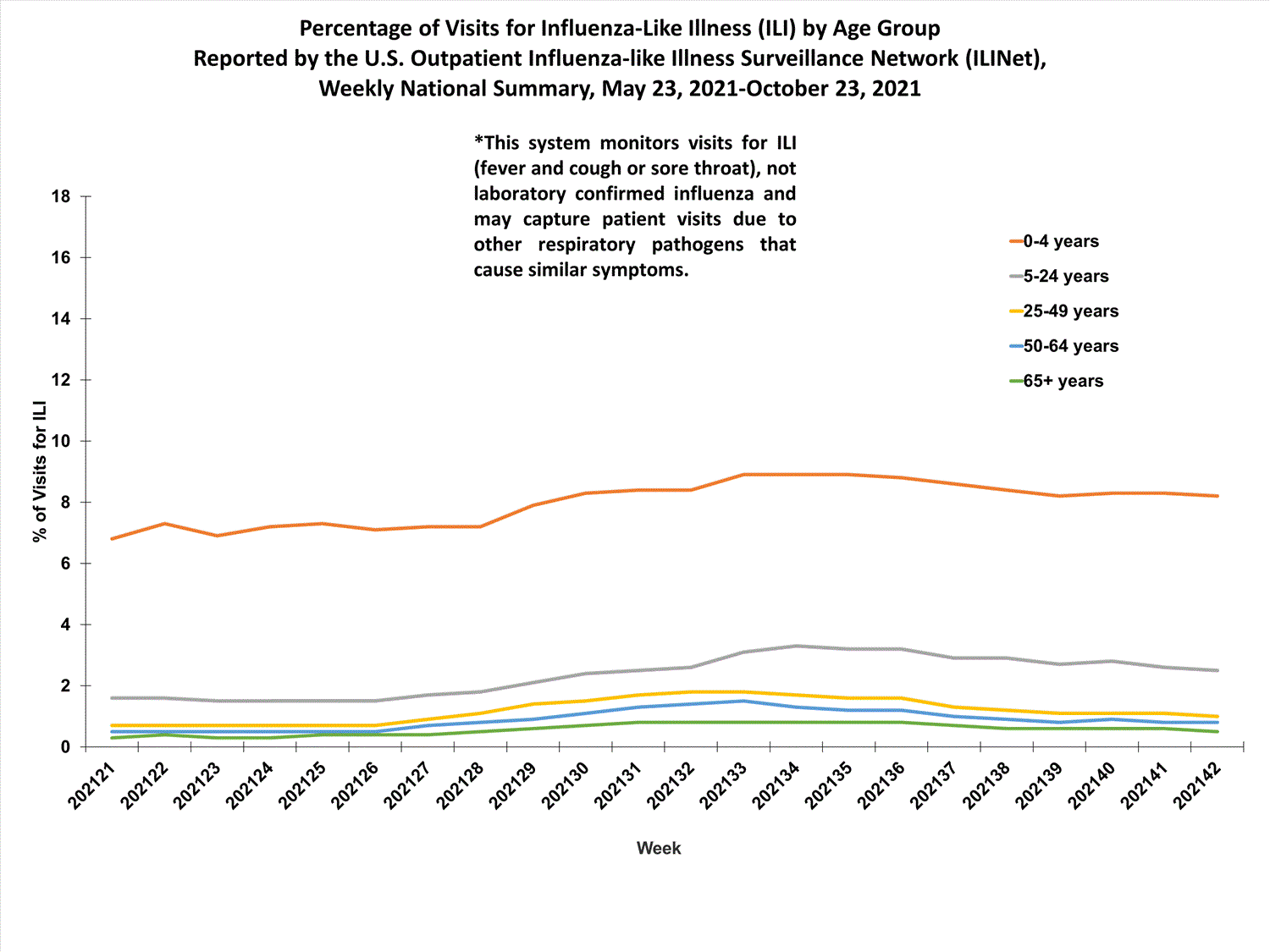
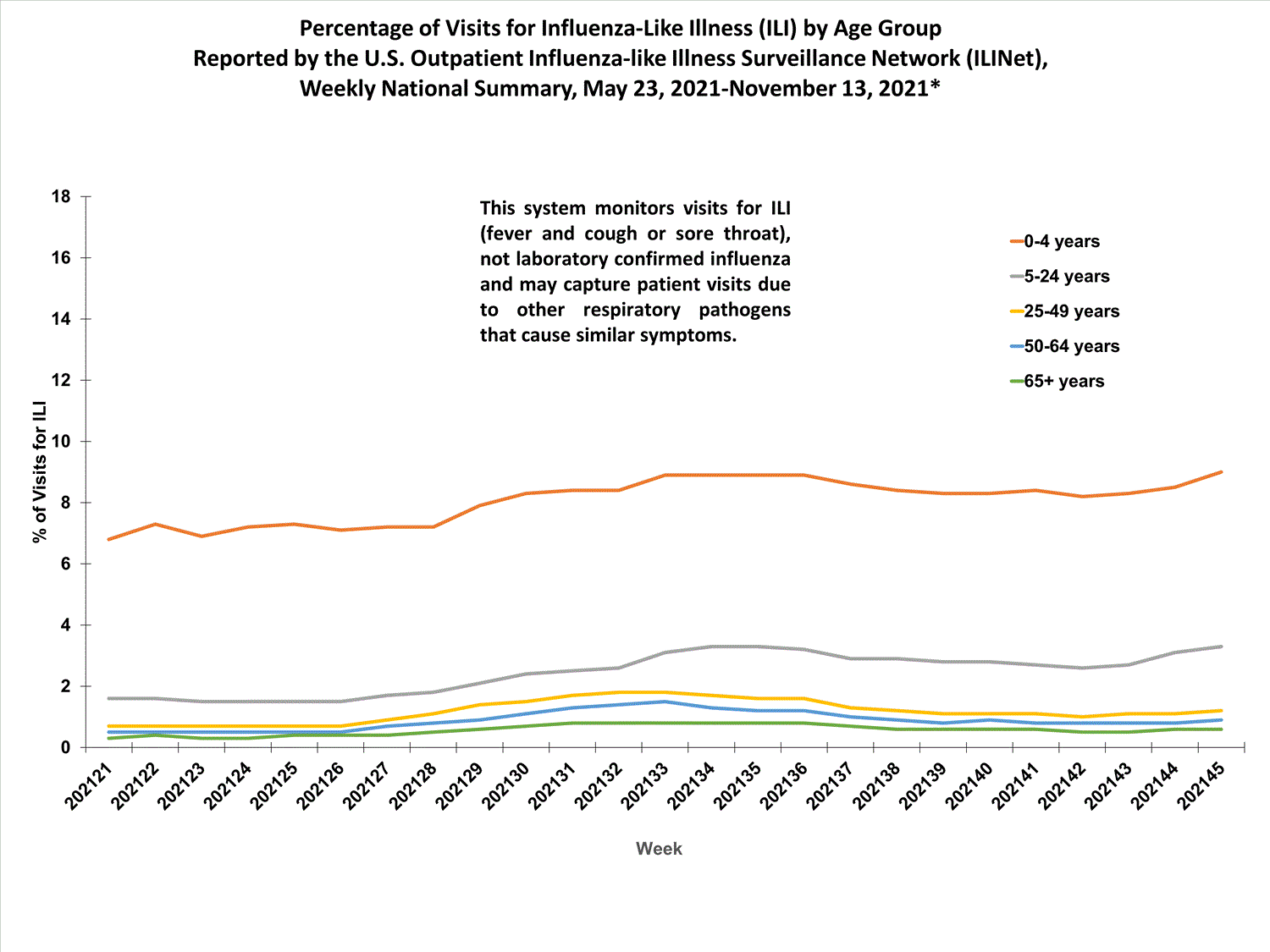
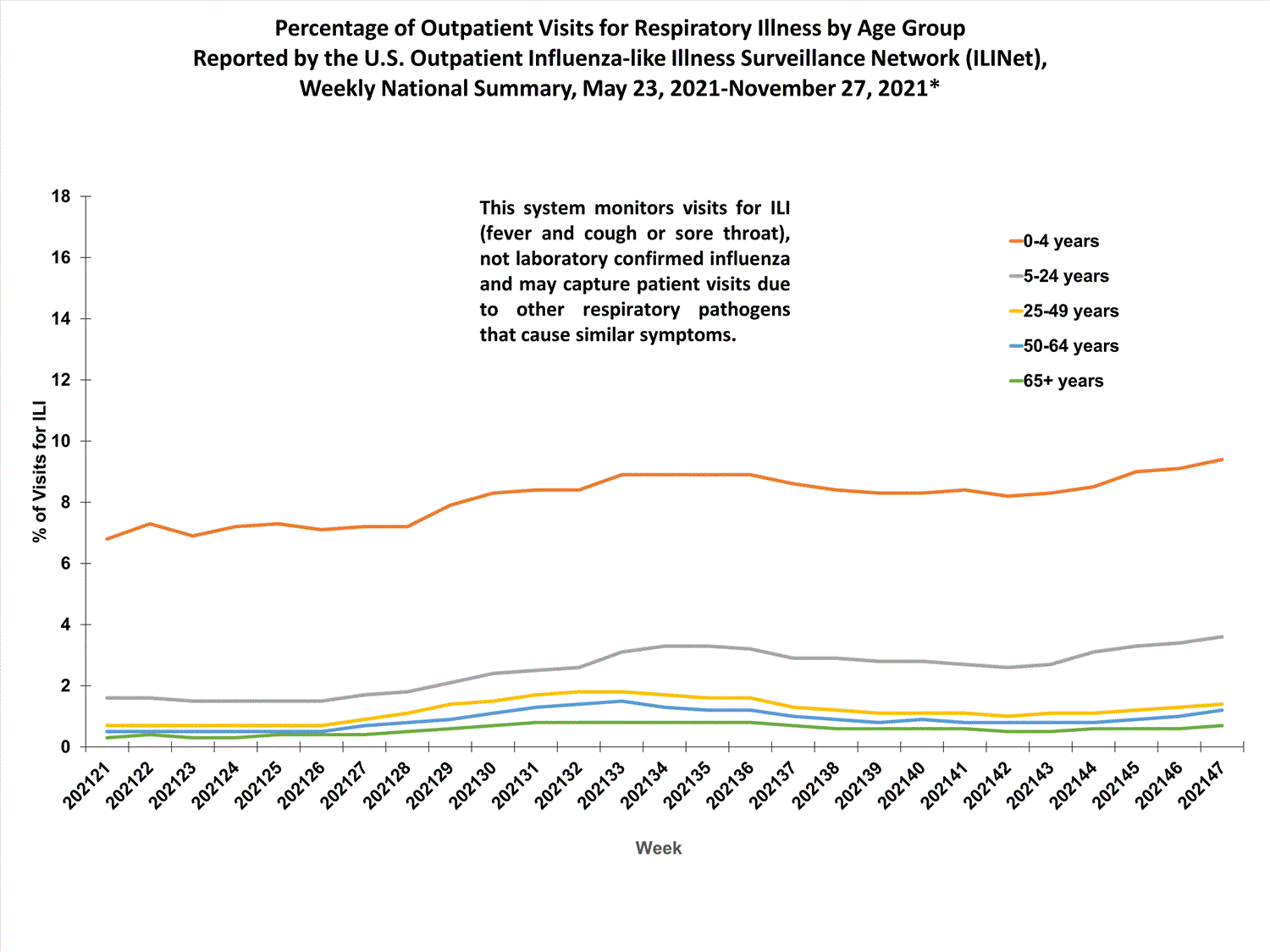
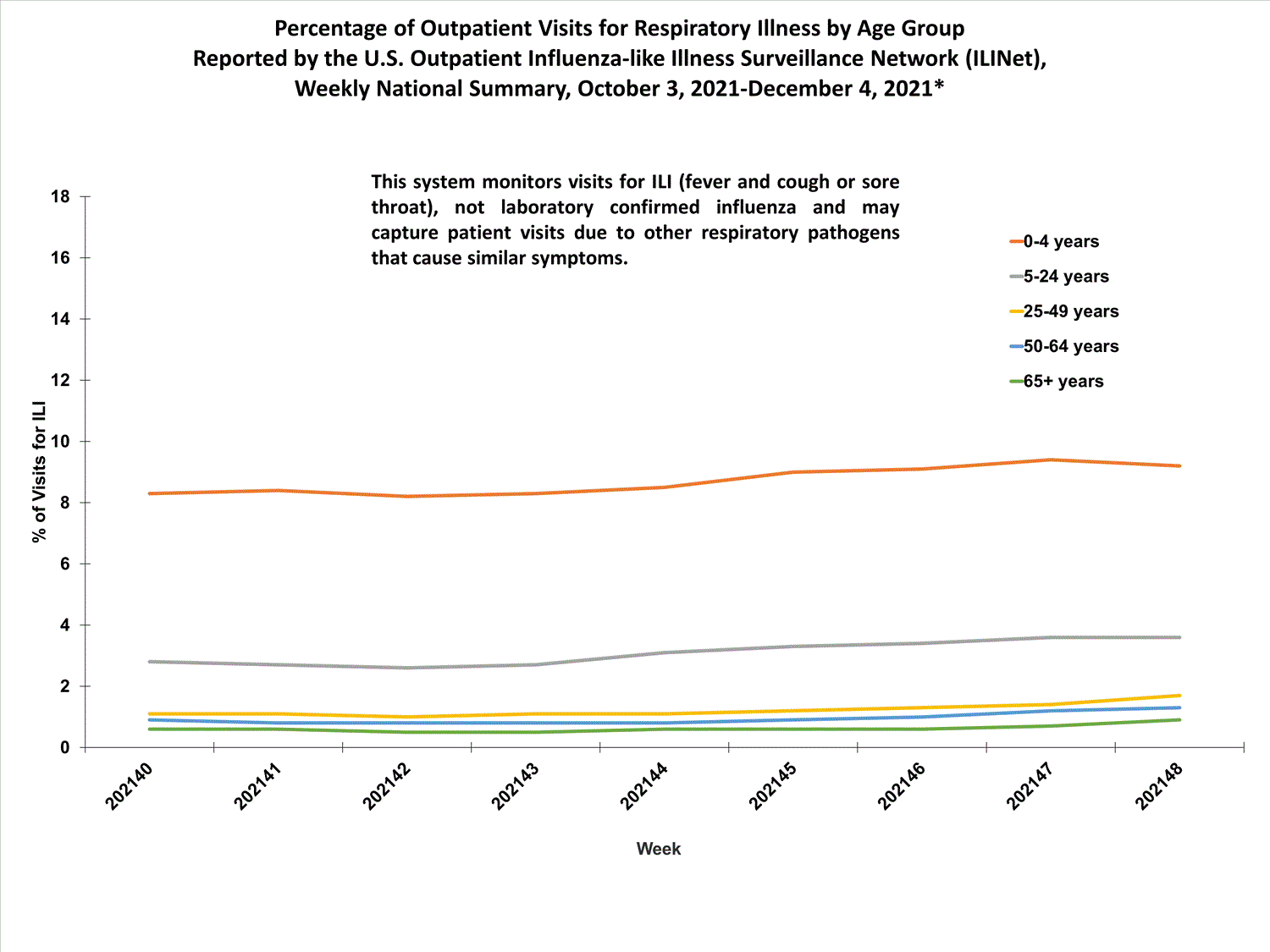
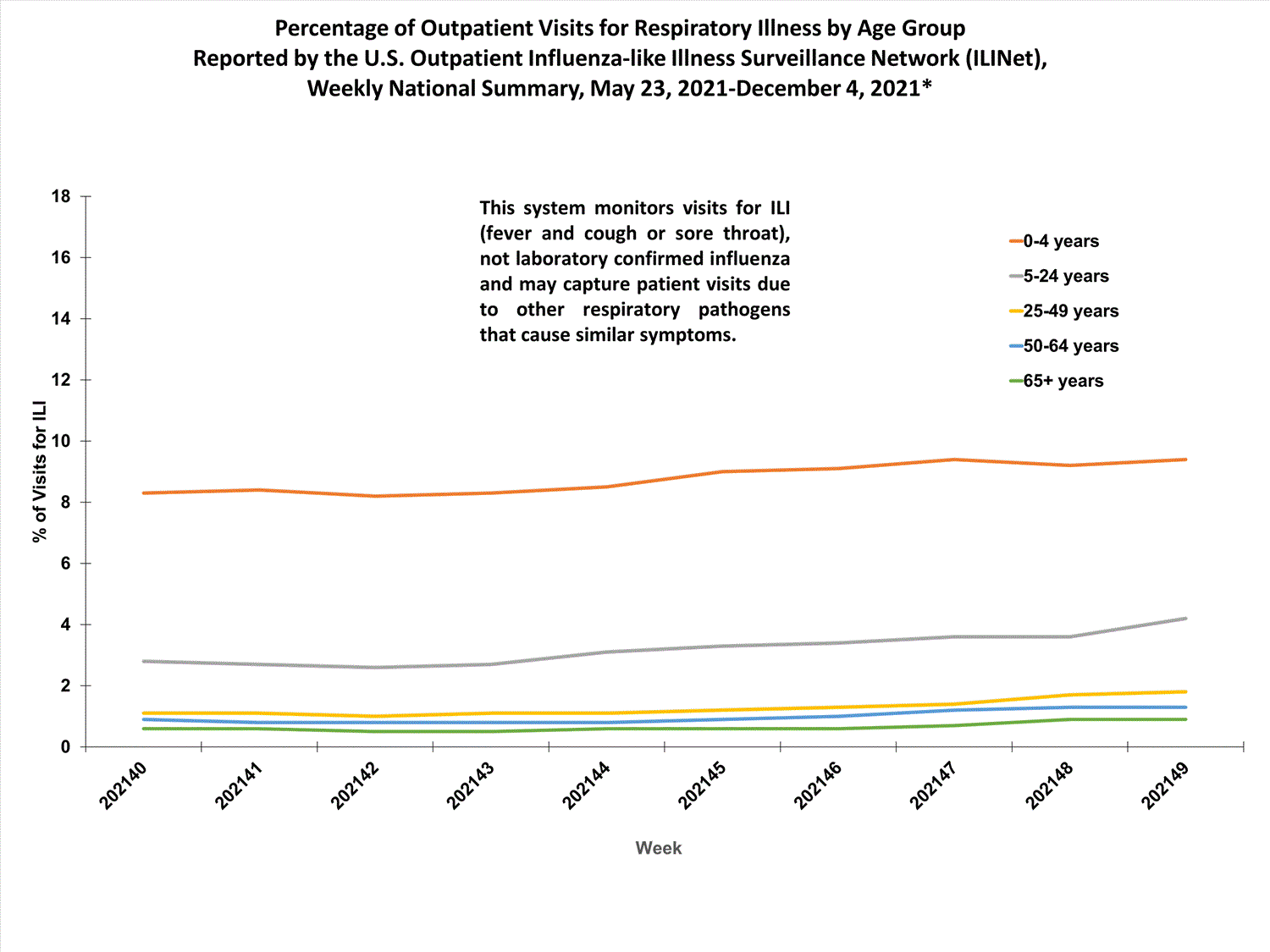
View Chart Data (current season only) | View Full ScreenILI Visits by Age Group
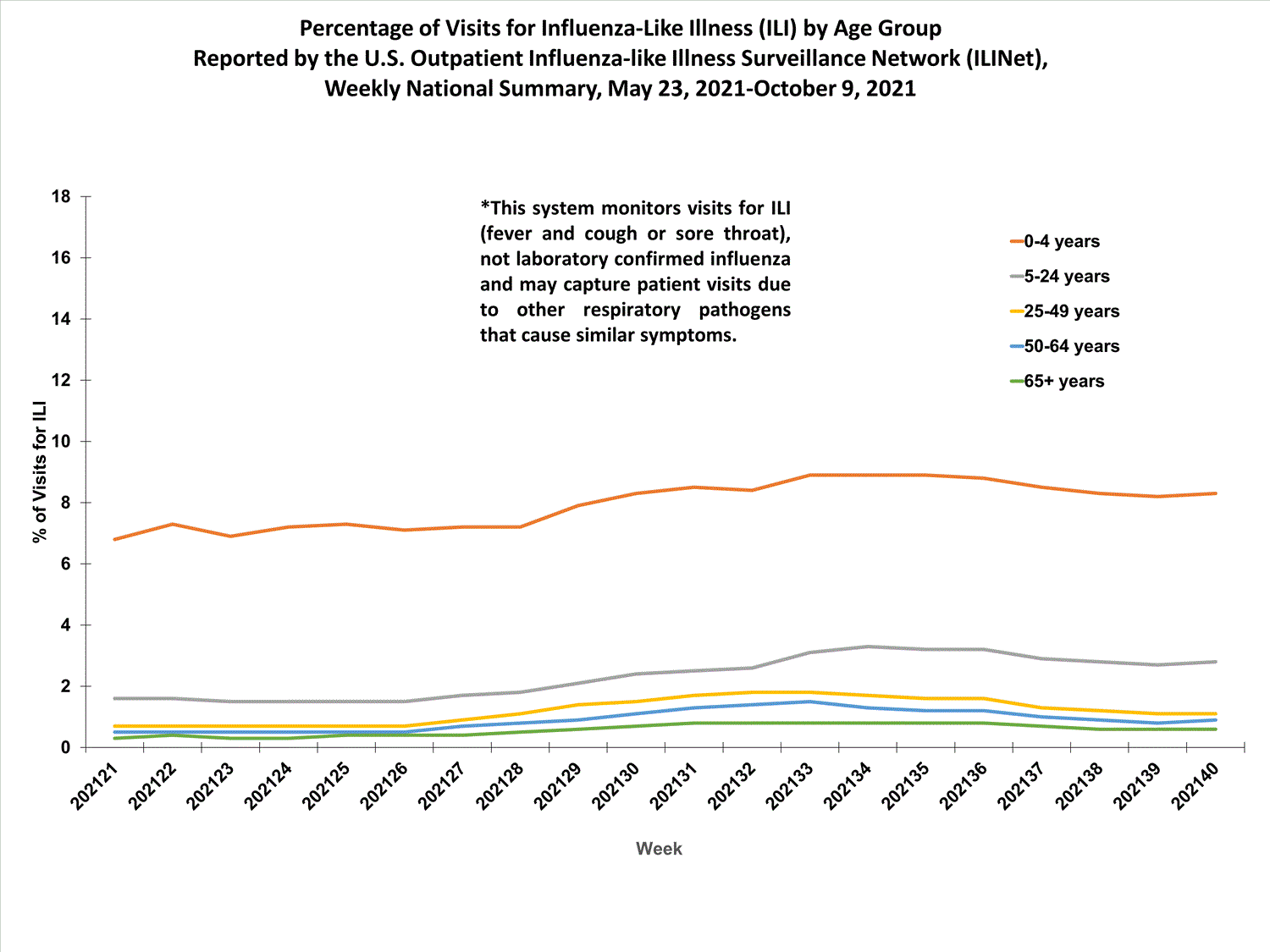
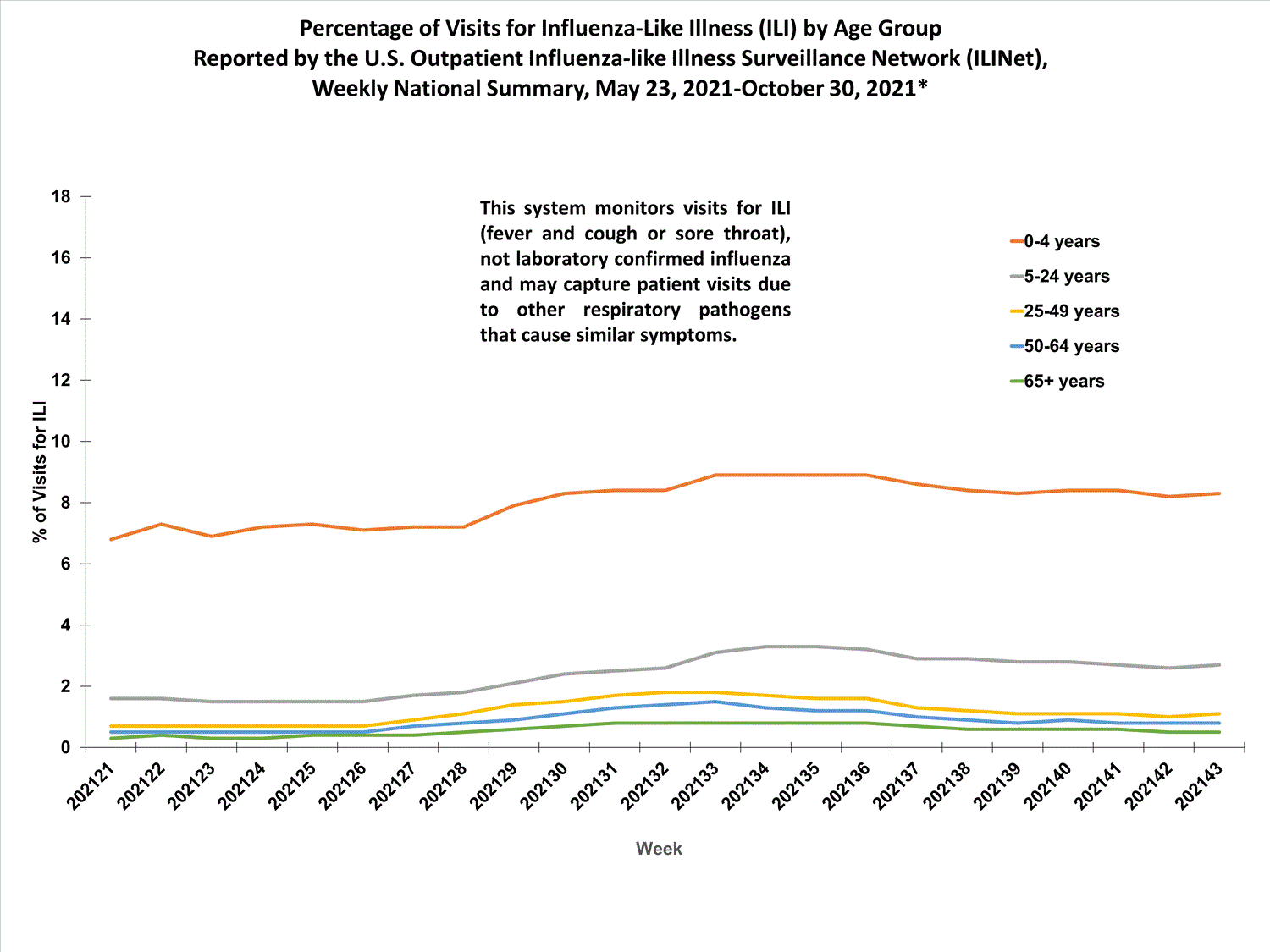
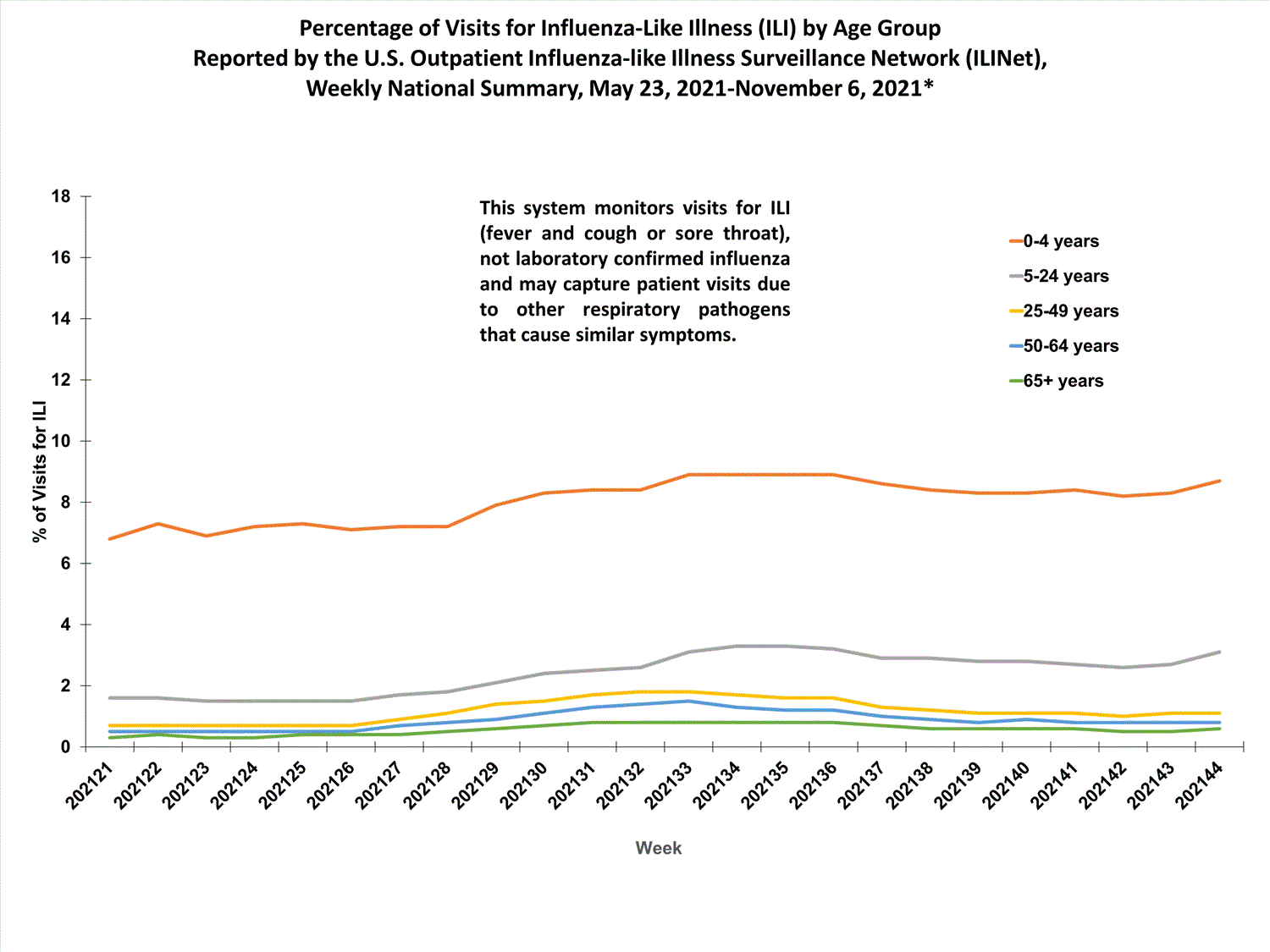
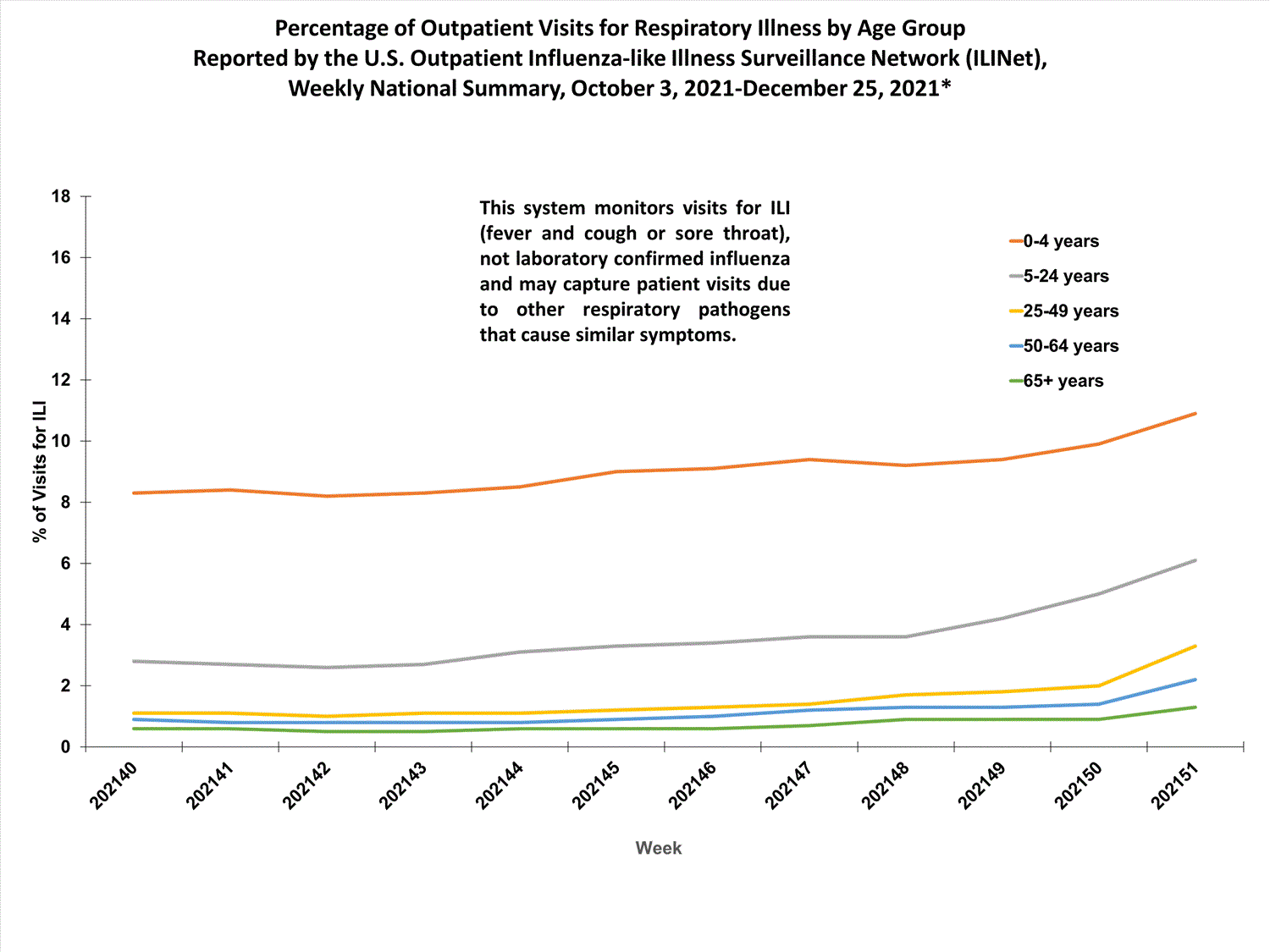
About 65% of ILINet participants provide both the number of patient visits for ILI and the total number of patient visits for the week broken out by age group. Data from this subset of providers are used to calculate the percentages of patient visits for ILI by age group.
The percentages of visits for ILI reported in ILINet are decreasing among all age groups (0-4 years, 5-24 years, 25-49 years, 50-64 years, and 65+ years).
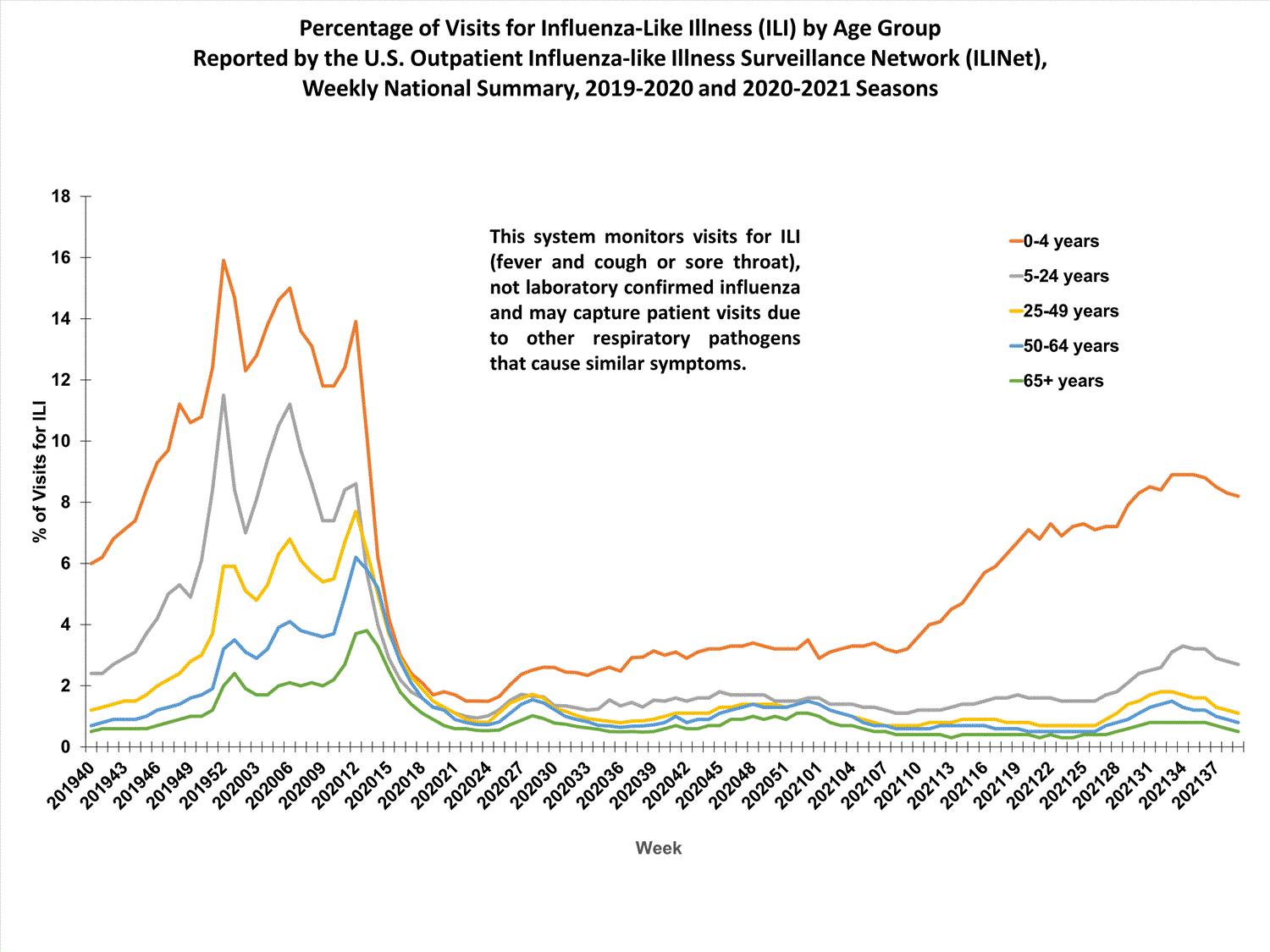
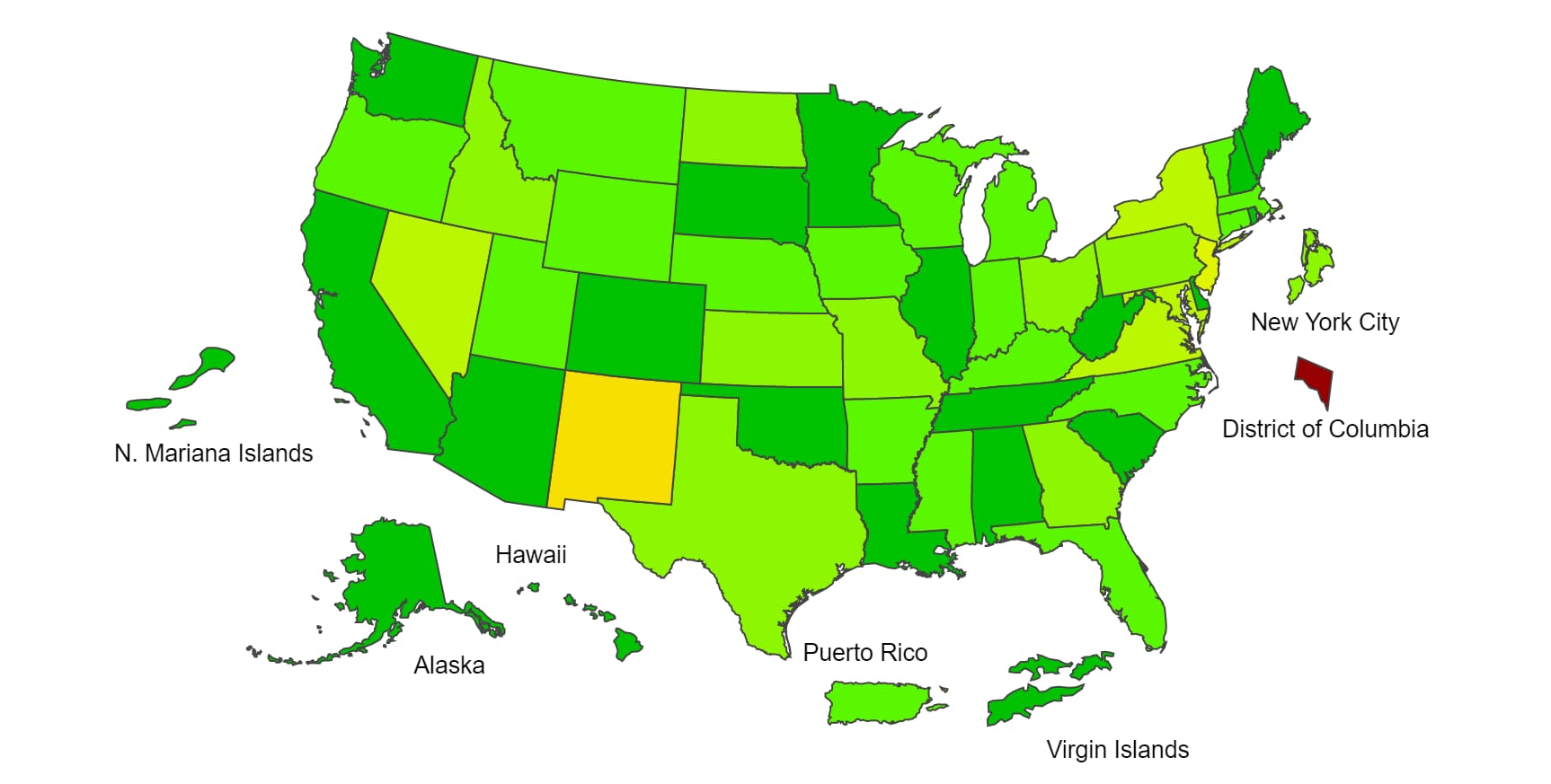
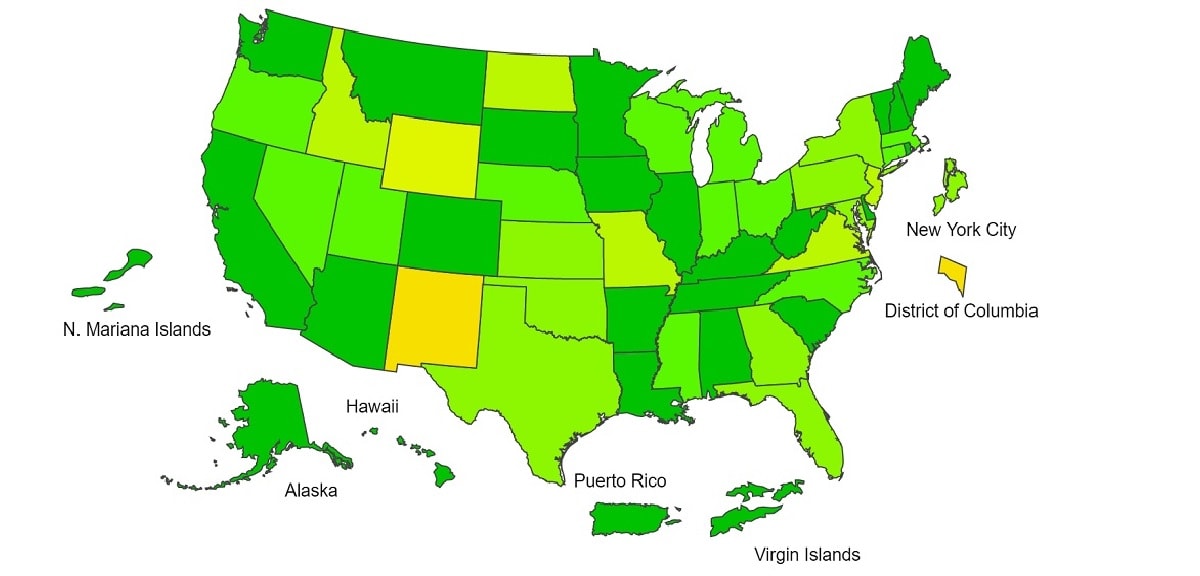
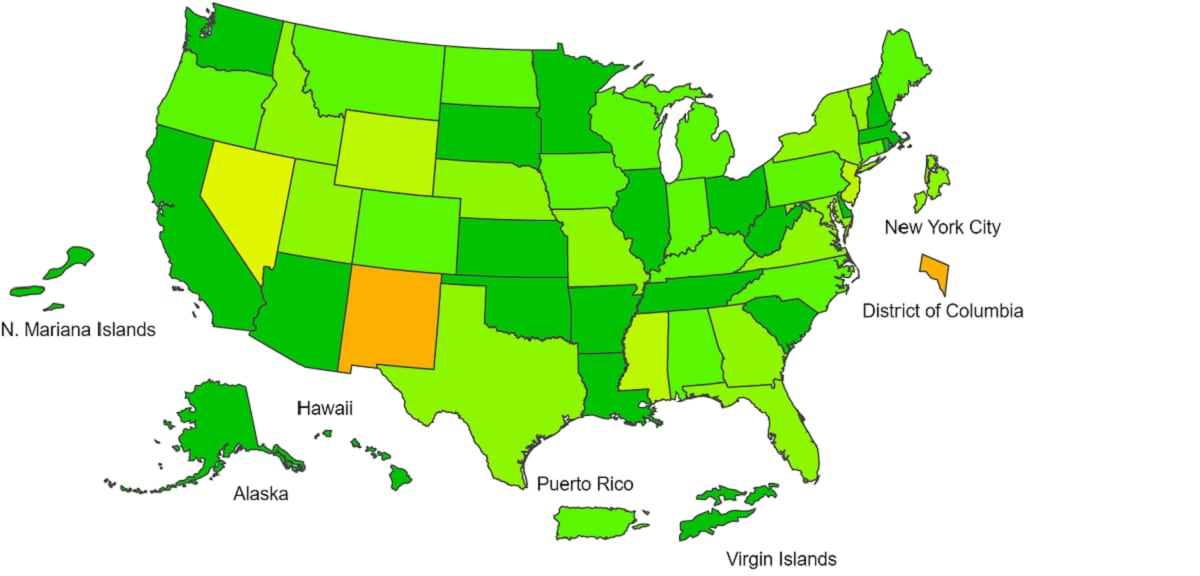
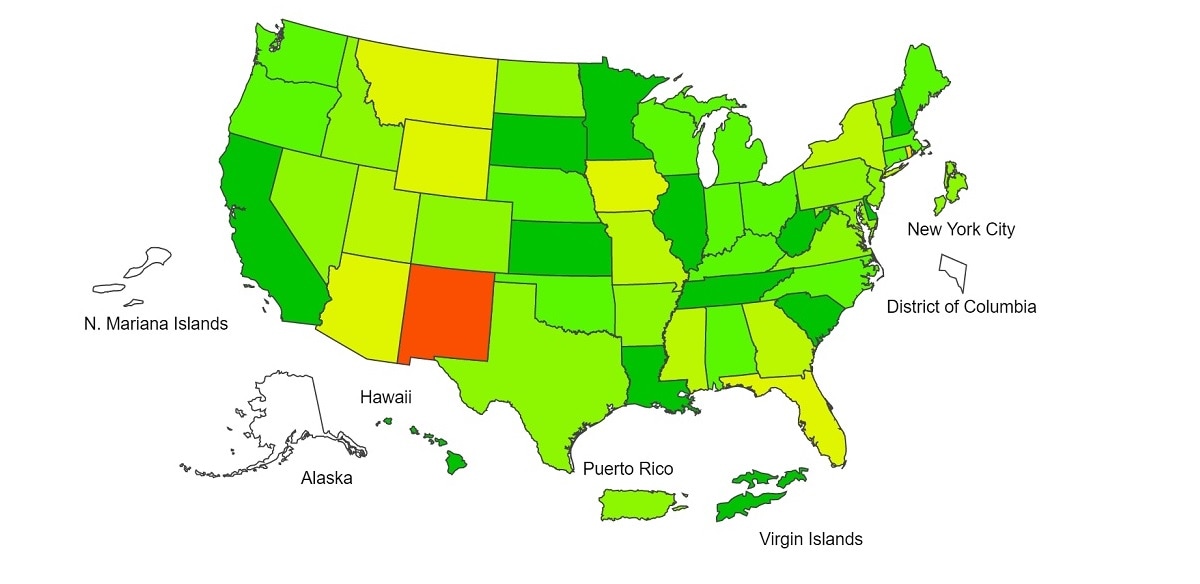
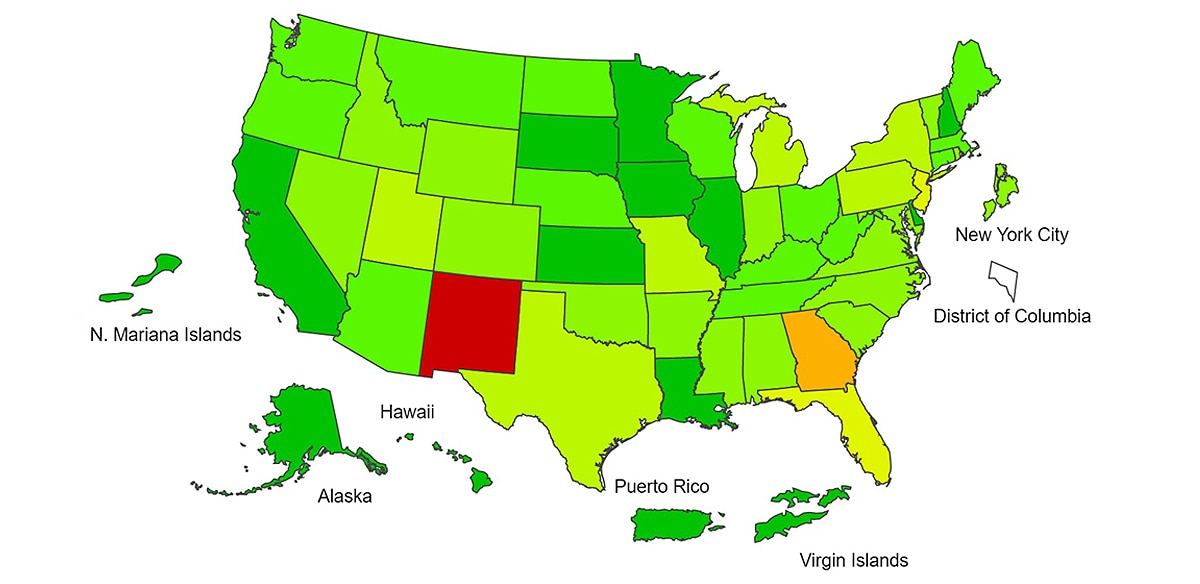
View Chart Data | View Full ScreenILI Activity Map
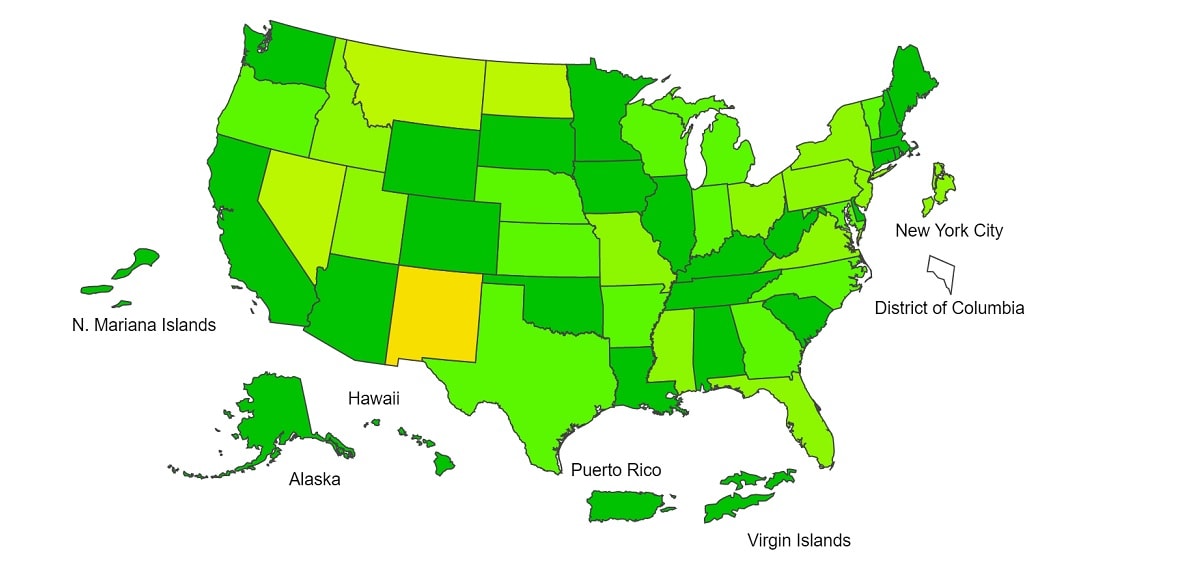
Data collected in ILINet are used to produce a measure of ILI activity* by state/jurisdiction and Core Based Statistical Areas (CBSA).
*Data collected in ILINet may disproportionally represent certain populations within a jurisdiction or CBSA, and therefore, may not accurately depict the full picture of influenza activity for the entire jurisdiction or CBSA. Differences in the data presented here by CDC and independently by some health departments likely represent differing levels of data completeness with data presented by the health department likely being the more complete.
Additional information about medically attended visits for ILI for current and past seasons:
Surveillance Methods | FluView Interactive: National, Regional, and State Data or ILI Activity Map
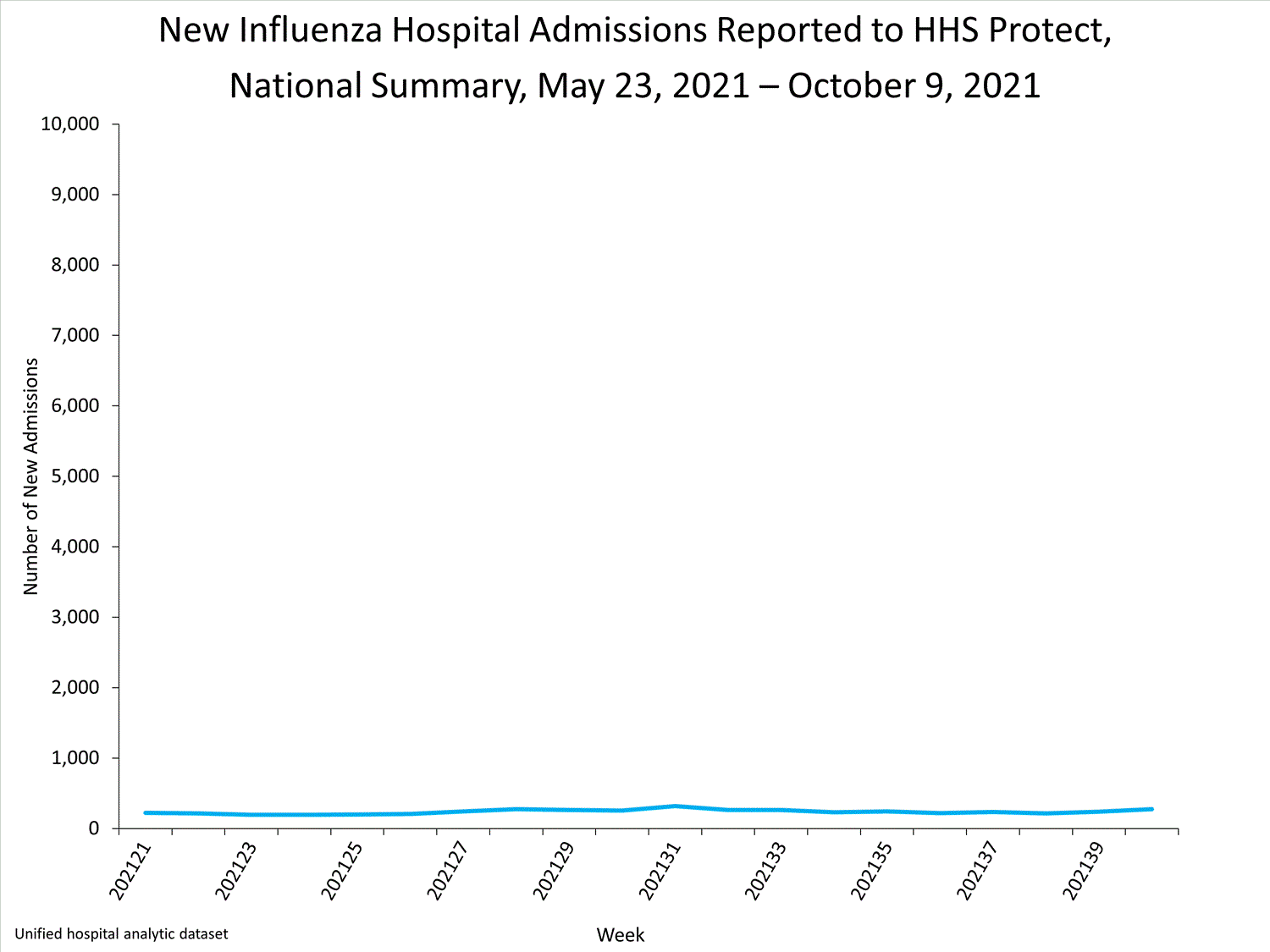
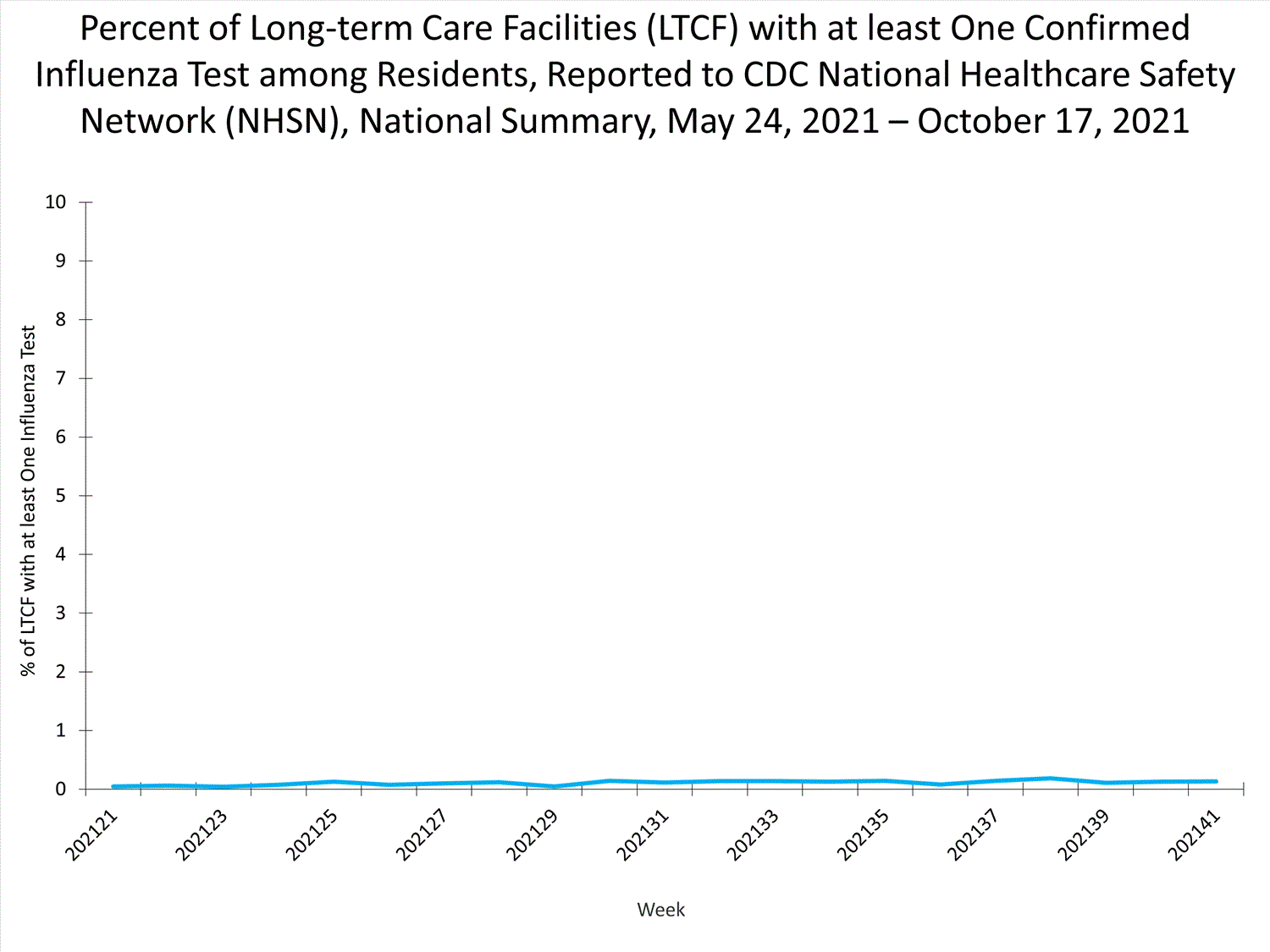
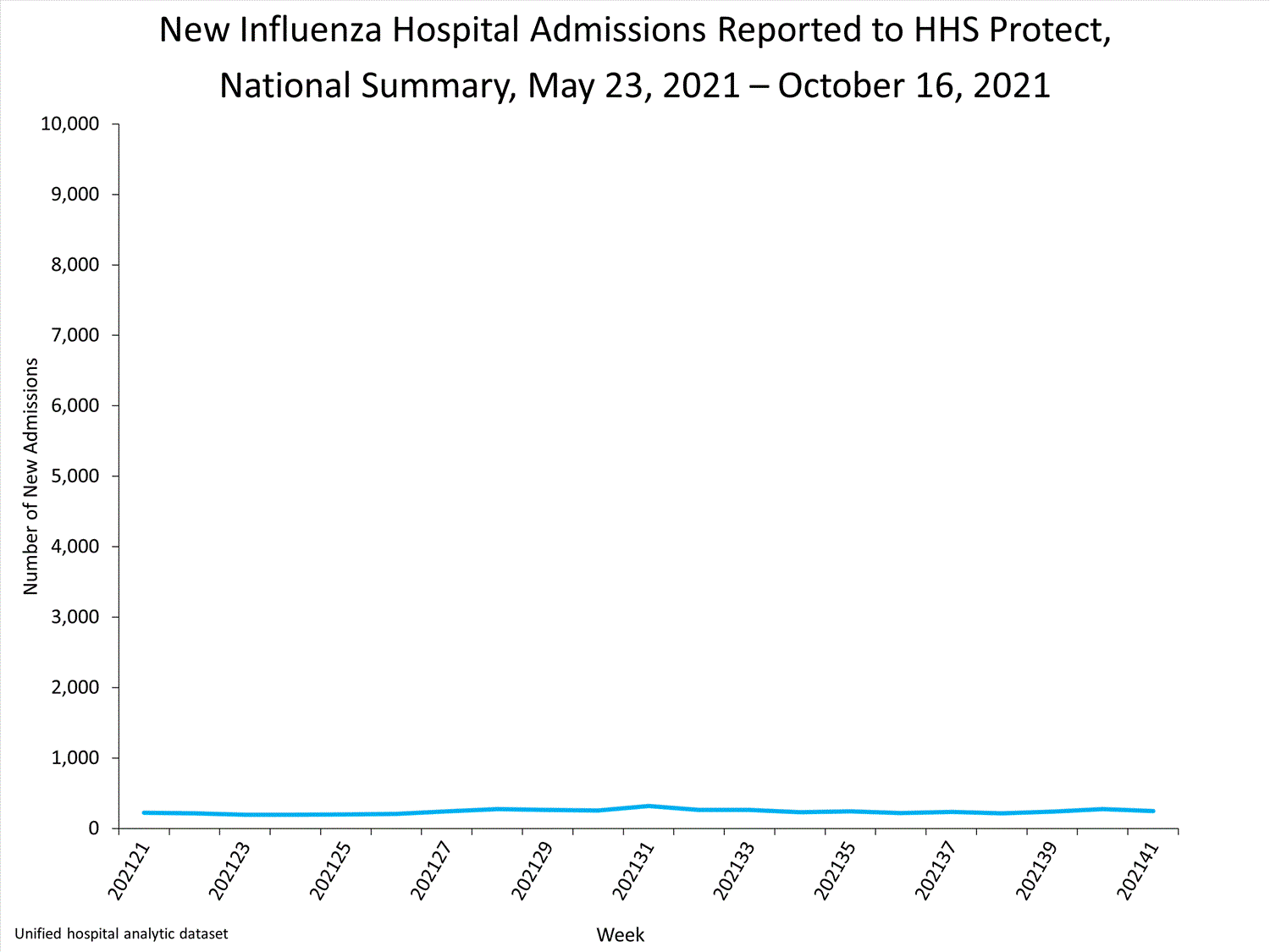
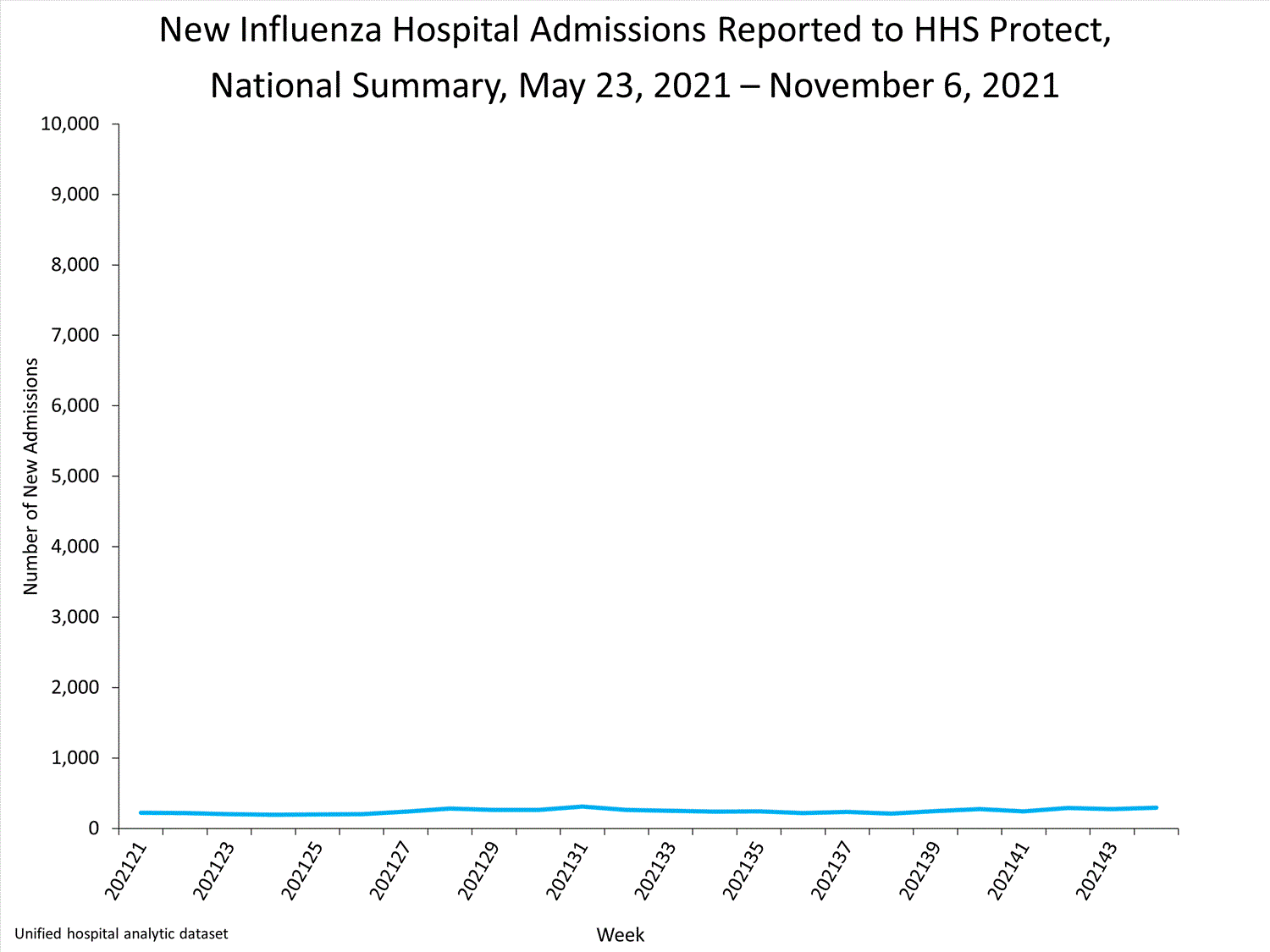
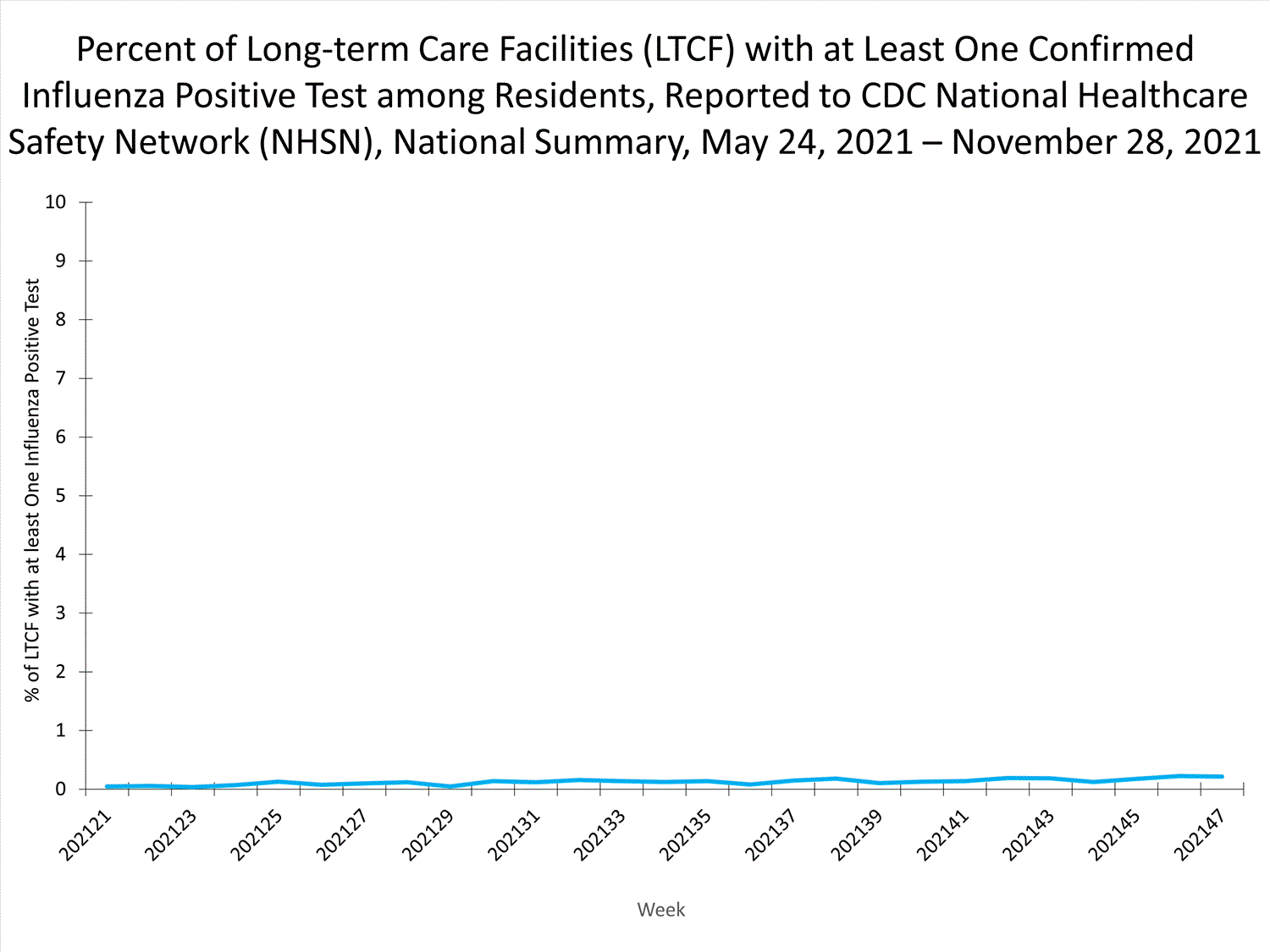
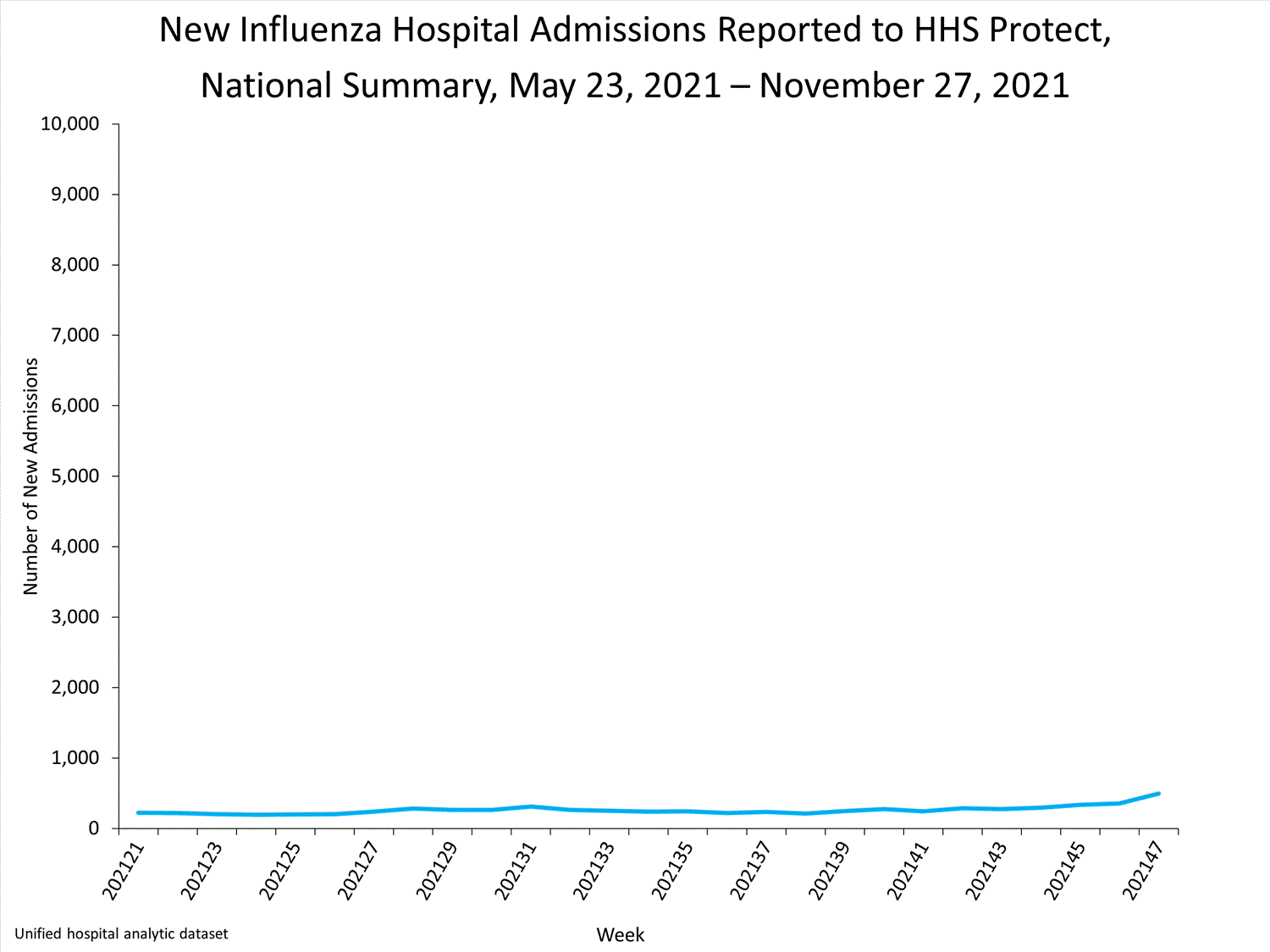
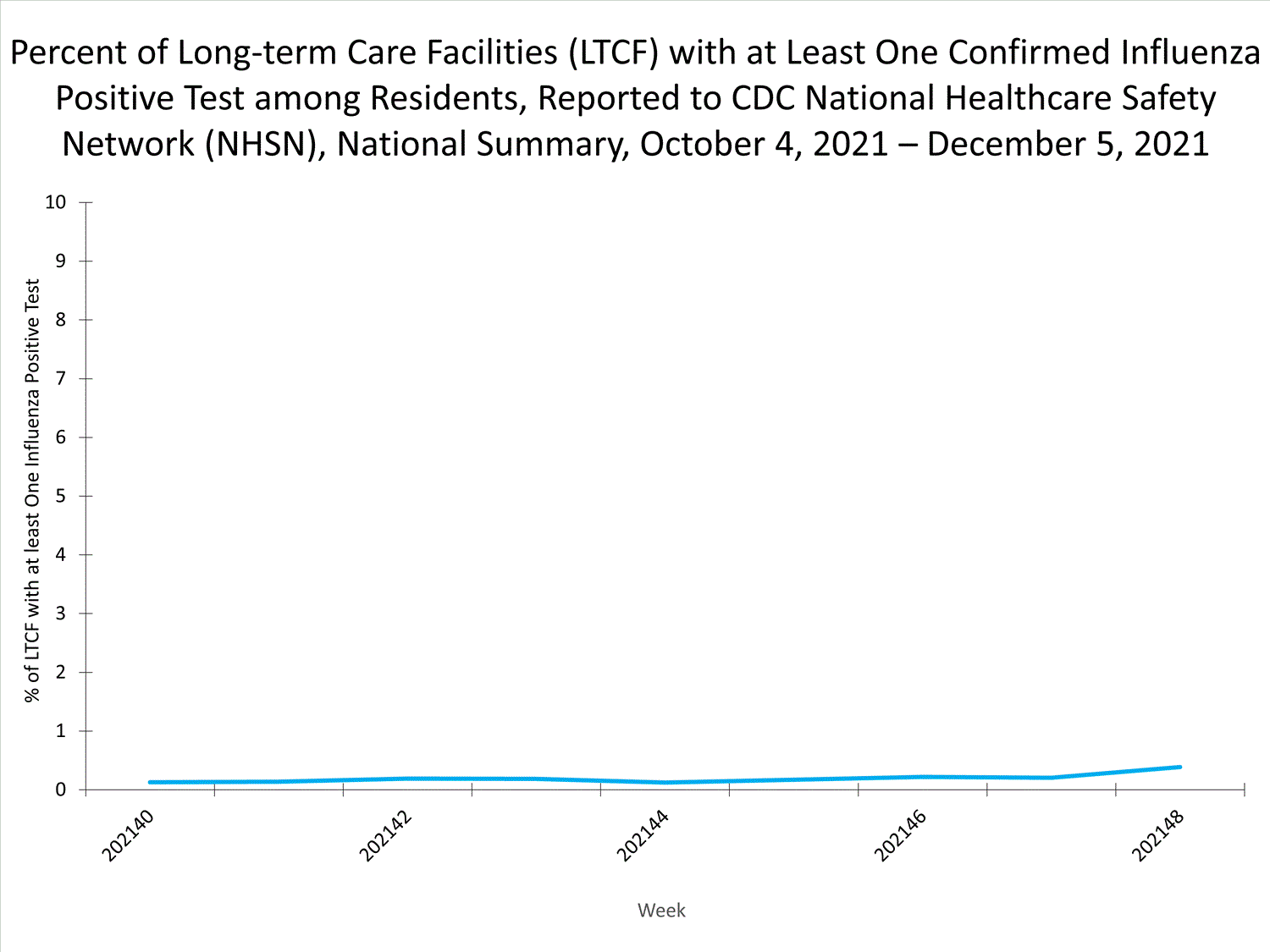
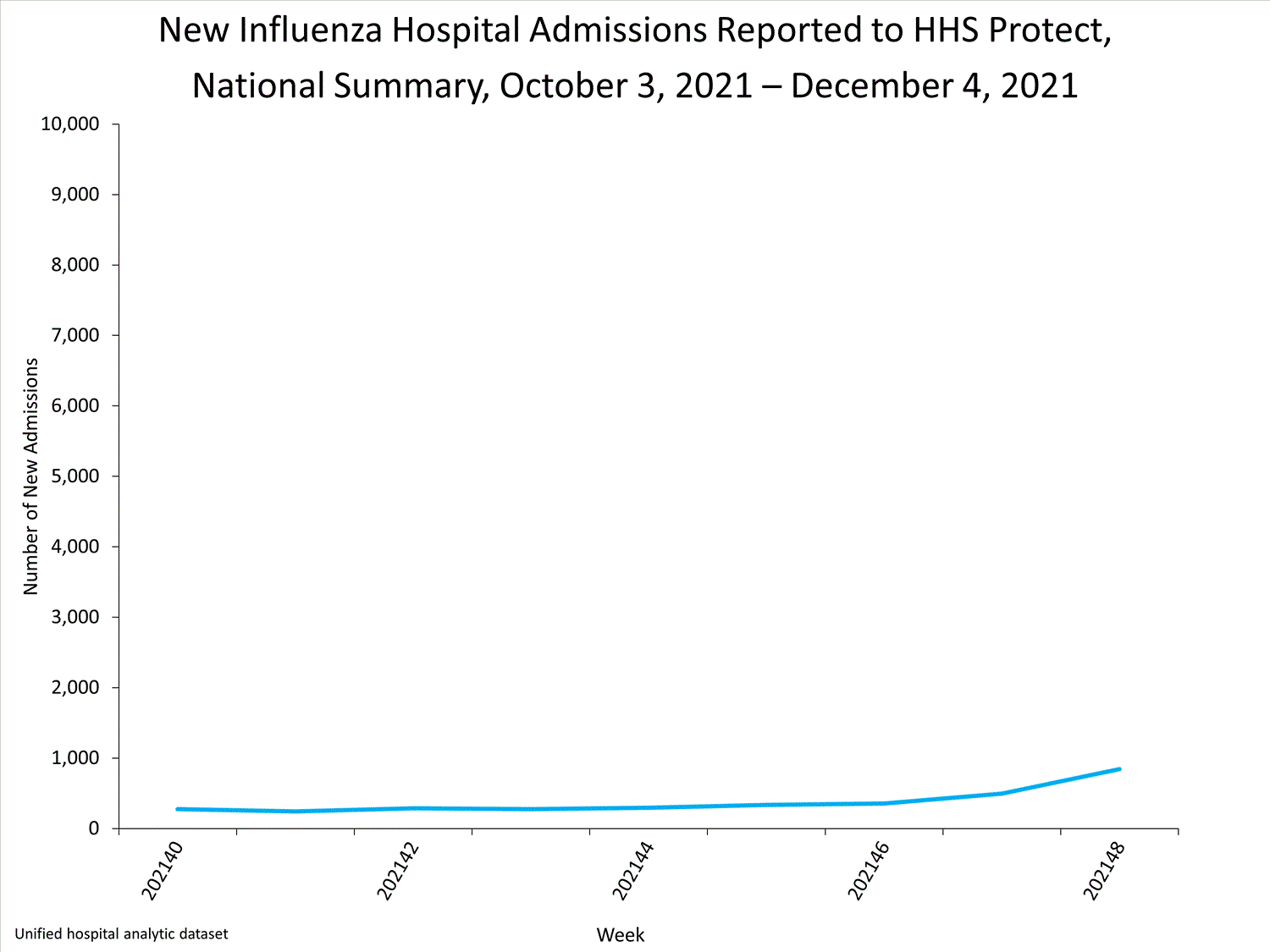
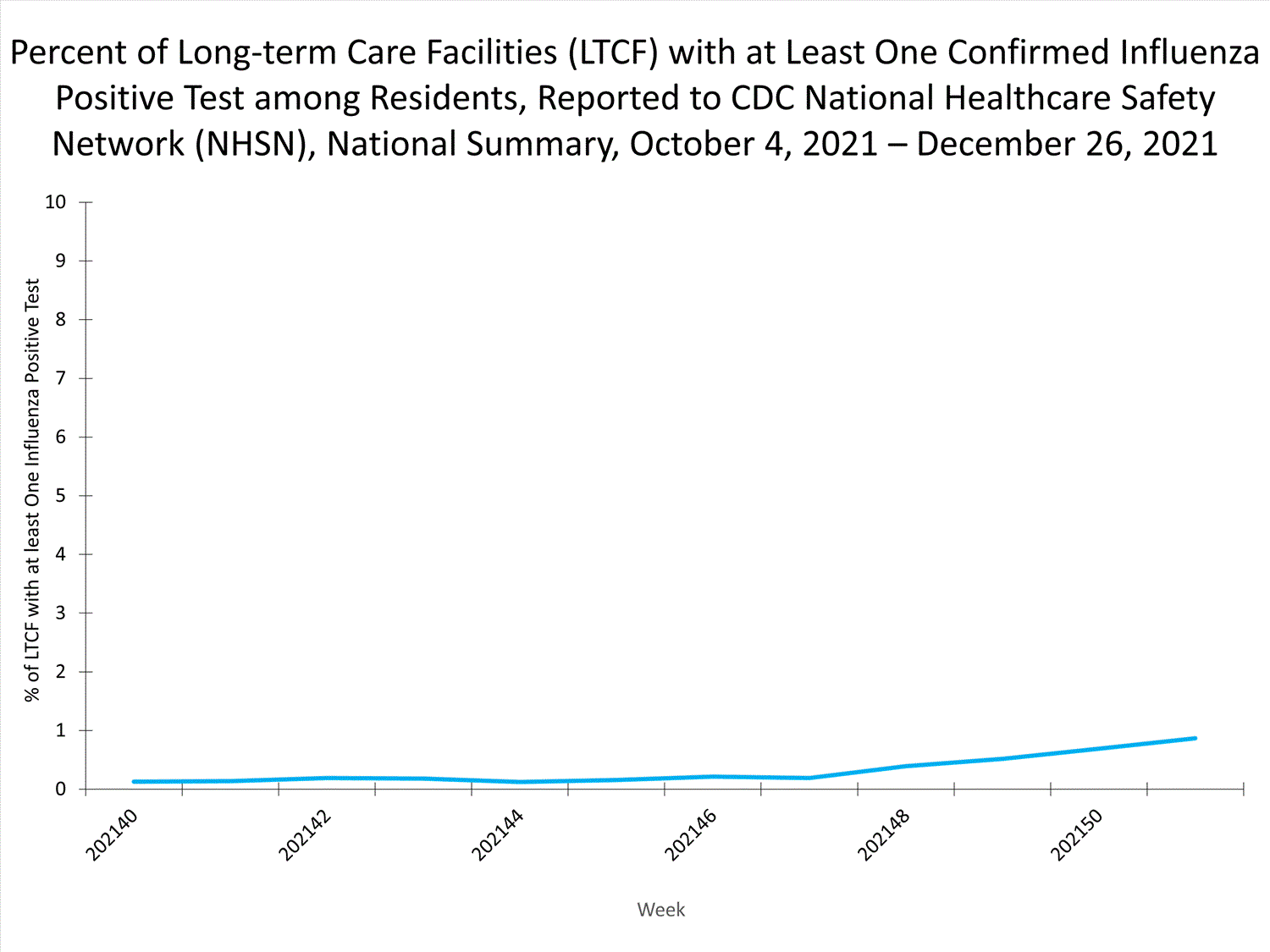
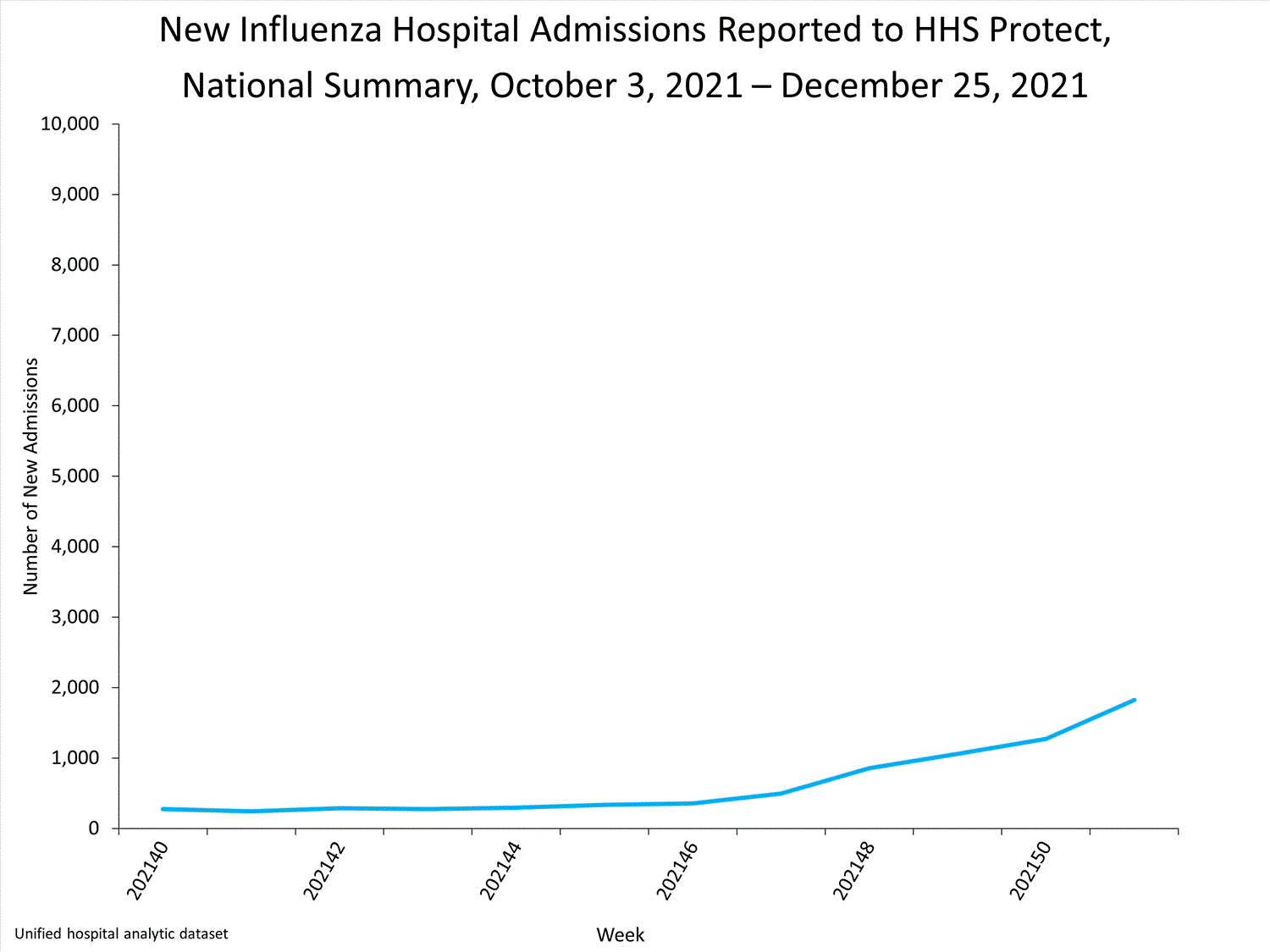
Influenza-Associated Hospitalizations:
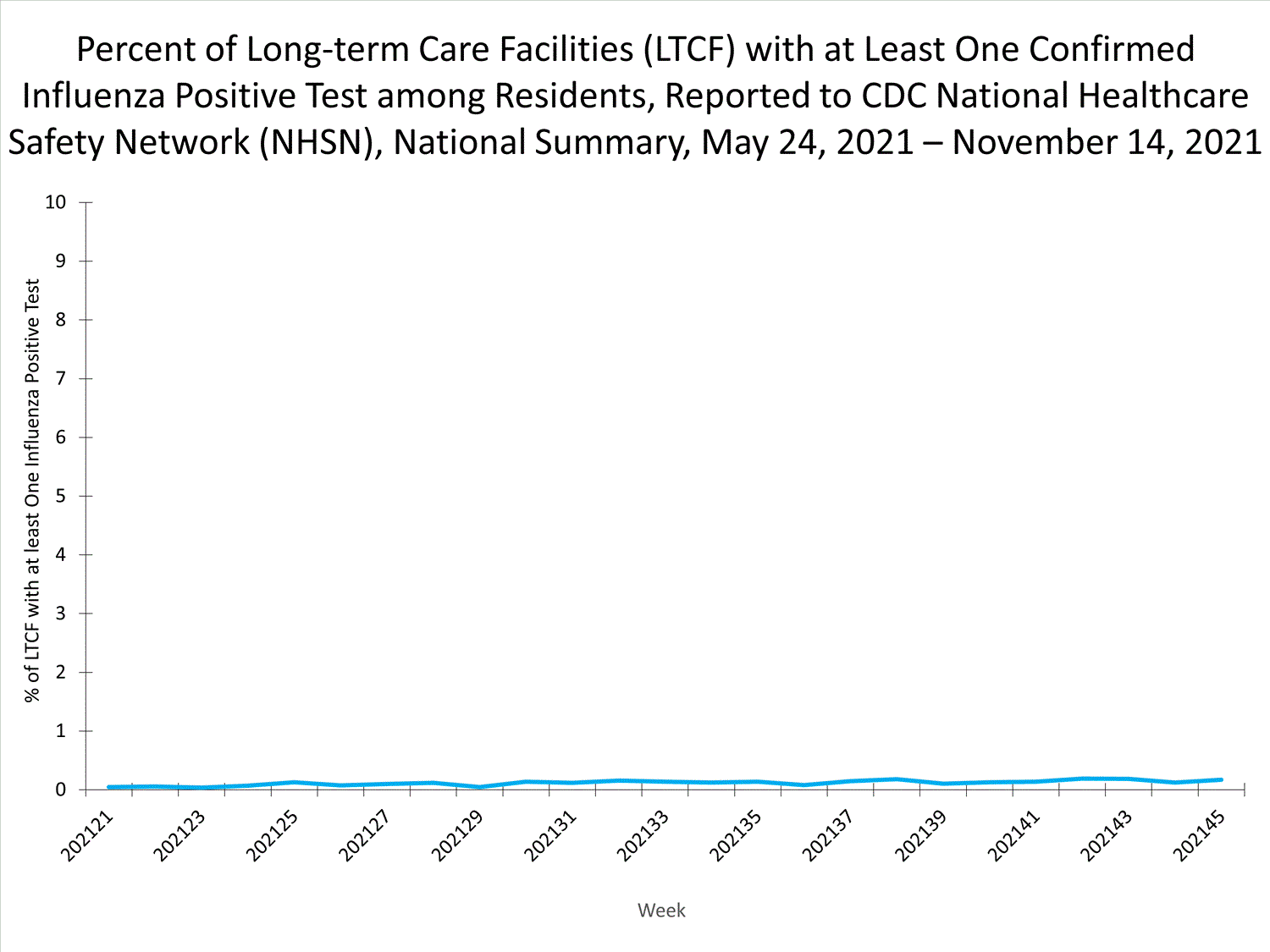
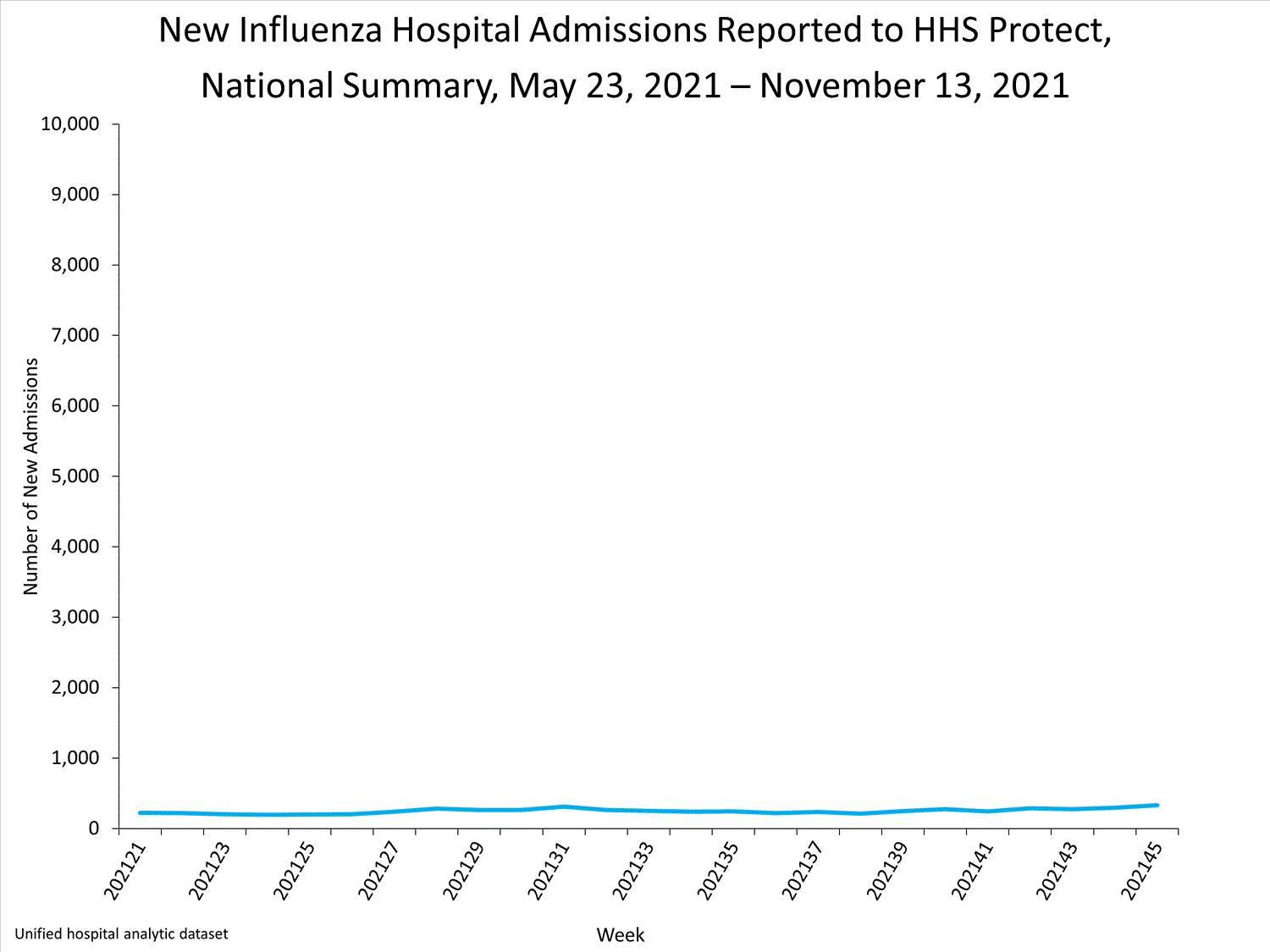
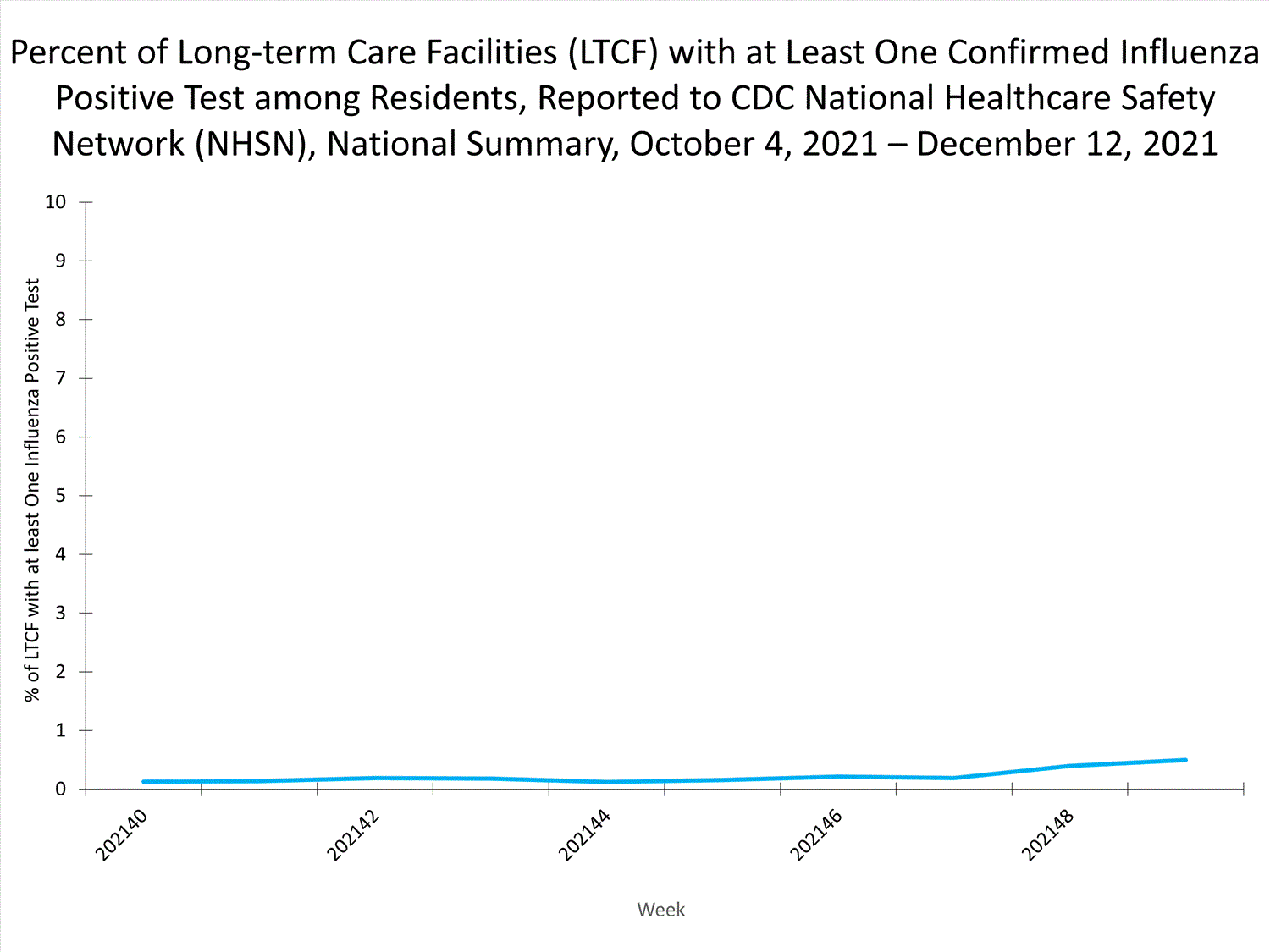
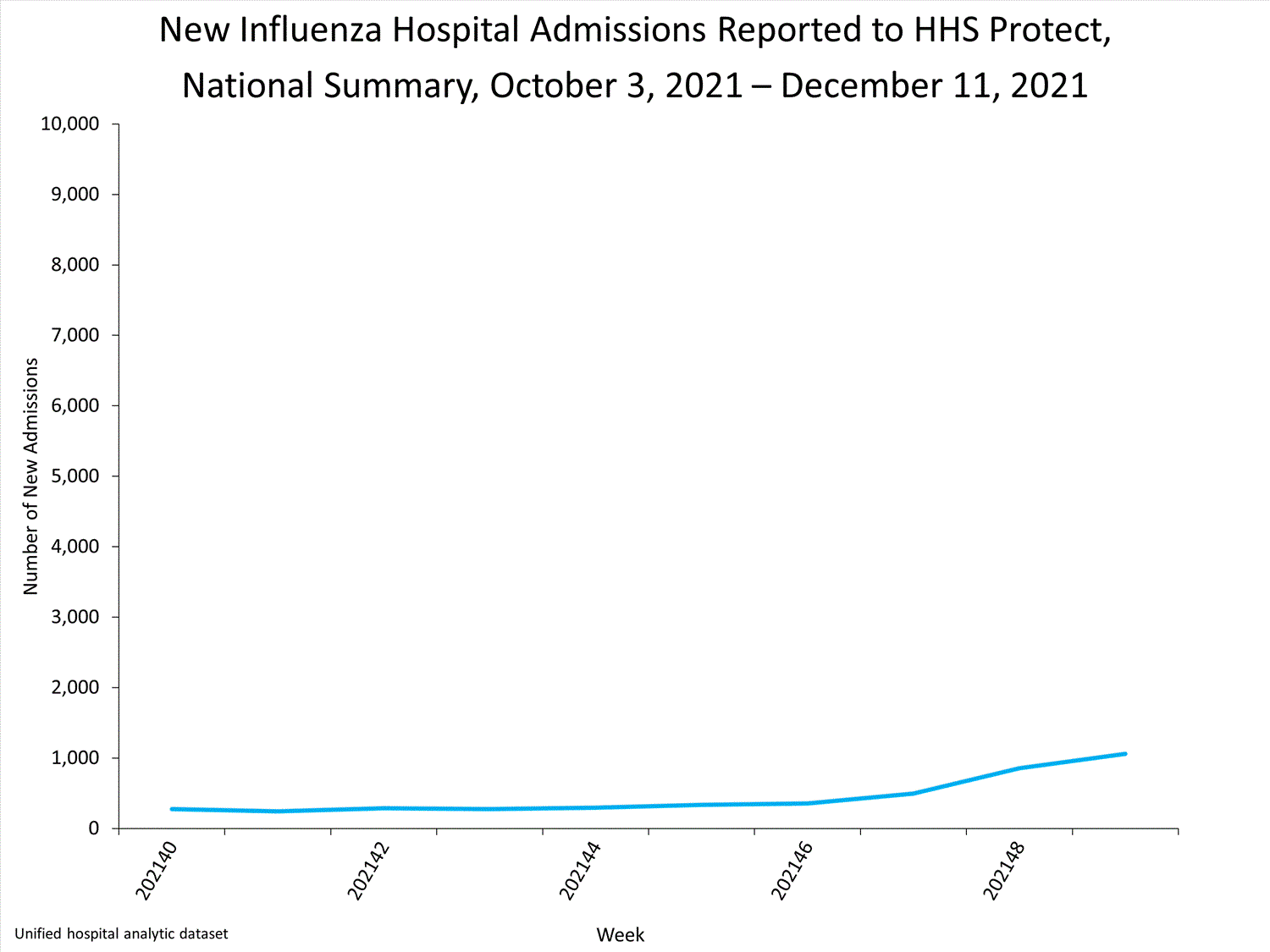
The Influenza Hospitalization Surveillance Network (FluSurv-NET) conducts population-based surveillance for laboratory-confirmed influenza-related hospitalizations in select counties in 14 states and represents approximately 9% of the U.S. population. As in previous seasons, patients admitted for laboratory-confirmed influenza-related hospitalization after April 30, 2021, will not be included in FluSurv-NET. Data on patients admitted through April 30, 2021, will continue to be updated as additional information is received.
Additional hospitalization surveillance information for current and past seasons and additional age groups:
Surveillance Methods | FluView Interactive: Rates by Age or Patient Characteristics
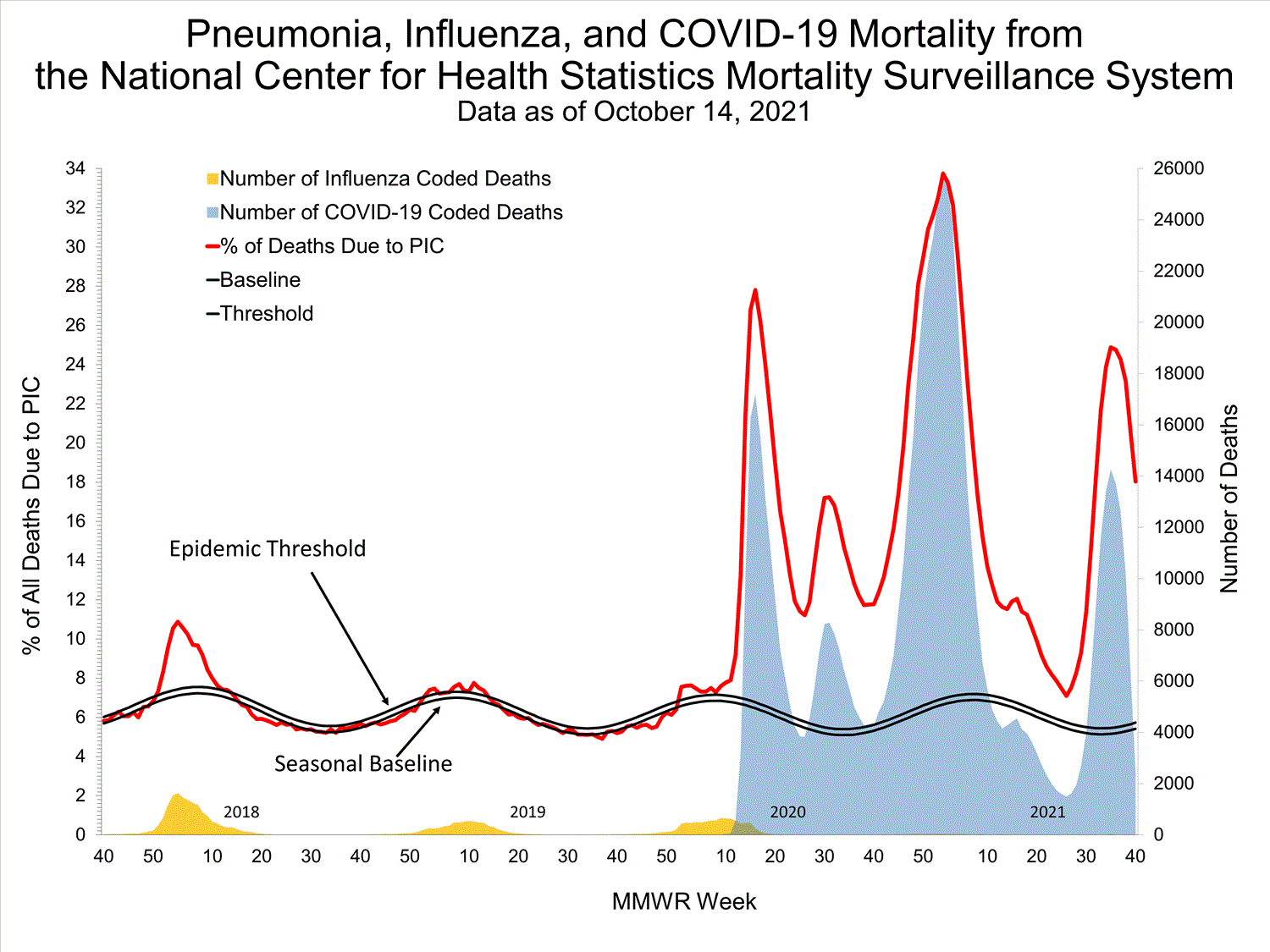
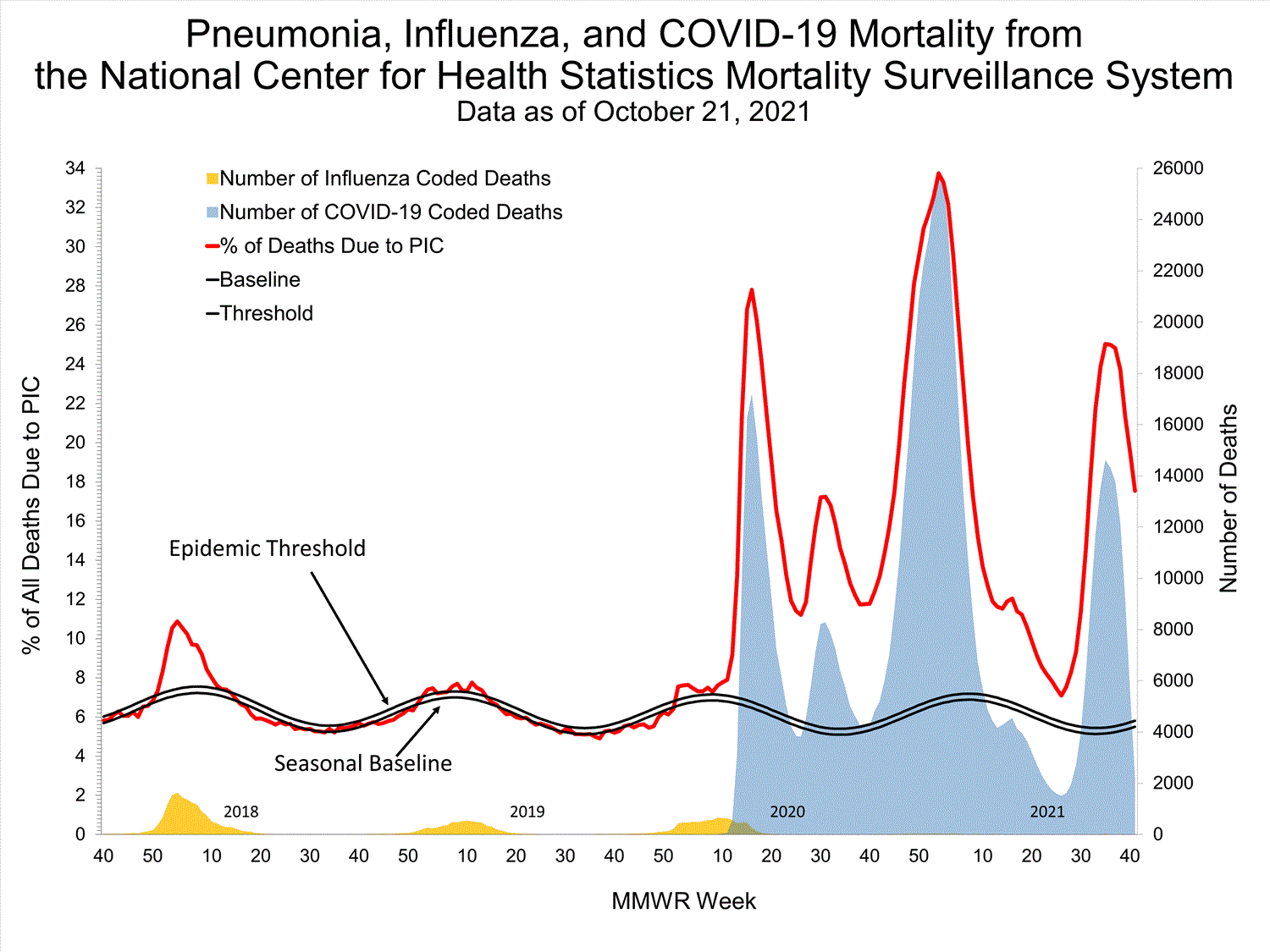
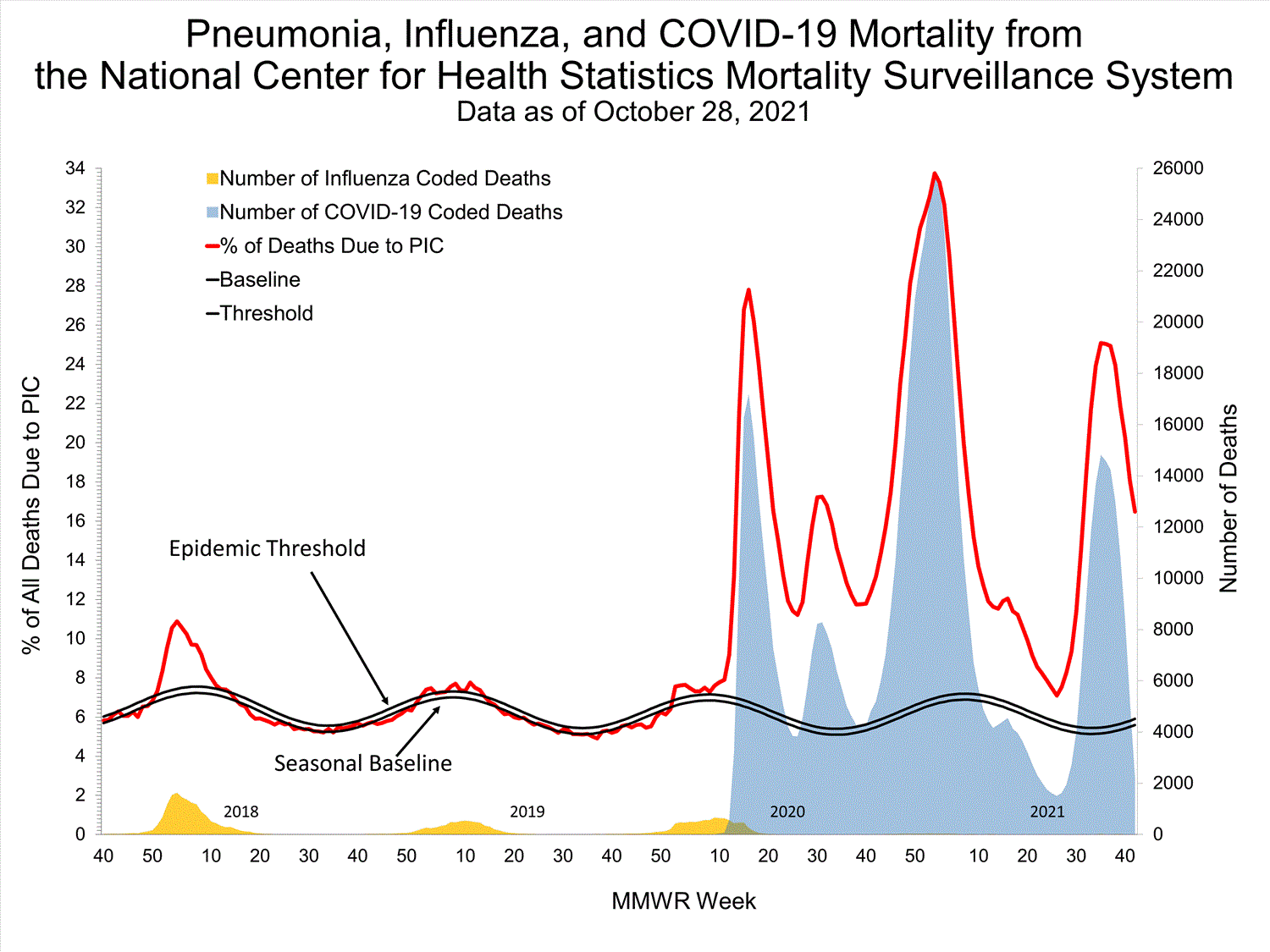
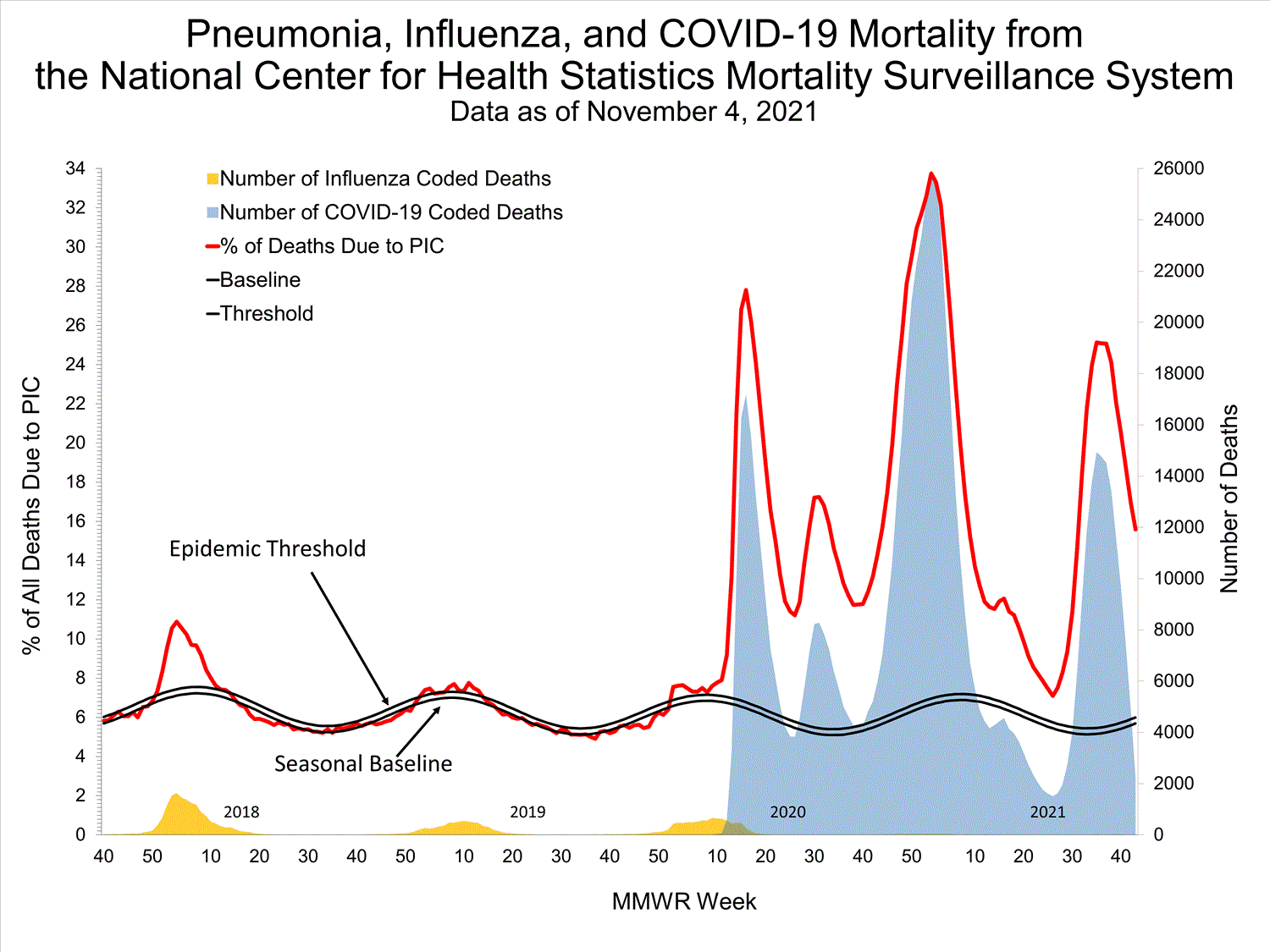
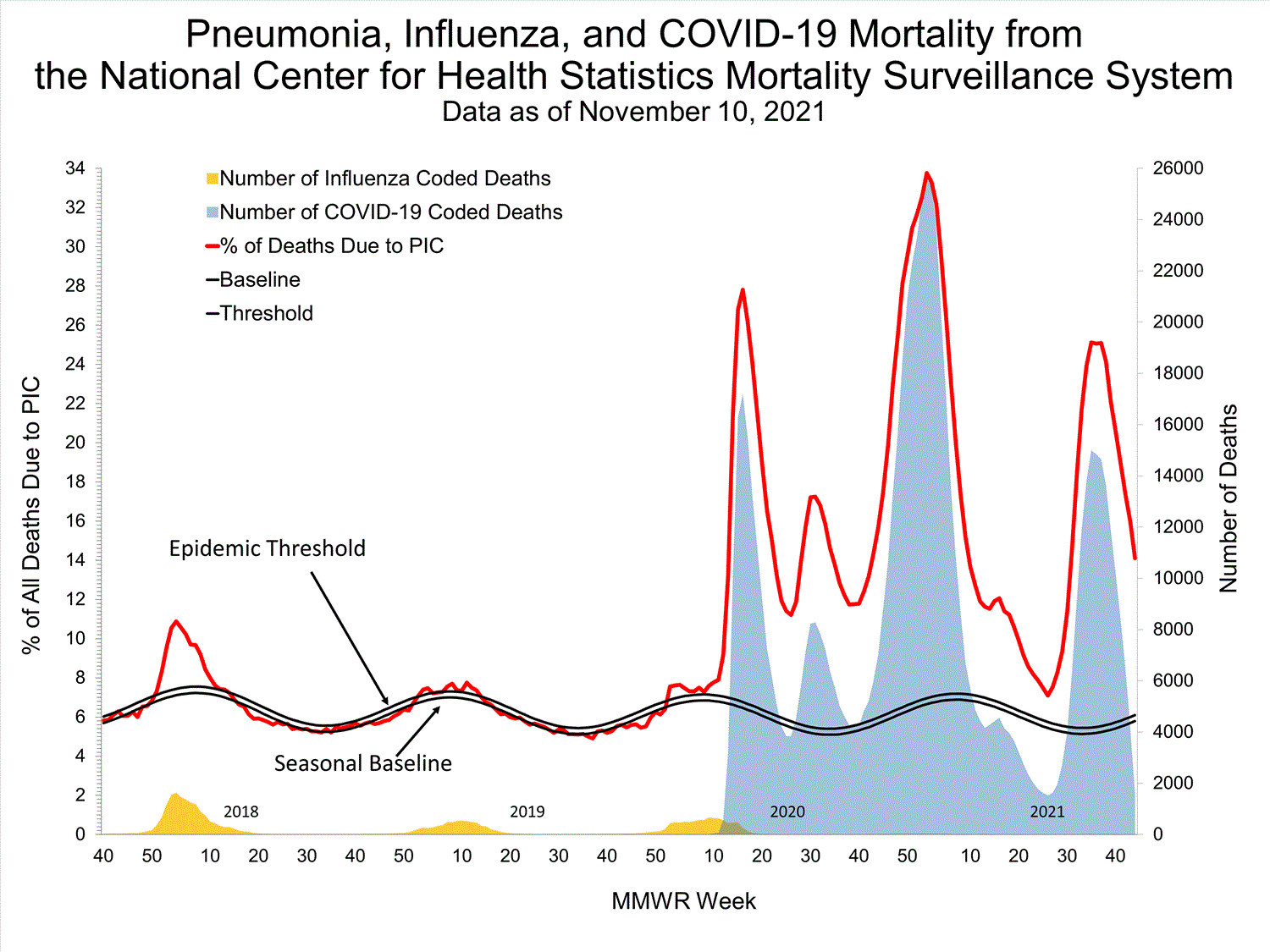
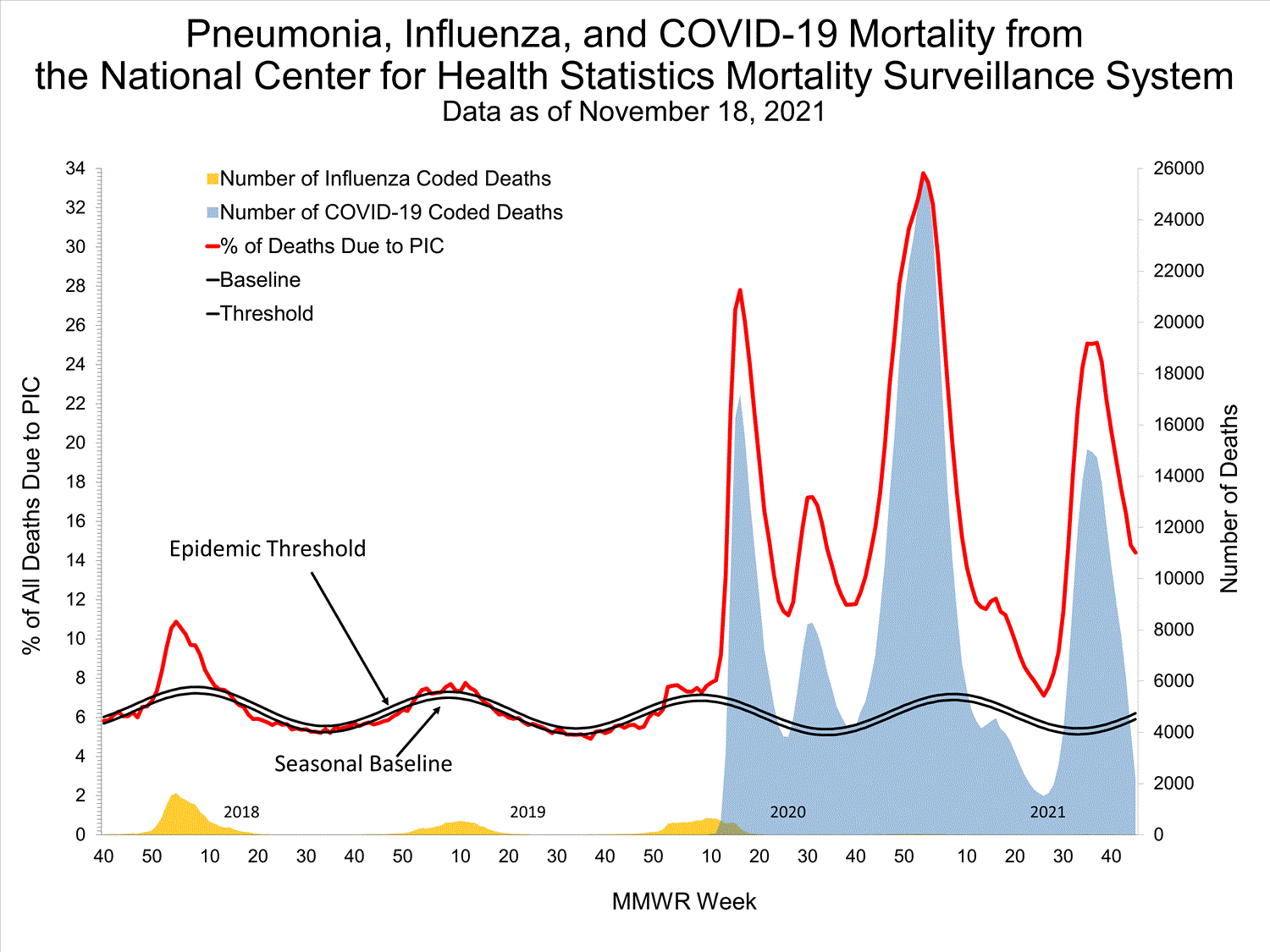
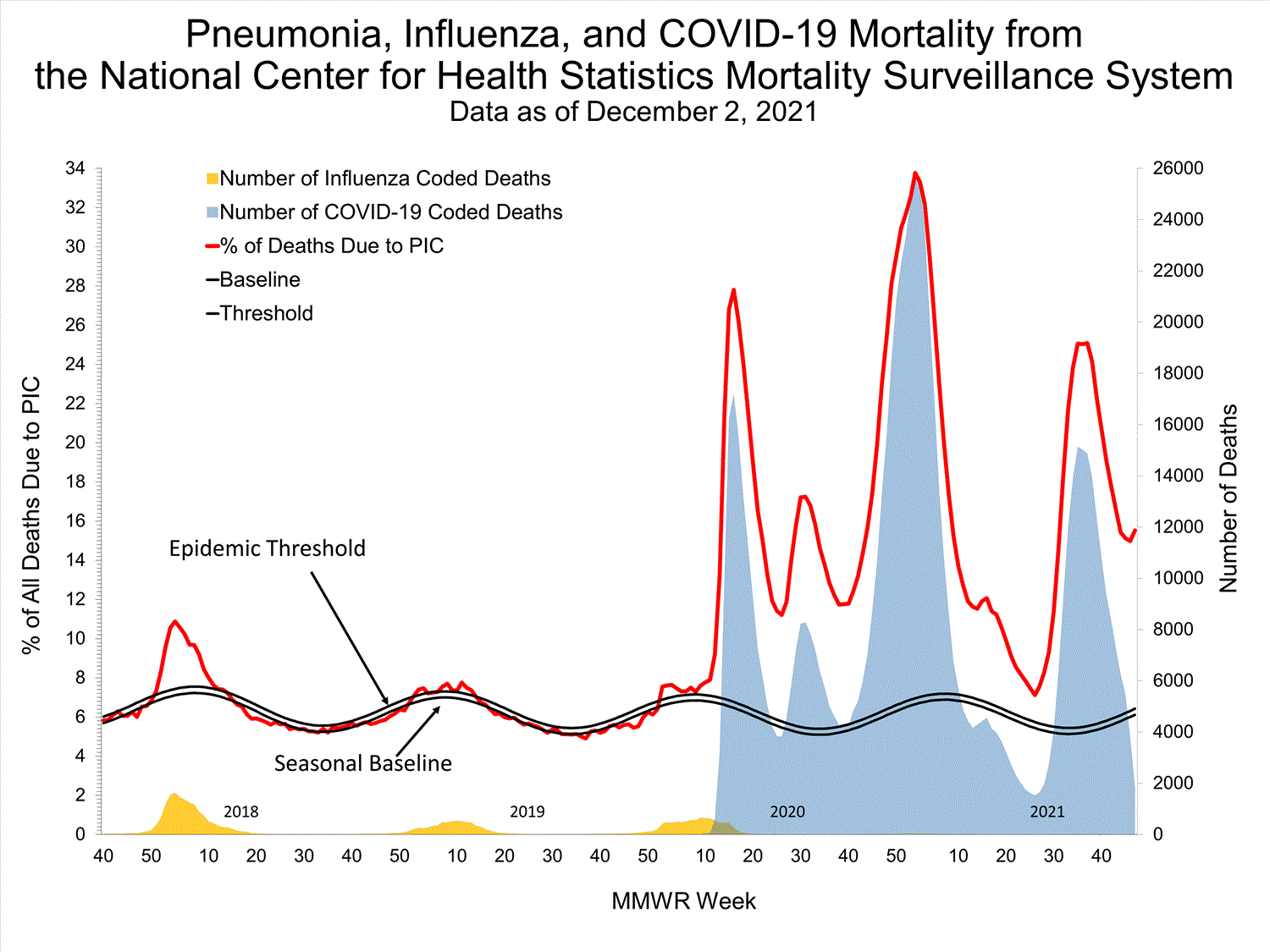
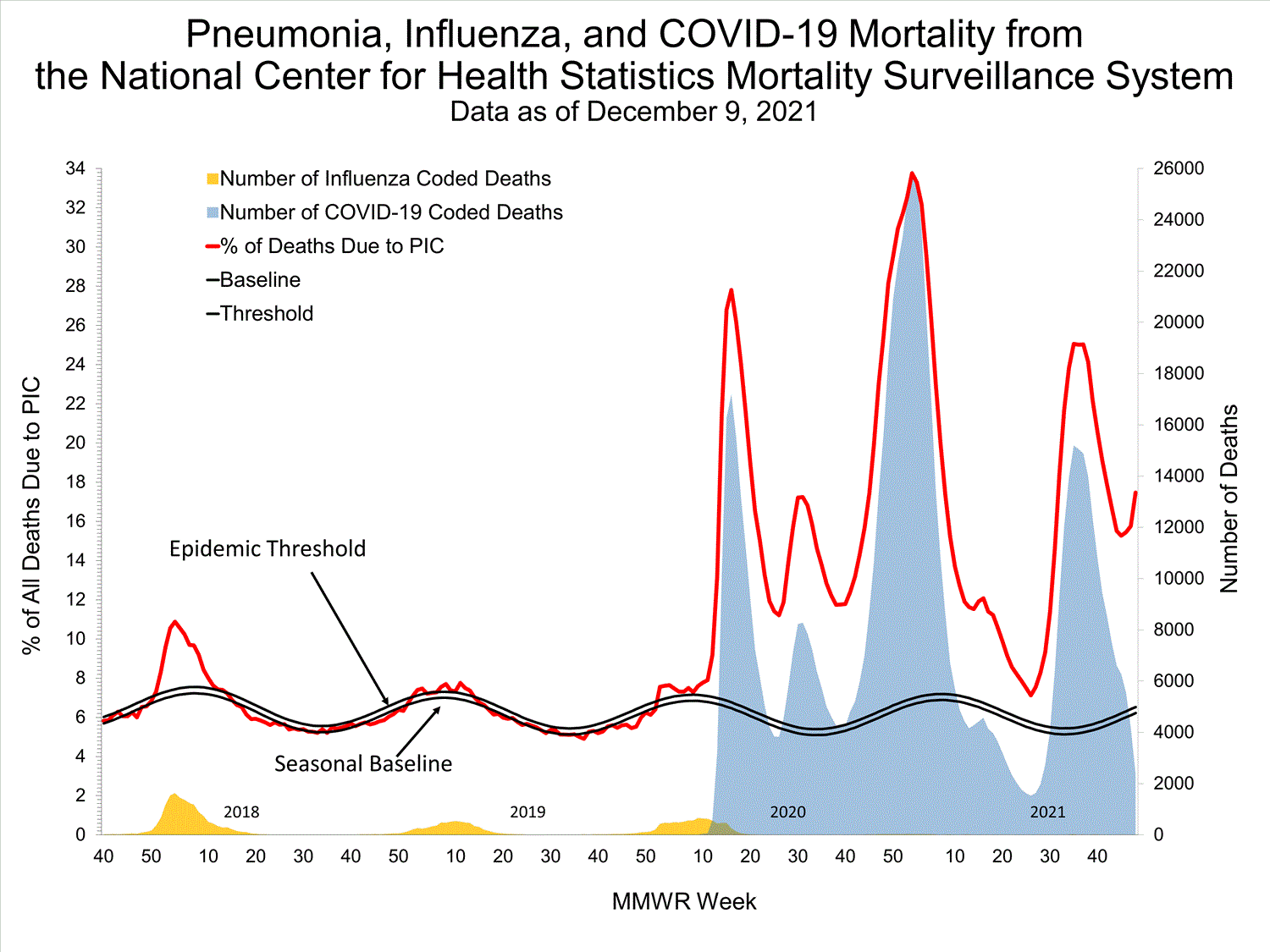
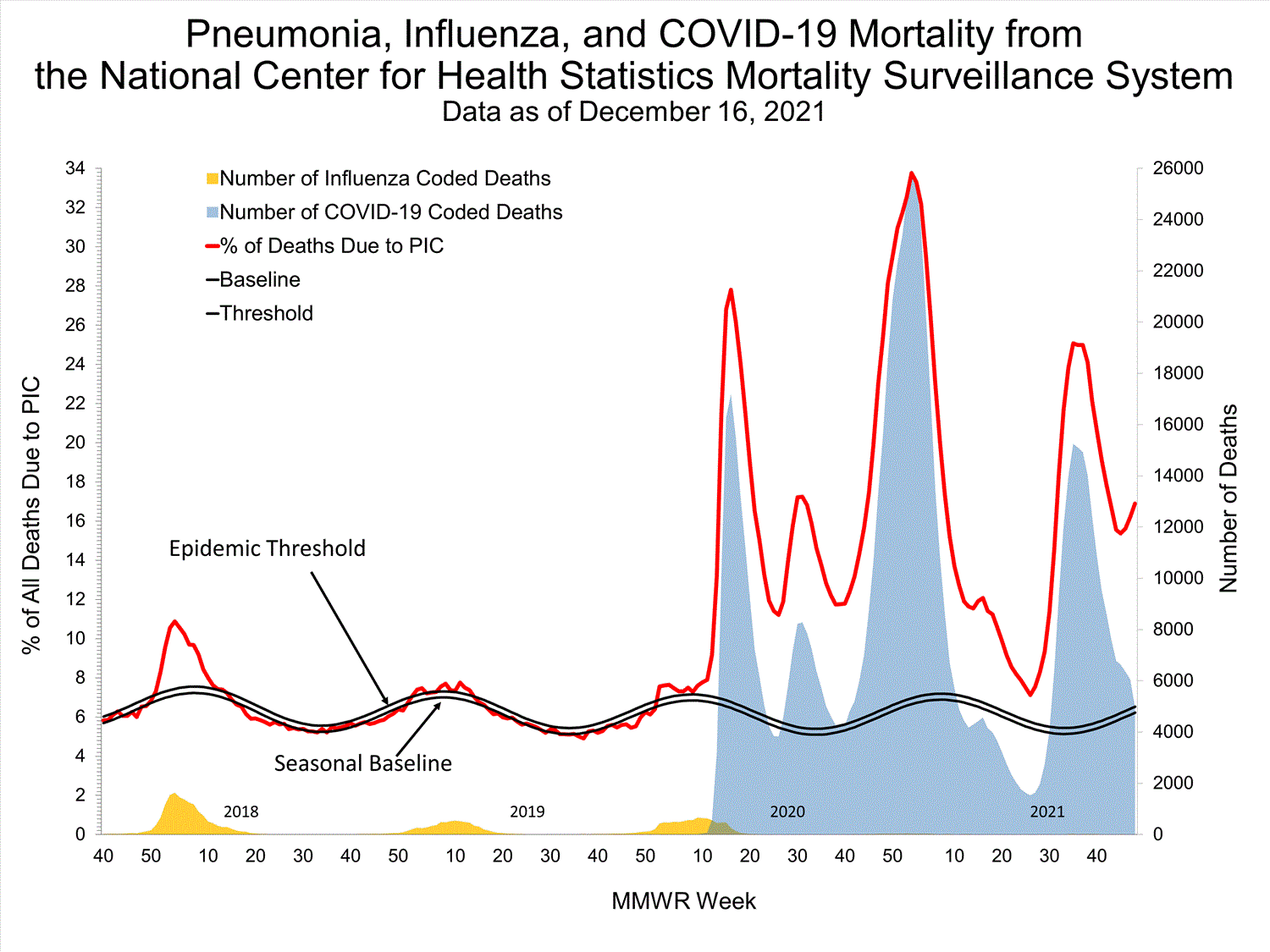
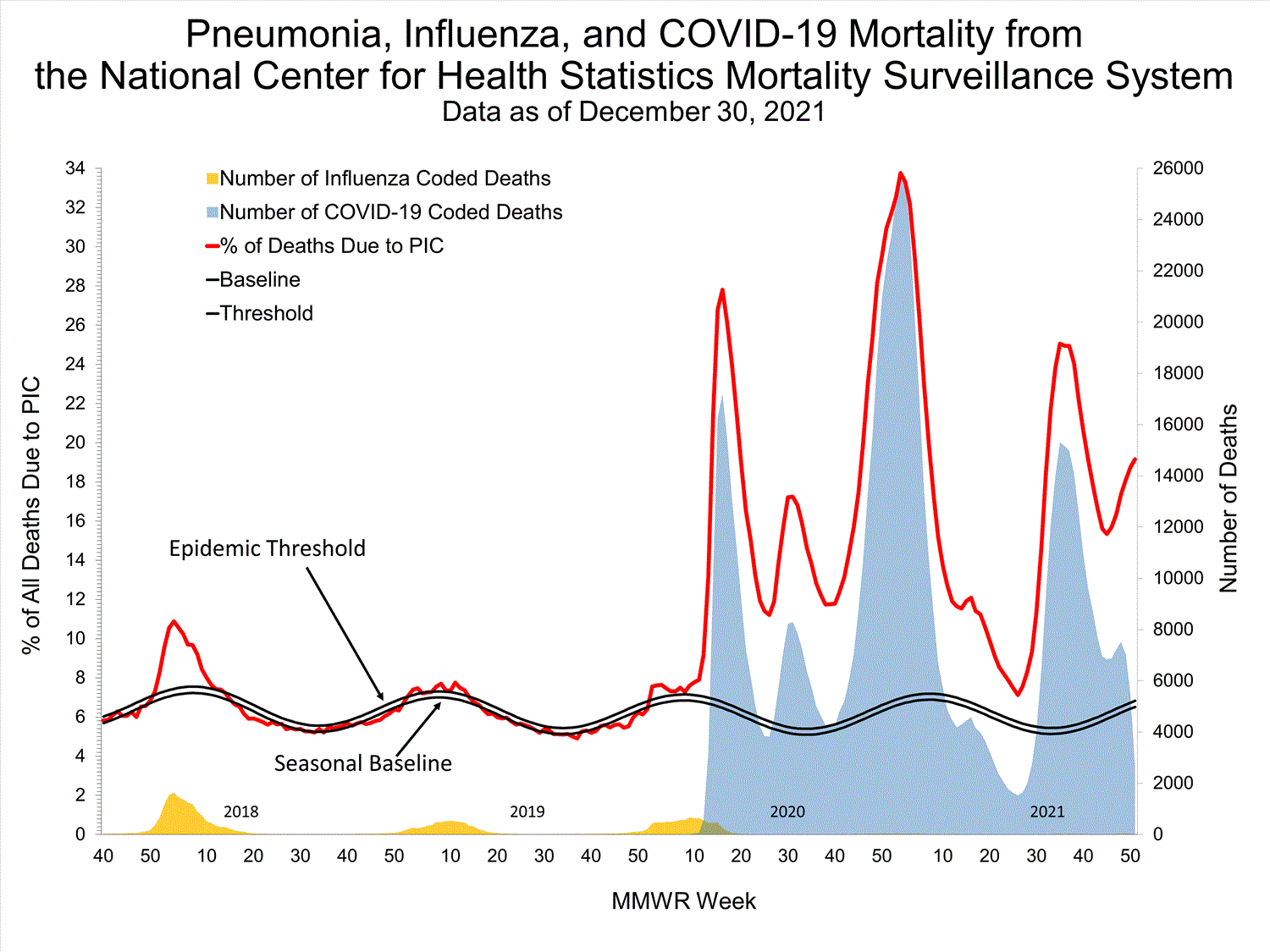
National Center for Health Statistics (NCHS) Mortality Surveillance
Based on NCHS mortality surveillance data available on October 7, 2021, 18.7% of the deaths that occurred during the week ending October 2, 2021 (week 39), were due to pneumonia, influenza, and/or COVID-19 (PIC). This percentage is above the epidemic threshold of 5.7% for this week. Among the 3,296 PIC deaths reported for this week, 2,607 had COVID-19 listed as an underlying or contributing cause of death on the death certificate, and four listed influenza, indicating that current PIC mortality is due primarily to COVID-19 and not influenza. The data presented are preliminary and may change as more data are received and processed.
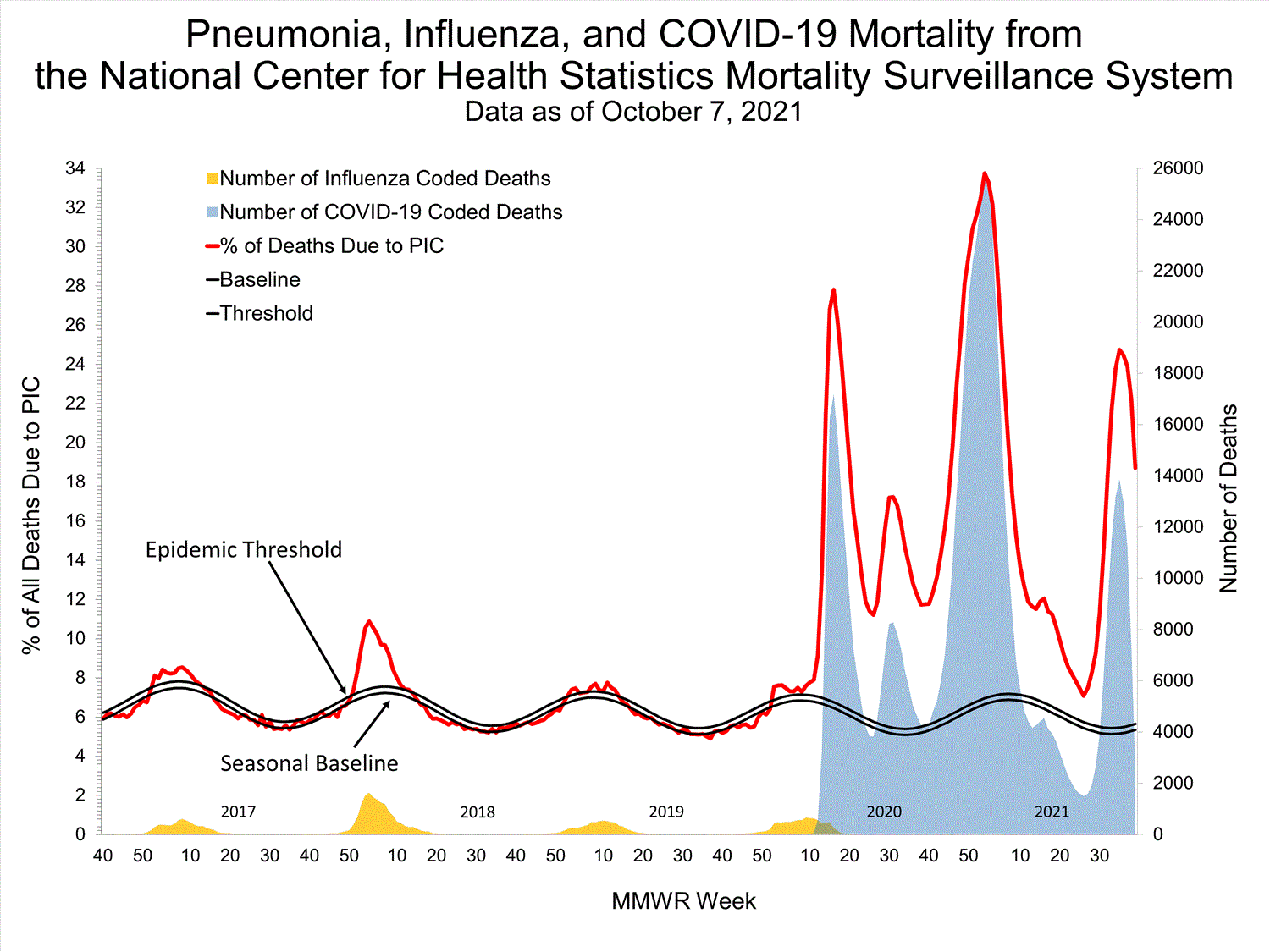
View Chart Dataexcel icon | View Full Screen
Additional pneumonia and influenza mortality surveillance information for current and past seasons:
Surveillance Methods | FluView Interactive
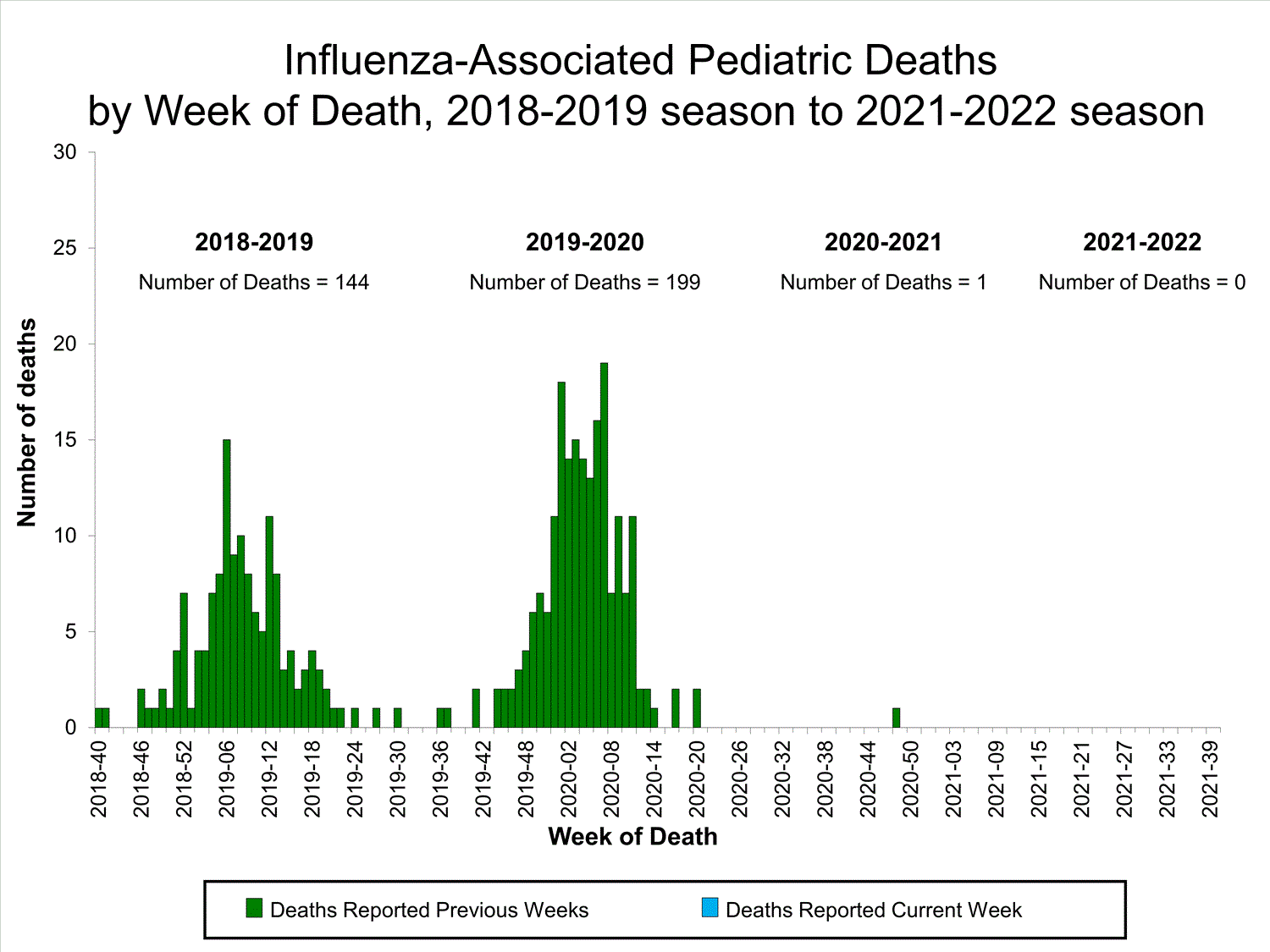
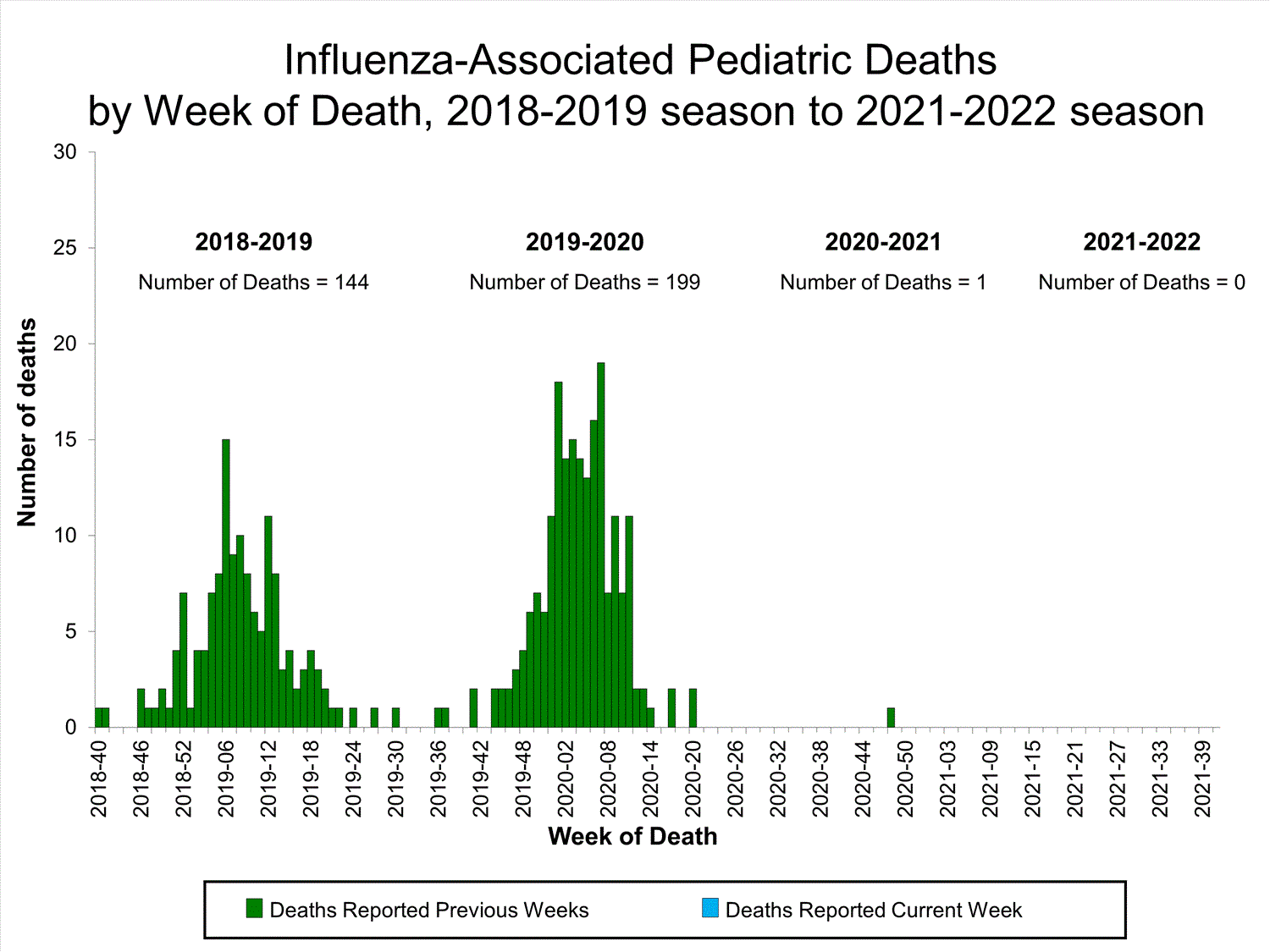
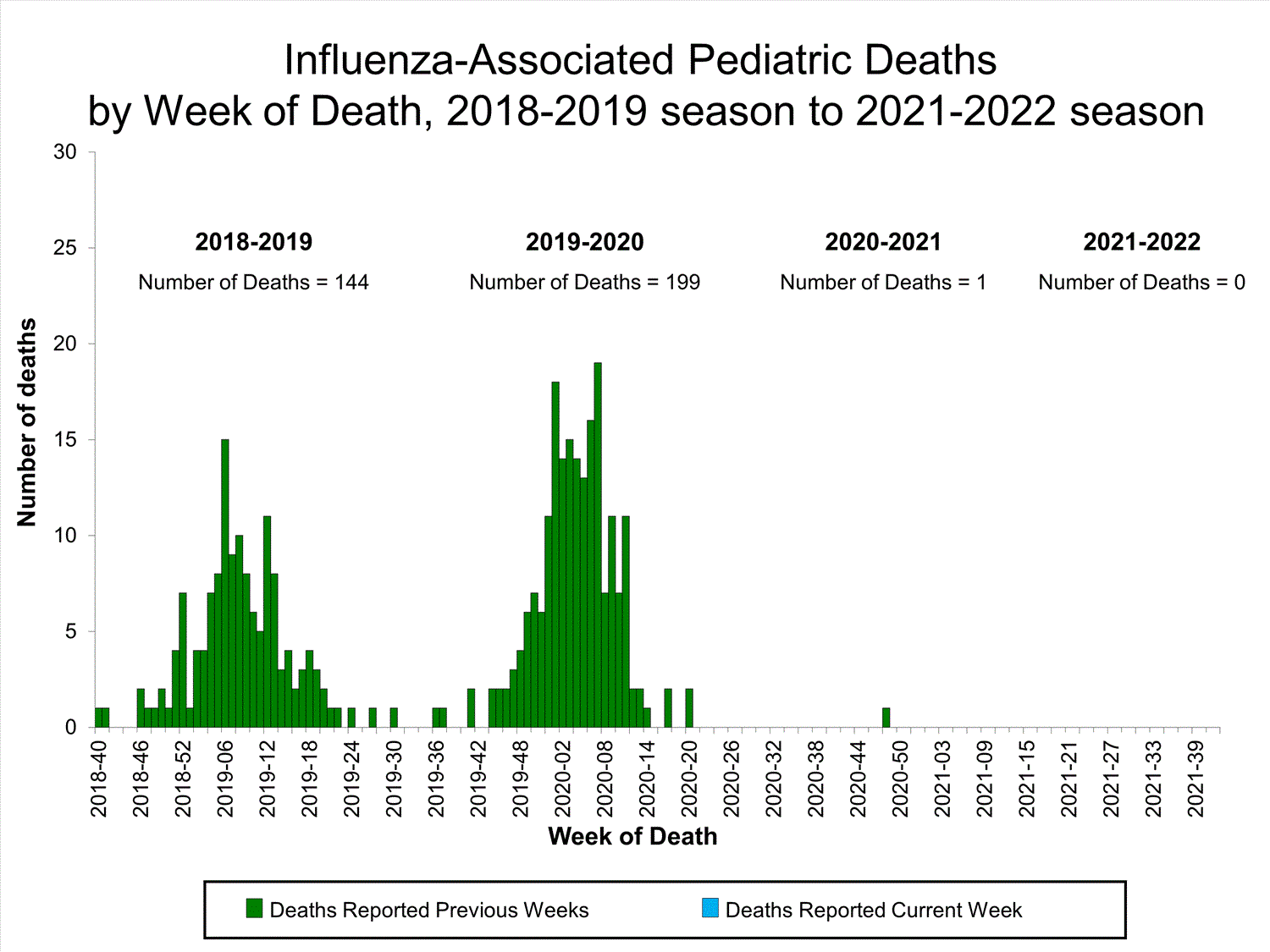
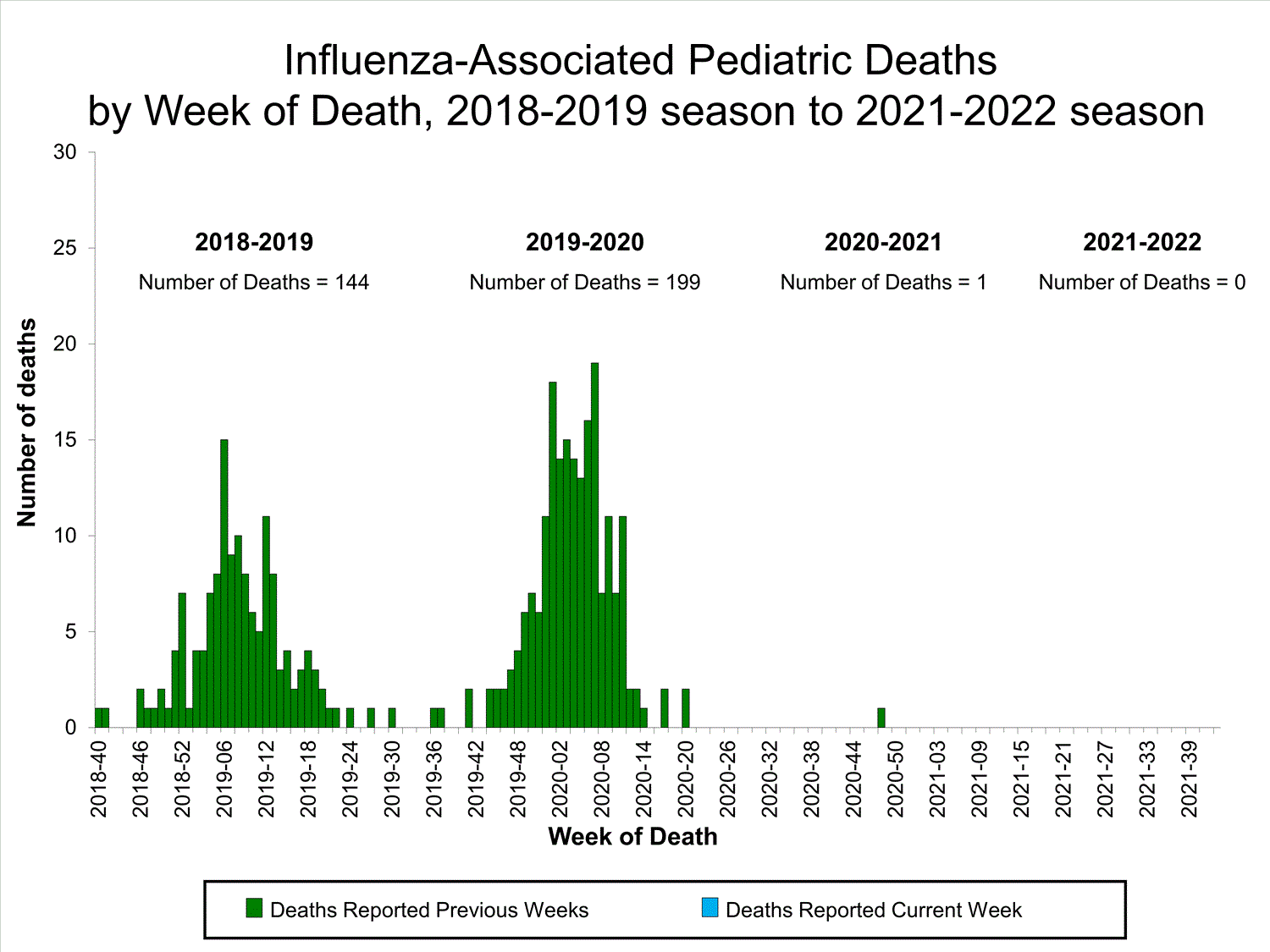
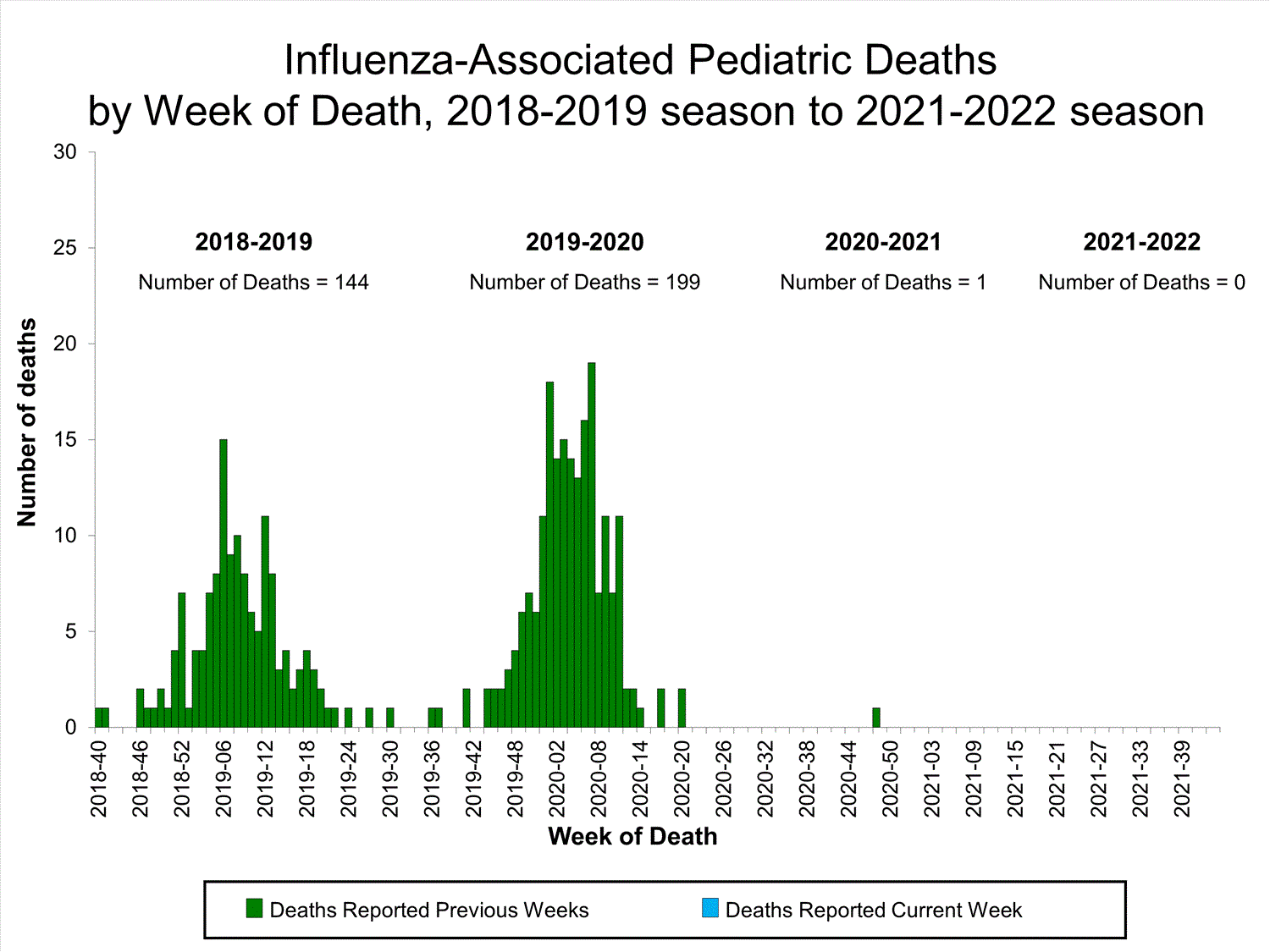
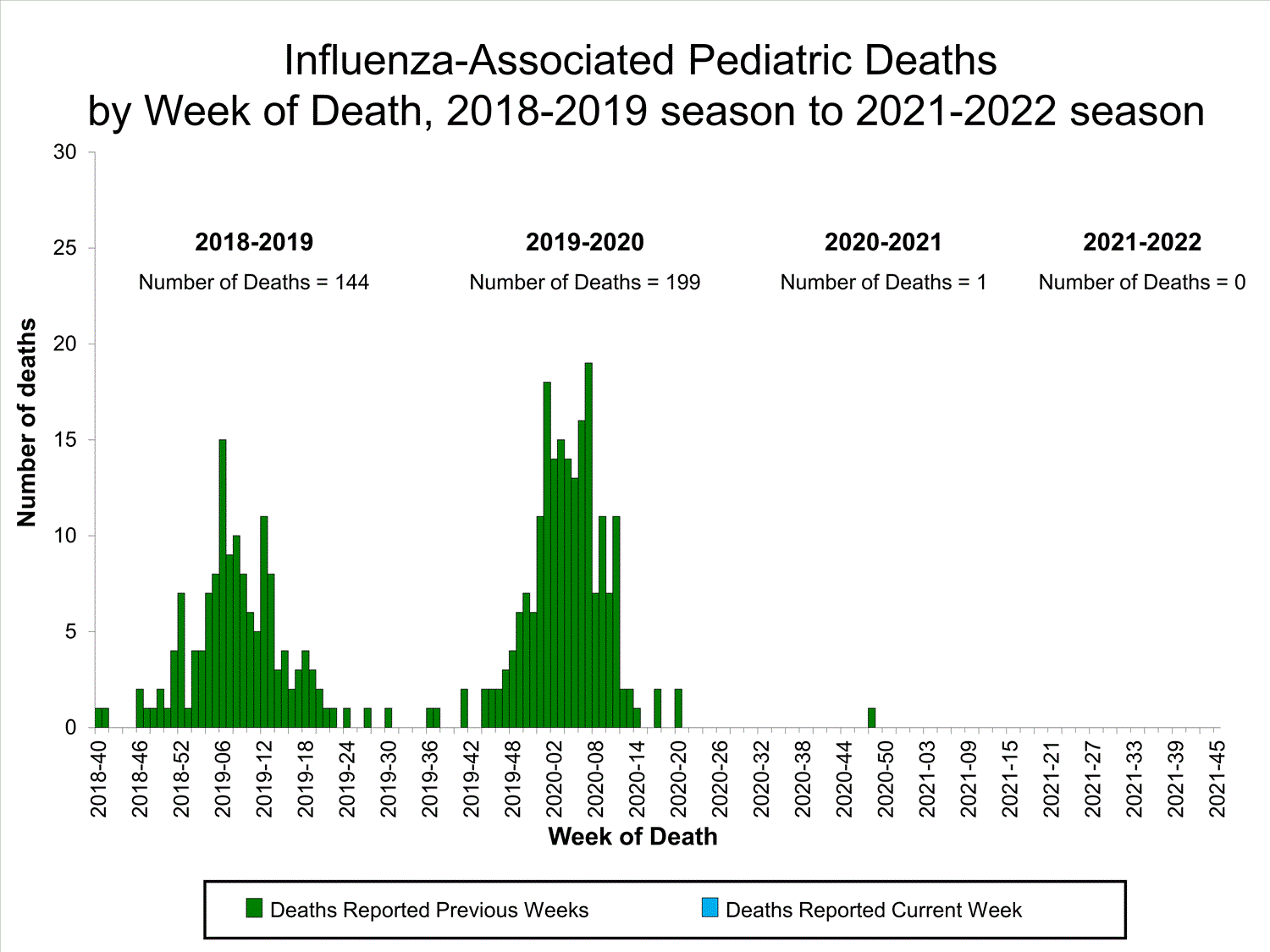
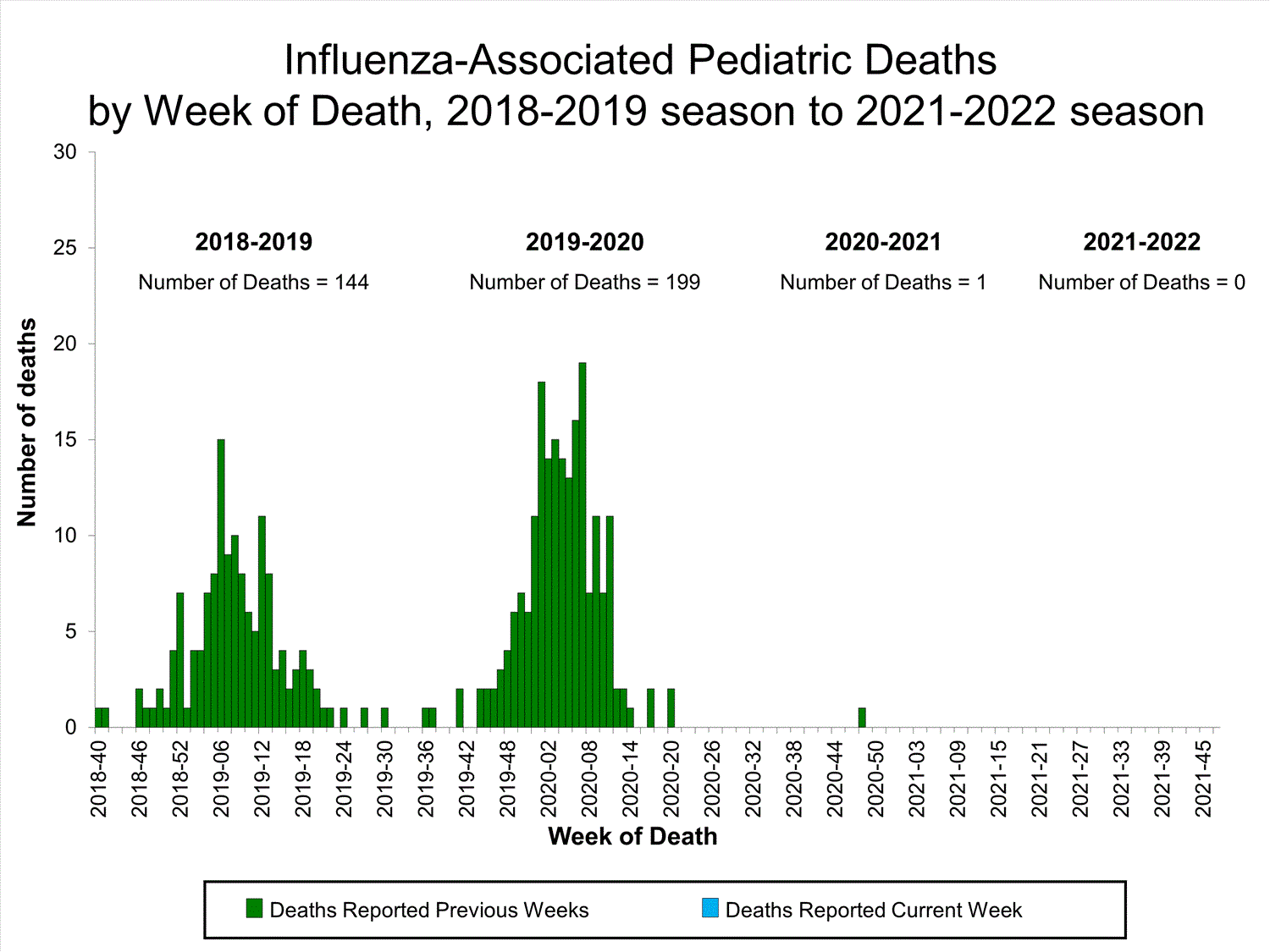
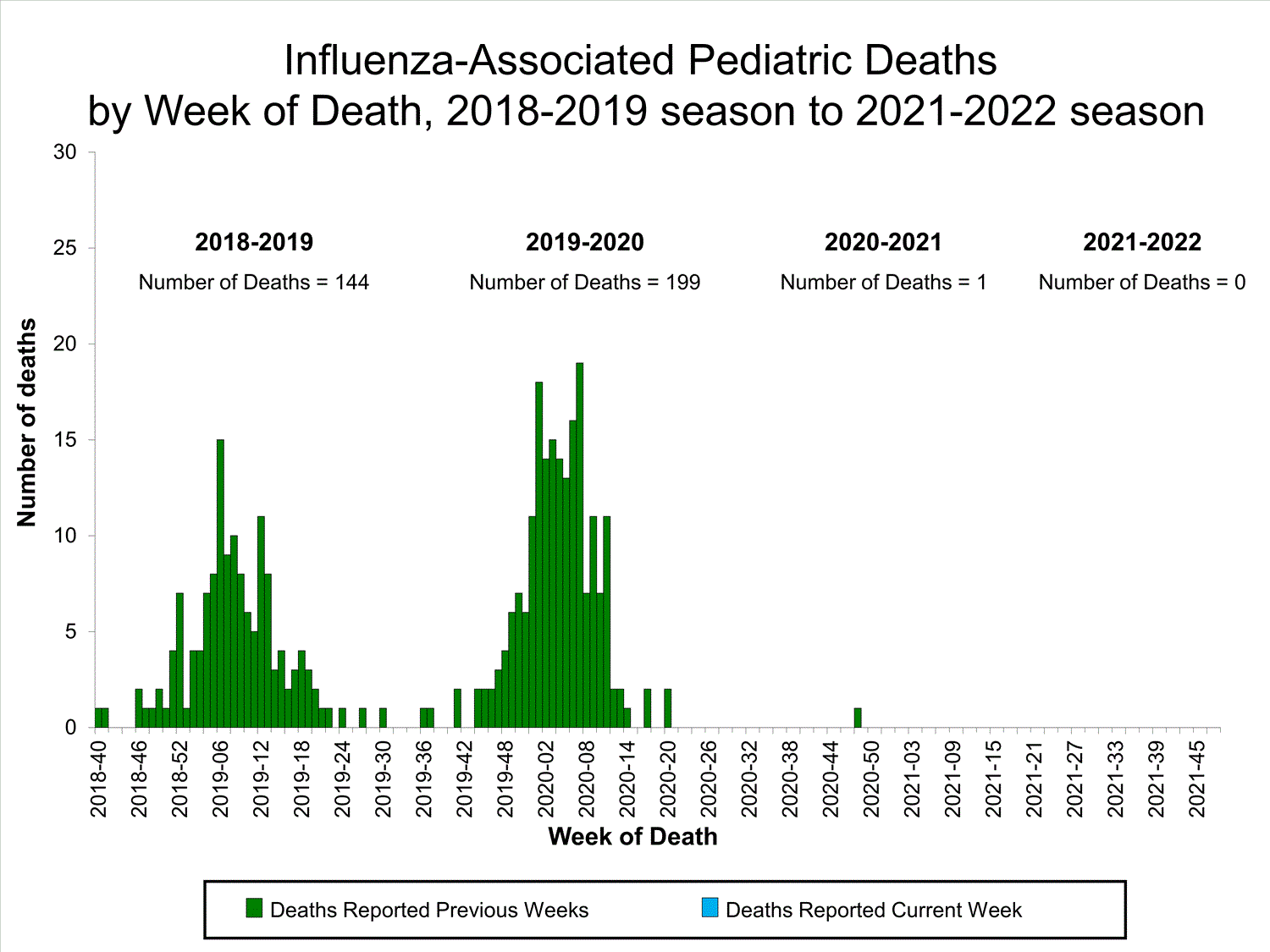
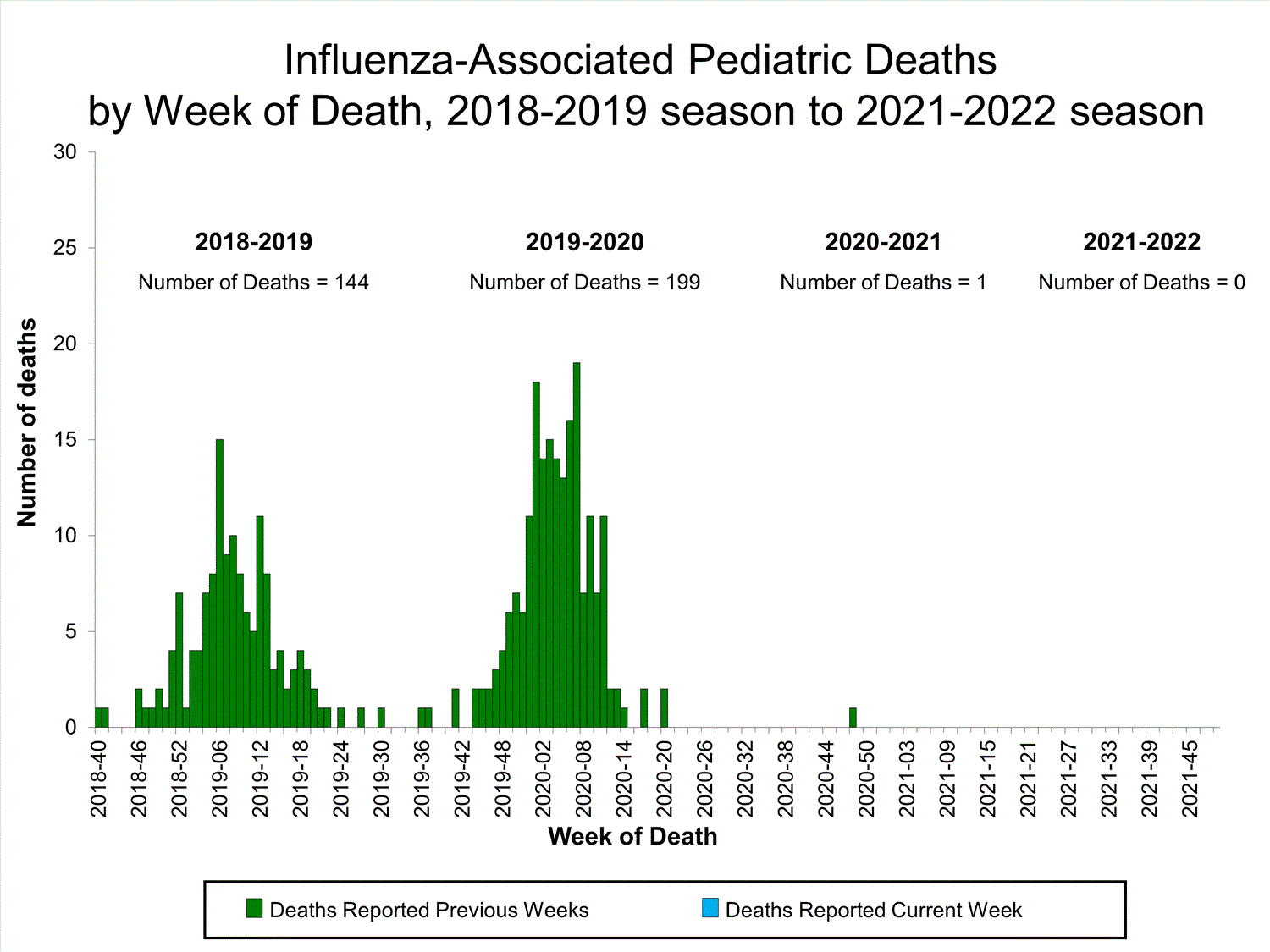
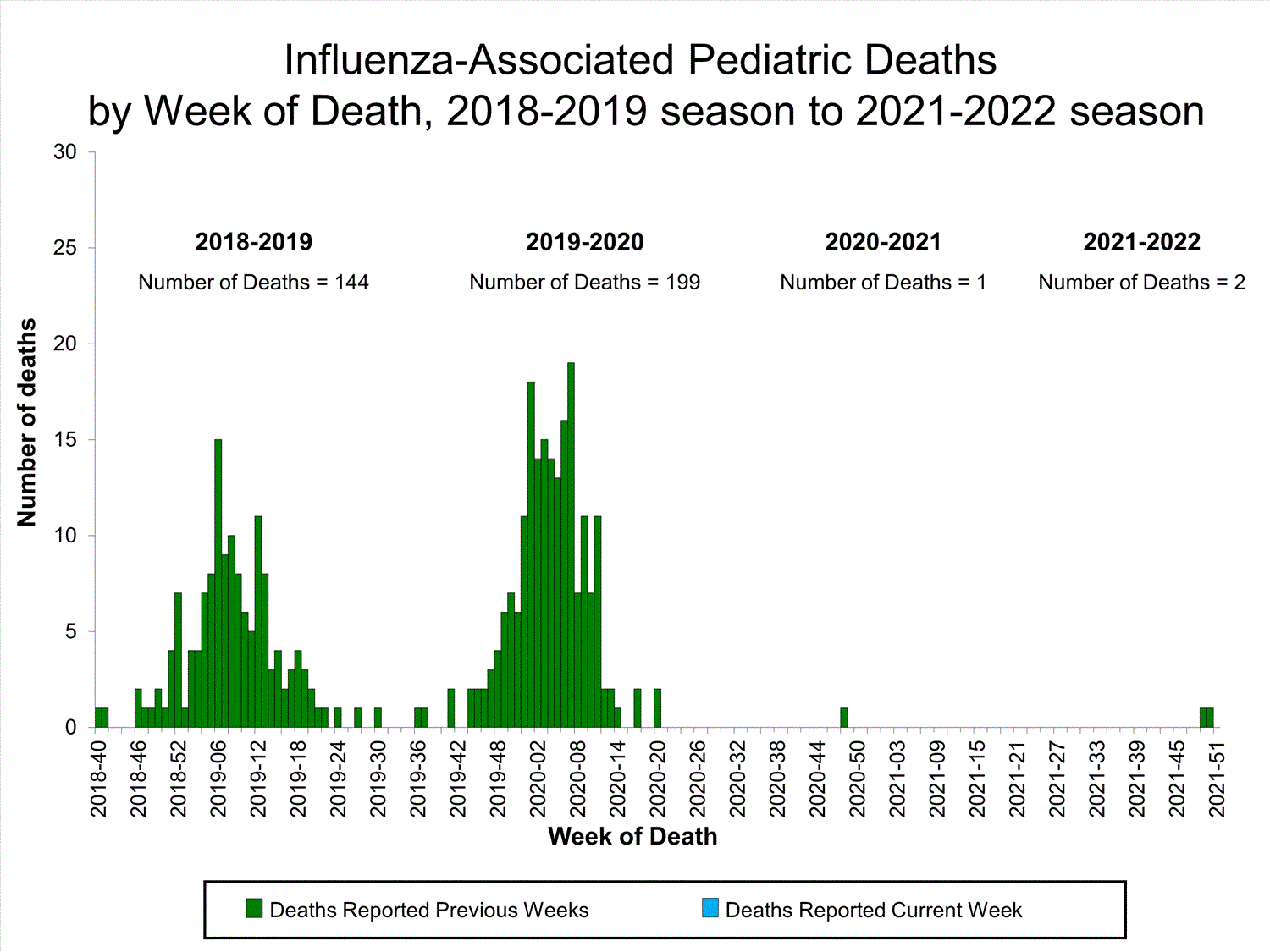
Influenza-Associated Pediatric Mortality
No influenza-associated pediatric deaths were reported to CDC during week 39.
One influenza-associated pediatric death occurring during the 2020-2021 season has been reported to CDC.
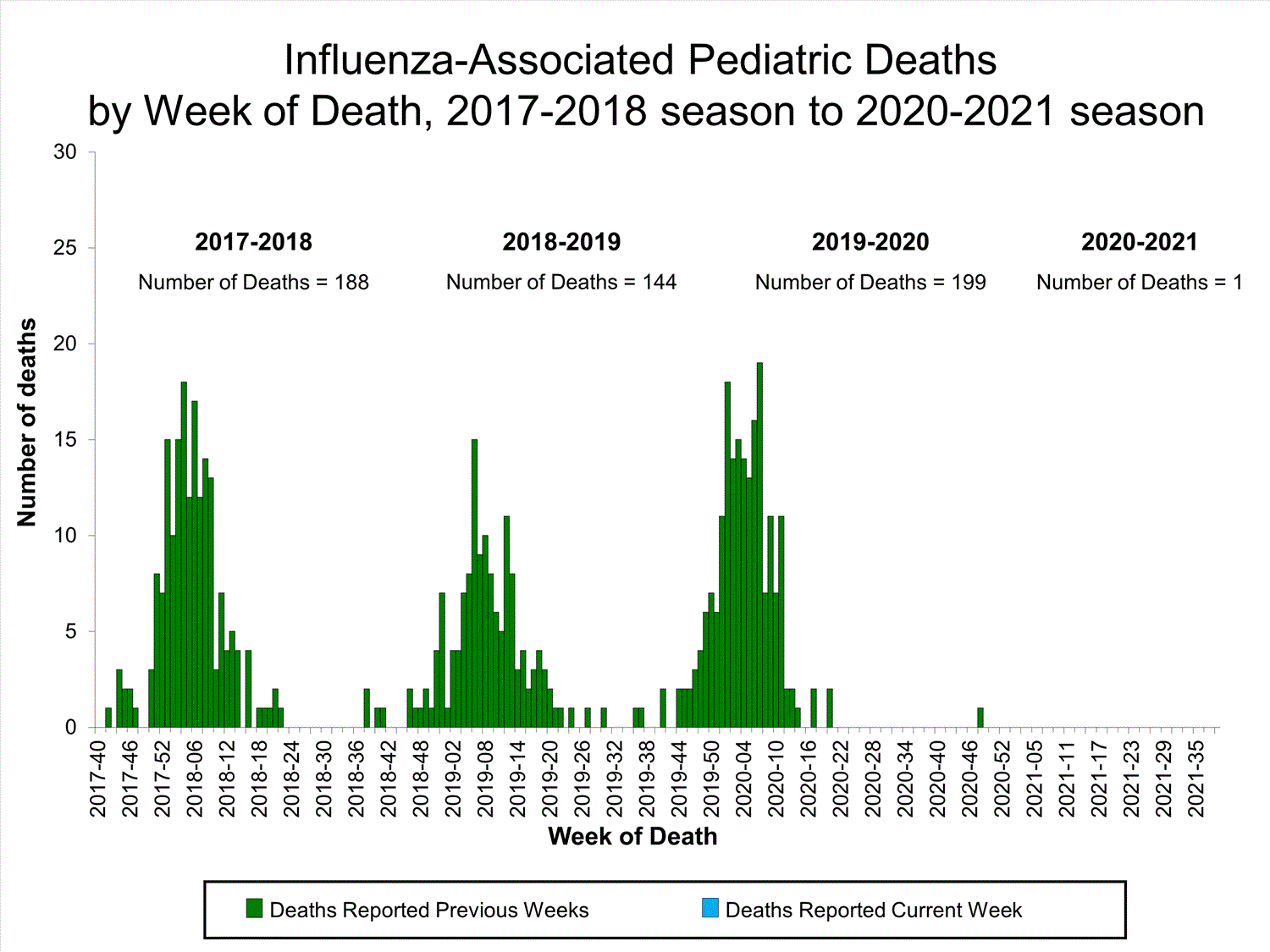 View Full Screen
View Full Screen
Additional pediatric mortality surveillance information for current and past seasons:
Surveillance Methods | FluView Interactive
Additional National and International Influenza Surveillance Information
FluView Interactive: FluView includes enhanced web-based interactive applications that can provide dynamic visuals of the influenza data collected and analyzed by CDC. These FluView Interactive applications allow people to create customized, visual interpretations of influenza data, as well as make comparisons across flu seasons, regions, age groups and a variety of other demographics.
National Institute for Occupational Safety and Health: Monthly surveillance data on the prevalence of health-related workplace absenteeism among full-time workers in the United States are available from NIOSH.
U.S. State and local influenza surveillance: Select a jurisdiction below to access the latest local influenza information.
Additional influenza surveillance information from participating WHO member nations is available through
FluNetexternal icon and the Global Epidemiology Reports.external icon
WHO Collaborating Centers for Influenza:
Australiaexternal icon, Chinaexternal icon, Japanexternal icon, the United Kingdomexternal icon, and the United States (CDC in Atlanta, Georgia)
Europe:
The most up-to-date influenza information from Europe is available from WHO/Europe and the European Centre for Disease Prevention and Controlexternal icon.
Public Health Agency of Canada:
The most up-to-date influenza information from Canada is available in Canada’s weekly FluWatch reportexternal icon.
Public Health England:
The most up-to-date influenza information from the United Kingdom is available from Public Health Englandexternal icon.
Any links provided to non-Federal organizations are provided solely as a service to our users. These links do not constitute an endorsement of these organizations or their programs by CDC or the Federal Government, and none should be inferred. CDC is not responsible for the content of the individual organization web pages found at these links.
A description of the CDC influenza surveillance system, including methodology and detailed descriptions of each data component is available on the surveillance methods page.
Page last reviewed: October 8, 2021, 11:00 AM
Weekly U.S. Influenza Surveillance Report

Note: CDC is tracking the COVID-19 pandemic in a weekly publication called COVID Data Tracker Weekly Review.
2020-2021 Influenza Season for Week 39, ending October 2, 2021
All data are preliminary and may change as more reports are received.
A description of the CDC influenza surveillance system, including methodology and detailed descriptions of each data component is available on the surveillance methods page.
Additional information on the current and previous influenza seasons for each surveillance component are available on FluView Interactive.
U.S. Virologic Surveillance:
Clinical Laboratories
Data from clinical laboratories (the percentage of specimens tested that are positive for influenza) are used to monitor whether influenza activity is increasing or decreasing.

View Chart Data | View Full ScreenPublic Health Laboratories
Data from public health laboratories are used to monitor the proportion of circulating viruses that belong to each influenza subtype/lineage.

View Chart Data | View Full Screen
Additional information about medically attended visits for ILI for current and past seasons:
Surveillance Methods | FluView Interactive: National, Regional, and State Data or ILI Activity Map
Outpatient Illness Surveillance
Please note, the U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet) monitors outpatient visits for influenza-like illness (ILI), not laboratory-confirmed influenza, and will capture visits due to other respiratory pathogens, such as SARS-CoV-2, that present with similar symptoms. In addition, health care-seeking behaviors have changed dramatically during the COVID-19 pandemic. Many people are accessing the health care system in alternative settings, which may or may not be captured as a part of ILINet. Therefore, ILI data, including ILI activity levels, should be interpreted with caution. It is particularly important at this time to evaluate syndromic surveillance data, including that from ILINet, in the context of other sources of surveillance data to obtain a complete and accurate picture of influenza, COVID-19, and other respiratory virus activity. CDC is tracking the COVID-19 pandemic in a weekly publication called COVID Data Tracker Weekly Review. Information about other respiratory virus activity can be found on CDC’s National Respiratory and Enteric Virus Surveillance System (NREVSS) website.
ILINet
Nationwide during week 39, 1.9% of patient visits reported through ILINet were due to ILI. The percentage of patient visits for ILI remains below the baseline of 2.6% nationally. All ten regions are below their region-specific baselines.
Influenza virus circulation remains low; therefore, increases in ILI activity are likely due to increased circulation of other respiratory viruses.

View Chart Data (current season only) | View Full ScreenILI Visits by Age Group
About 65% of ILINet participants provide both the number of patient visits for ILI and the total number of patient visits for the week broken out by age group. Data from this subset of providers are used to calculate the percentages of patient visits for ILI by age group.
The percentages of visits for ILI reported in ILINet are decreasing among all age groups (0-4 years, 5-24 years, 25-49 years, 50-64 years, and 65+ years).

View Chart Data | View Full ScreenILI Activity Map
Data collected in ILINet are used to produce a measure of ILI activity* by state/jurisdiction and Core Based Statistical Areas (CBSA).
| Week 39 (Week ending Oct. 2, 2021) |
Week 38 (Week ending Sep. 25, 2021) |
Week 39 (Week ending Oct. 2, 2021) |
Week 38 (Week ending Sep. 25, 2021) |
|
| Very High | 0 | 0 | 0 | 0 |
| High | 0 | 1 | 7 | 7 |
| Moderate | 1 | 0 | 16 | 32 |
| Low | 10 | 10 | 74 | 71 |
| Minimal | 42 | 44 | 474 | 495 |
| Insufficient Data | 2 | 0 | 358 | 324 |
*Data collected in ILINet may disproportionally represent certain populations within a jurisdiction or CBSA, and therefore, may not accurately depict the full picture of influenza activity for the entire jurisdiction or CBSA. Differences in the data presented here by CDC and independently by some health departments likely represent differing levels of data completeness with data presented by the health department likely being the more complete.
Additional information about medically attended visits for ILI for current and past seasons:
Surveillance Methods | FluView Interactive: National, Regional, and State Data or ILI Activity Map
Influenza-Associated Hospitalizations:
The Influenza Hospitalization Surveillance Network (FluSurv-NET) conducts population-based surveillance for laboratory-confirmed influenza-related hospitalizations in select counties in 14 states and represents approximately 9% of the U.S. population. As in previous seasons, patients admitted for laboratory-confirmed influenza-related hospitalization after April 30, 2021, will not be included in FluSurv-NET. Data on patients admitted through April 30, 2021, will continue to be updated as additional information is received.
Additional hospitalization surveillance information for current and past seasons and additional age groups:
Surveillance Methods | FluView Interactive: Rates by Age or Patient Characteristics
National Center for Health Statistics (NCHS) Mortality Surveillance
Based on NCHS mortality surveillance data available on October 7, 2021, 18.7% of the deaths that occurred during the week ending October 2, 2021 (week 39), were due to pneumonia, influenza, and/or COVID-19 (PIC). This percentage is above the epidemic threshold of 5.7% for this week. Among the 3,296 PIC deaths reported for this week, 2,607 had COVID-19 listed as an underlying or contributing cause of death on the death certificate, and four listed influenza, indicating that current PIC mortality is due primarily to COVID-19 and not influenza. The data presented are preliminary and may change as more data are received and processed.

View Chart Dataexcel icon | View Full Screen
Additional pneumonia and influenza mortality surveillance information for current and past seasons:
Surveillance Methods | FluView Interactive
Influenza-Associated Pediatric Mortality
No influenza-associated pediatric deaths were reported to CDC during week 39.
One influenza-associated pediatric death occurring during the 2020-2021 season has been reported to CDC.
 View Full Screen
View Full Screen Additional pediatric mortality surveillance information for current and past seasons:
Surveillance Methods | FluView Interactive
Additional National and International Influenza Surveillance Information
FluView Interactive: FluView includes enhanced web-based interactive applications that can provide dynamic visuals of the influenza data collected and analyzed by CDC. These FluView Interactive applications allow people to create customized, visual interpretations of influenza data, as well as make comparisons across flu seasons, regions, age groups and a variety of other demographics.
National Institute for Occupational Safety and Health: Monthly surveillance data on the prevalence of health-related workplace absenteeism among full-time workers in the United States are available from NIOSH.
U.S. State and local influenza surveillance: Select a jurisdiction below to access the latest local influenza information.
World Health Organization:
Additional influenza surveillance information from participating WHO member nations is available through
FluNetexternal icon and the Global Epidemiology Reports.external icon
WHO Collaborating Centers for Influenza:
Australiaexternal icon, Chinaexternal icon, Japanexternal icon, the United Kingdomexternal icon, and the United States (CDC in Atlanta, Georgia)
Europe:
The most up-to-date influenza information from Europe is available from WHO/Europe and the European Centre for Disease Prevention and Controlexternal icon.
Public Health Agency of Canada:
The most up-to-date influenza information from Canada is available in Canada’s weekly FluWatch reportexternal icon.
Public Health England:
The most up-to-date influenza information from the United Kingdom is available from Public Health Englandexternal icon.
Any links provided to non-Federal organizations are provided solely as a service to our users. These links do not constitute an endorsement of these organizations or their programs by CDC or the Federal Government, and none should be inferred. CDC is not responsible for the content of the individual organization web pages found at these links.
A description of the CDC influenza surveillance system, including methodology and detailed descriptions of each data component is available on the surveillance methods page.
Page last reviewed: October 8, 2021, 11:00 AM

Comment