French MOH Statement On New COVID Variant Under Investigation
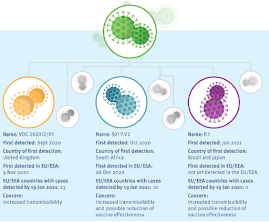
Mutation of SARS-CoV2 - current variants of concern - ECDC
#15,868
There are literally thousands of COVID variants, and the vast majority of them are of no greater consequence than the `wild type' COVID which dominated most of last year.
Late in 2020, however, several emerging variants emerged around the globe that appear to provide some biological advantages to the virus.
The three of greatest concern are B.1.1.7 - which is believed more transmissible and 30%-70% deadlier - and the P.1 and B.1.351 variants which are believed more transmissible, and have been linked to reduced antibody recognition, which in turn may increase reinfection risks and lower vaccine effectiveness.
There are other `lesser' variants under investigation (VUIs) around the globe, but at this time little is known about how much of a threat they may (or may not) pose (see UK PHE Now Tracking 9th Variant (VUI 202103/01) & Update On P.1 Variants).
Late yesterday the French Ministry of Health announced they were investigating a new variant, which - for reasons not fully explained - appears to elude some PCR tests.
At this time, there is no data to suggest this variant is more transmissible, or more severe, or evades current vaccines. It is, however, very early into their investigation.
First the press release, after which I'll have a bit more.
Following the identification by sequencing, in Brittany, of a new variant (derived from Clade 20C), the health authorities put in place the necessary monitoring and support measures.
published on 03.15.21
A new variant (derived from Clade 20C) has been detected in the context of a cluster at the Lannion hospital center, in the C?tes d'Armor. On March 13, 79 cases were identified, including 8 cases carrying the variant, confirmed by sequencing.
Based on the preliminary analysis of the mutations of this variant and the clinical and biological observations of carrier patients, the health authorities consider that it is reasonable to classify this variant under surveillance, that is to say in the VUI category. (variant under investigation) of the WHO, in particular due to the observation of several patients presenting typical symptoms suggesting an infection by SARS-CoV-2 but a negative RT-PCR test result on nasopharyngeal samples and for whom the diagnosis could be made by serology or by performing RT-PCR on deep respiratory samples.
The first analyzes of this new variant do not allow to conclude either an increased severity or transmissibility compared to the historical virus. In-depth investigations are underway in order to better understand this variant and its impact. Experiments will also take place to determine how this variant reacts to vaccination and to the antibodies developed during previous infections. Finally, the international authorities are informed of the discovery of this variant.
Viruses are constantly evolving through mutations in their genome. Specifically concerning the VUI variant derived from Clade 20C, a system for detecting and monitoring possible cases of infection or carriage has been set up by Public Health France and the National Reference Centers, in conjunction with the analysis laboratories. This system concerns a geographical area including Lannion, Guingamp, Saint-Brieuc and Morlaix. The general directorate of health has also warned all health establishments and health professionals in order to make them aware of the monitoring of this variant.
Finally, as a precaution, the local authorities, prefectures and ARS, are stepping up measures to curb the transmission of the virus , for example by speeding up vaccination, by reminding people of the importance of barrier gestures or by limiting gatherings.
PDFPress release | Following identification by sequencing, in (...)Download (216.5 kb)
DOCXPress release | Following identification by sequencing, in (...) Download (38.9 kb)
(Continue . . . )
This isn't the first time we've seen reports of a COVID variant eluding PCR testing. A little over 3 weeks ago, in What (Little) We Currently Know About the Fin-796H Variant, we looked at similar reports from Finland's Ministry of Health, albeit on a single patient.
We've seen a similar situation with the S-gene target failure (SGTF) on the B.1.1.7 variant with the Thermo Fisher Scientific TaqPath COVID-19 PCR test, but in that case, it actually has helped in the identification of potential variants.
Since different PCR tests target different parts of the virus, a variant that eludes one PCR test might not elude tests from other manufacturers. It isn't clear from this report if this is a problem with just one PCR test, or if it affects multiple brands.
This statement does indicate that the diagnosis could be made by `serology or by performing RT-PCR on deep respiratory samples', but that tests on several nasopharyngeal samples were negative. Whether that indicates a change in viral shedding isn't immediate apparent.
The first priority will be to improve testing procedures, in order to capture as many cases as possible. Only then can authorities begin to understand how widespread this variant might be, and what its other qualities might be.
Stay tuned.

Mutation of SARS-CoV2 - current variants of concern - ECDC
#15,868
There are literally thousands of COVID variants, and the vast majority of them are of no greater consequence than the `wild type' COVID which dominated most of last year.
Late in 2020, however, several emerging variants emerged around the globe that appear to provide some biological advantages to the virus.
The three of greatest concern are B.1.1.7 - which is believed more transmissible and 30%-70% deadlier - and the P.1 and B.1.351 variants which are believed more transmissible, and have been linked to reduced antibody recognition, which in turn may increase reinfection risks and lower vaccine effectiveness.
There are other `lesser' variants under investigation (VUIs) around the globe, but at this time little is known about how much of a threat they may (or may not) pose (see UK PHE Now Tracking 9th Variant (VUI 202103/01) & Update On P.1 Variants).
Late yesterday the French Ministry of Health announced they were investigating a new variant, which - for reasons not fully explained - appears to elude some PCR tests.
At this time, there is no data to suggest this variant is more transmissible, or more severe, or evades current vaccines. It is, however, very early into their investigation.
First the press release, after which I'll have a bit more.
Following the identification by sequencing, in Brittany, of a new variant (derived from Clade 20C), the health authorities put in place the necessary monitoring and support measures.
published on 03.15.21
A new variant (derived from Clade 20C) has been detected in the context of a cluster at the Lannion hospital center, in the C?tes d'Armor. On March 13, 79 cases were identified, including 8 cases carrying the variant, confirmed by sequencing.
Based on the preliminary analysis of the mutations of this variant and the clinical and biological observations of carrier patients, the health authorities consider that it is reasonable to classify this variant under surveillance, that is to say in the VUI category. (variant under investigation) of the WHO, in particular due to the observation of several patients presenting typical symptoms suggesting an infection by SARS-CoV-2 but a negative RT-PCR test result on nasopharyngeal samples and for whom the diagnosis could be made by serology or by performing RT-PCR on deep respiratory samples.
The first analyzes of this new variant do not allow to conclude either an increased severity or transmissibility compared to the historical virus. In-depth investigations are underway in order to better understand this variant and its impact. Experiments will also take place to determine how this variant reacts to vaccination and to the antibodies developed during previous infections. Finally, the international authorities are informed of the discovery of this variant.
Viruses are constantly evolving through mutations in their genome. Specifically concerning the VUI variant derived from Clade 20C, a system for detecting and monitoring possible cases of infection or carriage has been set up by Public Health France and the National Reference Centers, in conjunction with the analysis laboratories. This system concerns a geographical area including Lannion, Guingamp, Saint-Brieuc and Morlaix. The general directorate of health has also warned all health establishments and health professionals in order to make them aware of the monitoring of this variant.
Finally, as a precaution, the local authorities, prefectures and ARS, are stepping up measures to curb the transmission of the virus , for example by speeding up vaccination, by reminding people of the importance of barrier gestures or by limiting gatherings.
PDFPress release | Following identification by sequencing, in (...)Download (216.5 kb)
DOCXPress release | Following identification by sequencing, in (...) Download (38.9 kb)
(Continue . . . )
This isn't the first time we've seen reports of a COVID variant eluding PCR testing. A little over 3 weeks ago, in What (Little) We Currently Know About the Fin-796H Variant, we looked at similar reports from Finland's Ministry of Health, albeit on a single patient.
We've seen a similar situation with the S-gene target failure (SGTF) on the B.1.1.7 variant with the Thermo Fisher Scientific TaqPath COVID-19 PCR test, but in that case, it actually has helped in the identification of potential variants.
Since different PCR tests target different parts of the virus, a variant that eludes one PCR test might not elude tests from other manufacturers. It isn't clear from this report if this is a problem with just one PCR test, or if it affects multiple brands.
This statement does indicate that the diagnosis could be made by `serology or by performing RT-PCR on deep respiratory samples', but that tests on several nasopharyngeal samples were negative. Whether that indicates a change in viral shedding isn't immediate apparent.
The first priority will be to improve testing procedures, in order to capture as many cases as possible. Only then can authorities begin to understand how widespread this variant might be, and what its other qualities might be.
Stay tuned.
